Impact Factor ISSN: 1449-1907
Int J Med Sci 2020; 17(15):2328-2337. doi:10.7150/ijms.48872 This issue Cite
Research Paper
Downregulation of Cypher induces apoptosis in cardiomyocytes via Akt/p38 MAPK signaling pathway
1. Department of Cardiology, The First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China.
2. Department of Pathology and Pathophysiology, Zhejiang University School of Medicine, Hangzhou, China.
*These authors have equally contributed to this article as co-first authors.
Received 2020-5-30; Accepted 2020-8-17; Published 2020-8-27
Abstract
Background: Dilated cardiomyopathy (DCM) is considered as the most common form of non-ischemic cardiomyopathy with a high mortality worldwide. Cytoskeleton protein Cypher plays an important role in maintaining cardiac function. Genetic studies in human and animal models revealed that Cypher is involved in the development of DCM. However, the underlying molecular mechanism is not fully understood. Accumulating evidences suggest that apoptosis in myocytes may contribute to DCM. Thus, the purpose of this study is to define whether lack of Cypher in cardiomyocytes can elevate apoptosis signaling and lead to DCM eventually.
Methods and Results: Cypher-siRNA sufficiently inhibited Cypher expression in cardiomyocytes. TUNEL-positive cardiomyocytes were increased in both Cypher knockdown neonatal rat cardiomyocytes and Cypher knockout mice hearts, which were rare in the control group. Flow cytometry further confirmed that downregulation of Cypher significantly increased myocytes apoptosis in vitro. Cell counting kit-8 assay revealed that Cypher knockdown in H9c2 cells significantly reduced cell viability. Cypher knockdown was found to increase cleaved caspase-3 expression and suppress p21, ratio of bcl-2 to Bax. Cypher-deficiency induced apoptosis was linked to downregulation of Akt activation and elevated p-p38 MAPK accumulation. Pharmacological activation of Akt with SC79 attenuated apoptosis with enhanced phosphorylation of Akt and reduced p-p38 MAPK and Bax expression.
Conclusions: Downregulation of Cypher participates in the promotion of cardiomyocytes apoptosis through inhibiting Akt dependent pathway and enhancing p38 MAPK phosphorylation. These findings may provide a new potential therapeutic strategy for the treatment of DCM.
Keywords: apoptosis, Cypher, dilated cardiomyopathy, Akt, p38 MAPK
Introduction
Cypher/ZASP, encoded by LIM domain binding 3 (LDB3), is a cytoskeletal PDZ-LIM protein predominantly expressed in cardiac and skeletal muscle [1]. It is localized at the Z-line by directly complexing with α-actinin-2 through its PDZ domain to maintain sarcomere integrity during muscle contraction [2]. And the LIM domains are involved in the binding to a network of proteins such as protein kinase acting as a signaling center [3]. Genetic studies have revealed that mutations in the LDB3 gene are shown to be associated with human myopathies such as skeletal myopathy, isolated non-compaction of the left ventricular myocardium, hypertrophic cardiomyopathy and dilated cardiomyopathy (DCM) [4]. Moreover, the LDB3 gene, mapped on chromosome 10q22.3-10q23.2, overlaps with a DCM locus [5]. DCM, the most common cause of congestive heart failure, is a cardiac muscle disease characterized by left ventricular dilation with systolic dysfunction which often necessitates cardiac transplantation [6]. Although the pathogenesis of DCM are highly heterogeneous, up to 35% of cases are caused by mutations, mainly in cytoskeletal and sarcomeric genes [7]. Among them, Cypher/ZASP plays a critical role in the pathogenesis of DCM in both human and animal models [8-11].
