Impact Factor ISSN: 1449-1907
Int J Med Sci 2022; 19(1):47-64. doi:10.7150/ijms.58902 This issue Cite
Research Paper
G6PD upregulates Cyclin E1 and MMP9 to promote clear cell renal cell carcinoma progression
1. Department of Biochemistry and Molecular Biology, School of Basic Medical Sciences, Kunming Medical University, Yunnan, Kunming 650500, P.R. China.
2. Departments of Pathology, The First Affiliated Hospital of Kunming Medical University, Yunnan, Kunming 650032, P.R. China.
3. Department of Clinical Laboratory, The Second Hospital of Jingzhou, Jingzhou, Hubei 434000, P.R. China.
4. Department of Medical Oncology, The Third Affiliated Hospital of Kunming Medical University (Tumor Hospital of Yunnan Province), Yunnan, Kunming 650118, P.R. China.
5. Department of Clinical Laboratory, The Third Affiliated Hospital of Kunming Medical University (Tumor Hospital of Yunnan Province), Yunnan, Kunming 650118, P.R. China.
6. Departments of Organ Transplantation, The First Affiliated Hospital of Kunming Medical University, Yunnan, Kunming 650032, P.R. China.
# Current address: Department of Biochemistry and Biophysics, School of Basic Medical Sciences, Beijing Key Laboratory of Protein Posttranslational Modifications and Cell Function, Peking University Health Science Center, Beijing 100191, P.R. China.
* These authors contributed equally.
Abstract
Background: Clear cell renal cell carcinoma (ccRCC) is a cell metabolic disease with high metastasis rate and poor prognosis. Our previous studies demonstrate that glucose-6-phosphate dehydrogenase (G6PD), the first and rate-limiting enzyme of the pentose phosphate pathway, is highly expressed in ccRCC and predicts poor outcomes of ccRCC patients. The aims of this study were to confirm the oncogenic role of G6PD in ccRCC and unravels novel mechanisms involving Cyclin E1 and MMP9 in G6PD-mediated ccRCC progression.
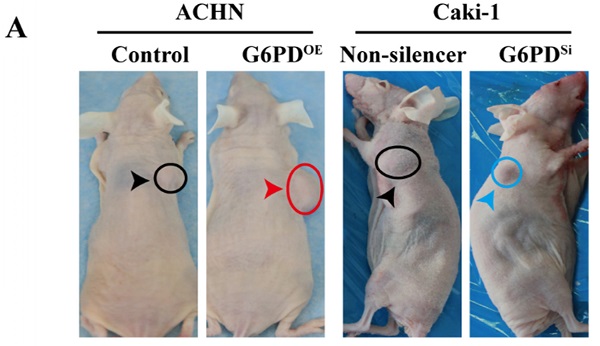
Methods: Real-time RT-PCR, Western blot and immunohistochemistry were used to determine the expression patterns of G6PD, Cyclin E1 and MMP9 in ccRCC. TCGA dataset mining was used to identify Cyclin E1 and MMP9 correlations with G6PD expression, relationships between clinicopathological characteristics of ccRCC and the genes of interest, as well as the prognosis of ccRCC patients. The role of G6PD in ccRCC progression and the regulatory effect of G6PD on Cyclin E1 and MMP9 expression were investigated by using a series of cytological function assays in vitro. To verify this mechanism in vivo, xenografted mice models were established.
Results: G6PD, Cyclin E1 and MMP9 were overexpressed and positively correlated in ccRCC, and they were associated with poor prognosis of ccRCC patients. Moreover, G6PD changed cell cycle dynamics, facilitated cells proliferation, promoted migration in vitro, and enhanced ccRCC development in vivo, more likely through enhancing Cyclin E1 and MMP9 expression.
Conclusion: These findings present G6PD, Cyclin E1 and MMP9, which contribute to ccRCC progression, as novel biomarkers and potential therapeutic targets for ccRCC treatment.
Keywords: ccRCC, G6PD, Cyclin E1, MMP9, proliferation, migration

 Global reach, higher impact
Global reach, higher impact