Impact Factor
ISSN: 1449-1907
Int J Med Sci 2021; 18(14):3249-3260. doi:10.7150/ijms.61510 This issue Cite
Research Paper
Therapeutic Antitumor Efficacy of Cancer Stem Cell-Derived DRibble Vaccine on Colorectal Carcinoma
1. Department of Regenerative Medicine, School of Pharmaceutical Sciences, Jilin University, Changchun, Jilin 130021, China
2. Stanford University Medical School, VA Palo Alto Health Care System, Palo Alto, CA 94304, USA,
3. Institute of Oceanography, Minjiang University, Fuzhou, Fujian 350108, China
4. Medical Institute of Regeneration Sciences, Jilin University, Changchun, Jilin 130021, China.
* Equal contributions
Abstract
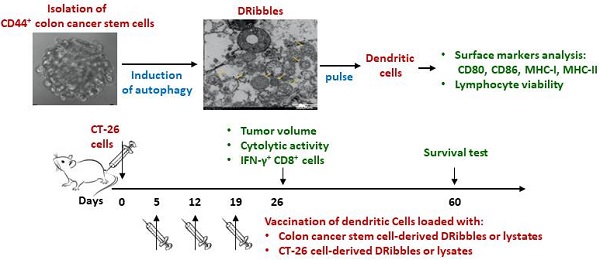
Dendritic cell (DC)-based immunotherapy has been a promising strategy for colon cancer therapy, but the efficacy of dendritic cell vaccines is in part limited by immunogenicity of loaded antigens. In this study, we aimed to identify a putative tumor antigen that can generate or enhance anti-tumor immune responses against colon cancer. CD44+ colon cancer stem cells (CCSCs) were isolated from mouse colorectal carcinoma CT-26 cell cultures and induced to form defective ribosomal products-containing autophagosome-rich blebs (DRibbles) by treatment with rapamycin, bortezomib, and ammonium chloride. DRibbles were characterized by western blot and transmission electron microscopy. DCs generated from the mice bone marrow monocytes were cocultured with DRibbles, then surface markers of DCs were analyzed by flow cytometry. Meanwhile, the efficacy of DRibble-DCs was examined in vivo. Our results showed that CCSC-derived DRibbles upregulated CD80, CD86, major histocompatibility complex (MHC)-I, and MHC-II on DCs and induced proliferation of mouse splenic lymphocytes and CD8+ T cells. In a model of colorectal carcinoma using BALB/c mice with robust tumor growth and mortality, DC vaccine pulsed with CCSC-derived DRibbles suppressed tumor growth and extended survival. A lactate dehydrogenase test indicated a strong cytolytic activity of cytotoxic T-cells derived from mice vaccinated with CCSC-derived DRibbles against CT-26 cells. Furthermore, flow cytometry analyses showed that the percentages of IFN-γ-producing CD8+ T-cells were increased in SD-DC group compare with the other groups. These findings provide a rationale for novel immunotherapeutic anti-tumor approaches based on DRibbles derived from colon cancer stem cells.
Keywords: DRips-Containing Blebs (DRibbles), Dendritic Cells, Cancer Stem Cells, Autophagosome, Colorectal Cancer

 Global reach, higher impact
Global reach, higher impact