In zebrafish, knockdown of Cypher results in a DCM phenotype which is characterized by cardiac dilation alongside with significant thinning of the ventricular wall [9]. In mice, both global and cardiac-restricted Cypher knockout induce a severe form of DCM with heart failure, which lead to premature lethality [10]. A similar phenotype is observed in a transgenic mouse model (S196L) [11]. Moreover, Cypher deficiency induced DCM in mouse models leads to abnormalities of multiple signaling pathways. For example, calcineurin signaling is overactivated which is a Ser/Thr phosphatase of the L-type calcium channel (LTCC) in Cypher deficient mouse [12,13]. Cypher also can tether protein kinase A (PKA) to the LTCC and phosphorylate Ser1928 through specifically interacting with PKA, termed an AKAP [13]. The mechanosensing protein Ankrd2 and the nuclear phosphoprotein p53 are another two binding partners of Cypher [14]. And they also found that the Cypher acts as a negative regulator to decrease the p53-mediated activation of Bax and MDM2 promoters [14]. The DCM related D626N mutation of Cypher/ZASP increases its affinity to protein kinase C (PKC), further suggesting that Cypher/ZASP plays an important signaling role to maintain cardiac function [15].
Accumulating studies using zebrafish, mouse, and human genetics have revealed a pivotal role of Cypher/ZASP for maintaining cardiac structure and function. However, the molecular mechanisms underlying the deficiencies in Cypher/ZASP resulting in DCM are still poorly understood. Further investigations are essential for providing initial insights into the pathophysiology for Cypher/ZASP related DCM as well as developing new therapeutic strategies to prevent progression and mortality due to DCM. Therefore, we investigated whether DCM induced by cypher-deficiency is mediated by apoptosis which plays an important role in the pathogenesis of DCM.
Materials and Methods
Experimental animals
All procedures conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication no. 85-23, revised 1996) and were approved by the ethics committee of the First Affiliated Hospital, School of Medicine, Zhejiang University. C57BL/6J global Cypher knockout mice were a generous gift of Dr. Cheng. All mice were housed and bred in an accredited facility, which was maintained at 20-25°C, 55% humidity, with a 12/12 hrs light/dark cycle. Mice were sacrificed at birth and the hearts were collected.
Neonatal rat cardiomyocytes (NRVCM) culture
Neonatal rat cardiomyocytes were prepared from 1-3 days old Sprague-Dawley rats bred at Zhejiang Academy of Medical Sciences by adapting previously published protocols [16].
Cell culture
HEK293T cell line and H9c2 cell line were purchased from Chinese Academy of Sciences. Cells were cultured in high glucose DMEM supplemented with 10% FBS, 100 U/mL of penicillin/streptomycin. Cells were incubated at 37°C in the presence of 5% CO2. For some groups, the H9c2 cells were treated with Akt agonists SC79 (HY-18749, 10 mM, 30 mins, MCE), and then the cells were harvested in 30 mins for Western blot analysis.
siRNA transfection
siRNA transfections were performed with Lipofectamine RNAiMAX reagent (Invitrogen, USA) following the manufacturer's instructions. The siRNA to lipofectamine RNAiMAX reagent concentration used was 10 pmol to 7.5 µL. After incubation for 48 hrs, the H9c2 cells or primary neonatal rat cardiomyocytes were used for subsequent experiments. The siRNA targeting rat Cypher (5'-GGAACAGCCUCUUCCACAUTT-3') or nontargeting scrambled siRNA (5'-UUCUCCGAACGUGUCACGUTT-3') were synthesized by GenePharma (Shanghai, China).
TdT-mediated dUTP nick end labeling (TUNEL) assay
TUNEL assay was performed using the In situ Cell Death Detection Kit (Roche Diagnostics, Mannheim, Germany) to detect apoptotic cells in the mouse hearts (experiments were accomplished by Wuhan Seville Biological Technology, Ltd.). Positive staining which exhibited dark brown, brownish and light yellow nuclei under a light microscope was analyzed by Quant center software. The cell apoptosis positive rate = number of apoptotic cells/number of total cells×100%. Apoptosis of neonatal rat cardiomyocytes was detected using the TUNEL FITC Apoptosis Detection kit (Beyotime) according to the manufacturer's instructions. In brief, cells were washed with PBS once and fixed in 4% paraformaldehyde for 30 mins. Cells were then treated with 0.3% Triton-X for 5 min, rinsed with PBS, and subsequently incubated with a mixture of Biotin-dUTP and the TdT enzyme in a humidified atmosphere for 60 mins at 37°C. Finally, DAPI was used to dye the nucleus. Finally, TUNEL positive cells were observed under fluorescence microscope.
Cell counting kit-8 (CCK-8) assay
Si-Cypher-transfected, si-scramble-transfected and untreated H9c2 cells were seeded into 96-well plates (1×105 cells/mL) for the CCK-8 cell proliferation assay (lot: K10183133EF5E, Beyotime). According to the manufacturer's instructions, cells were incubated with the CCK-8 reagent at 37°C for 2 hrs, with the absorbance of each sample scanned on a microplate reader equipped to read absorbance values at 450 nm.
Assessment of apoptosis by flow cytometric analysis
Cells were treated with siRNA for 48 hrs. Following treatment, cells were stained with Annexin V/PI Apoptosis Detection Kit (catalogue: 556547, BD Biosciences, USA). Briefly, after digestion with 0.25% trypsin, cells were harvested by centrifugation, washed twice with cold PBS, and then resuspended in 500 μL 1×Binding Buffer at a concentration of 1×105 cells/mL. The cells were then incubated with 5 µL Annexin V-FITC and 5 µL PI at room temperature in dark for 15 mins with gentle vortexing. Stained cells were analyzed by flow cytometry. Data analysis was performed with FlowJo 10.5.0 (FlowJo LLC). The experiment was repeated for three times.
Quantitative Real time Polymerase Chain Reaction (RT-qPCR) analysis
Total RNA was extracted from cell samples using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.). 500 ng of isolated RNA was reverse transcribed into cDNA using PrimeScript™ RT Master Mix (Takara Biotechnology Co., Ltd., China) according to the manufacturer's protocol. And RT-qPCR reaction systems were prepared using SYBR Green qPCR Master Mix (Vazyme Biotech Co. Ltd). The thermocycling conditions were: 95°C for 30 secs; followed by 39 cycles at 95°C for 10 secs and 60°C for 30 secs. Relative transcript abundance was normalized against GAPDH. The 2-ΔΔCT method was used to calculate the relative transcript level.
Western blotting
Western blotting was performed as previously described [17]. Blots were incubated overnight at 4°C with following primary antibodies: GAPDH (Cell Signaling Technology, CST), LDB3 (Abcam), p21 (Abcam), Bax (Abcam), Bad (CST), phospho-Bad (Ser112, CST), Bcl-2 (Proteintech), caspase3 (CST), cleaved caspase-3 (CST), Akt (CST), phospho-Akt (Ser473, CST), phospho-Akt (Thr308, CST), p38 MAPK (CST), phospho-p38 MAPK (Thr180/Tyr182, CST), Erk1/2 (CST), phospho-Erk1/2 (Thr202/Tyr204, CST), SAPK/JNK (#9252, CST), phospho-SAPK/JNK (Thr183/Tyr185, CST), p53 (CST), GST (Abcam), HA (EΛRTH). The bands were quantified by image J software.
Co-immunoprecipitation (Co-IP)
Plasmid expressing GST-Cypher and HA-Akt were co-transfected into HEK293T cells using Lipofectamine 2000 (Invitrogen, USA) according to the manufacturer's protocol. Cell lysates were collected 48 h post-transfection and subsequently incubated with GST-tag Cypher, HA-tag Akt, or control IgG antibodies overnight at 4°C on a rotator. Thereafter, protein A/G agarose beads were added for another 2 hrs at room temperature. The precipitates were then washed three times and boiled at 95°C for 10 mins. The eluted proteins were analyzed by western blot.
Statistical analysis
The data were expressed as means ± standard deviations (SDs). Statistical comparisons between two groups were performed with an unpaired Student's t test. Statistical analysis was performed using GraphPad Prism 7.0 (GraphPad Software Inc., San Diego, CA, USA). P values < 0.05 were considered statistically significant.
Results
Knockdown of Cypher promoted cardiomyocyte apoptosis in vivo and vitro
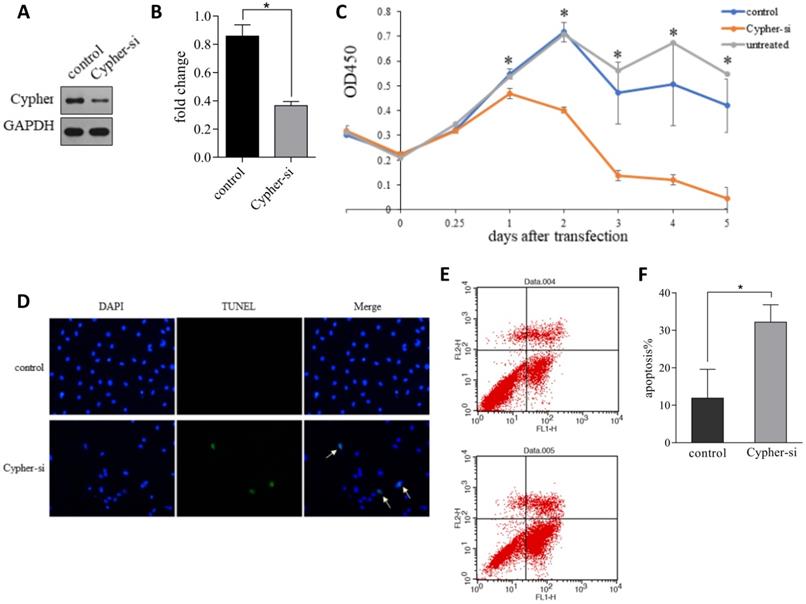
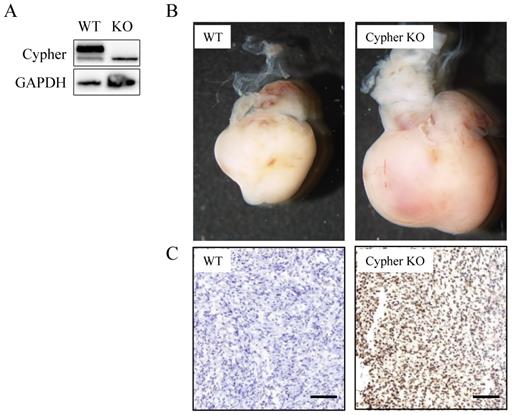
To investigate the potential role of Cypher, the siRNA-mediated specific knockdown of Cypher was applied to downregulate its expression level. Scramble siRNA, a non-functional and non-targeting siRNA, was used as the control group. And the highly transfection efficiency was confirmed by western blotting. As shown in Figure 1A and 1B, the protein levels of Cypher were significantly diminished relative to the control group (p<0.05) and a decrease in intercellular adhesion and cell number, as well as cell shrinkage, were observed in cells treated with Cypher siRNA. Thus, CCK-8 assay was used to detect cell viability. Inhibition of Cypher in H9c2 cells efficiently reduced cell viability (Fig. 1C). The effect began to appear on the first day following Cypher knockdown, and continued to deteriorate until the end of the experiment which lasted for 5 days. To determine whether deficient Cypher was involved in NRVCMs apoptosis, the TUNEL staining and flow cytometry were performed. As shown in Figure 1D, knockdown of Cypher notably increased the proportion of TUNEL positive cells compared with the controls. Quantitative analysis using flow cytometry confirmed that knockdown of Cypher significantly increased the apoptotic rate of the primary cells. As shown in Fig.1E and 1F, the number of Annexin V+ and Annexin V+/PI+ cells, standing for early and late stage of apoptosis, were increased compared with the controls. To further assess the relationship between Cypher and cardiomyocyte, Cypher knockout mice were used (Fig. 2A). Morphologically, Cypher knockout mice exhibited larger heart than wild type littermates (Fig. 2B). As shown in Figure 2C, TUNEL-negative nuclei appeared blue, and TUNEL-positive nuclei were dark brown, brownish and light yellow. A marked increase in the number of TUNEL positive nuclei was observed in cardiac tissue of the Cypher knockout mice with apoptotic indices of 0.32, more than twice the wide type. Taken together, we strongly suggested that Cypher deficiency may induce cardiomyocytes apoptosis, which eventually causes dilated cardiomyocyte.
Downregulation of Cypher induced apoptosis in vitro. (A and B) Representative western blot bands and statistical results of Cypher protein expression normalized by GAPDH. *P < 0.05 versus control cells. (C) Cell viability was measured using the CCK-8 assay. *P < 0.05 Cypher-si versus control. (D) Representative images of TUNEL staining showing the apoptotic cells (stained in green) and nucleus (stained in blue with DAPI). Arrowheads in the pictures indicate apoptotic cell nuclei. (E) Annexin V-FITC/propidium iodide (PI) staining by flow cytometry was performed to detect the apoptosis of H9c2 cells. Early apoptosis (Annexin V-FITC+/PI), late apoptosis (Annexin V-FITC+/PI+), and necrosis (Annexin V-FITC/PI+). (F) The quantitative presentation of apoptotic cell population by Annexin V-FITC/PI staining. *P < 0.05 versus control. Experiments were repeated three times.
Effects of Cypher in mice hearts. (A) Representative western blot bands of Cypher protein expression was shown. (B) The effects of Cypher knockout on heart volume. (C) Representative TUNEL staining images from hearts of wild type mice (left) and Cypher knockout mice (right), scale bar: 100 µM.
Effect of Cypher-siRNA transfection on H9c2 cell apoptosis
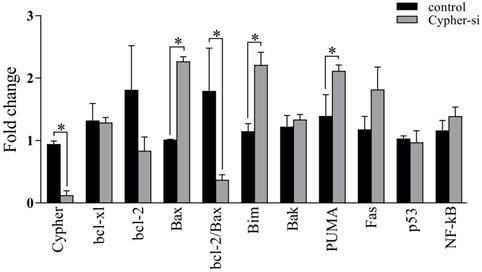
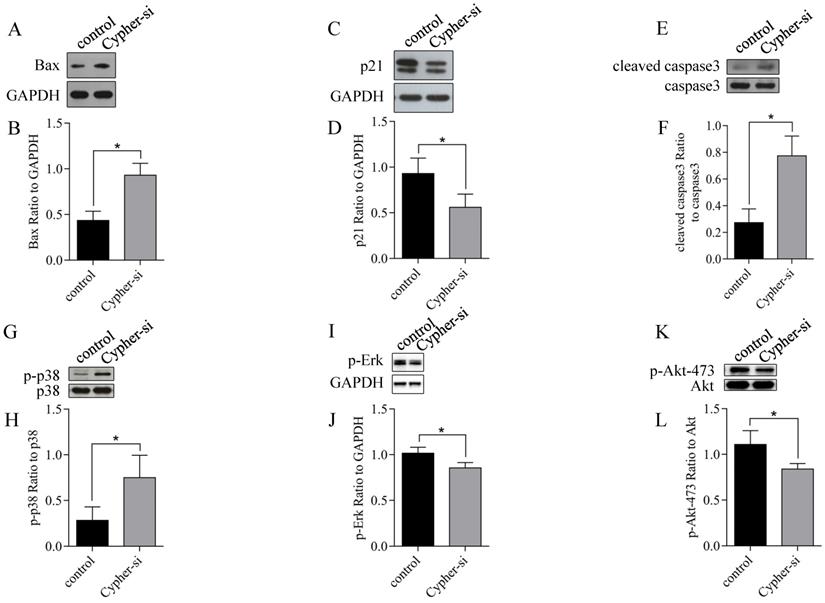
As the knockdown of Cypher induced cell apoptosis, we employed RT-qPCR and western blot analysis to identify the expression of genes related to apoptosis following the reduction in Cypher expression. The results of RT-qPCR revealed that administration of Cypher siRNA resulted in decreased expression of bcl-2 and caused upregulation of apoptosis-inducing agents such as Bim, PUMA and Bax (Fig. 3). Then, the ratio of bcl-2 to Bax expression, which is the determining factor for the induction of apoptosis, was markedly decreased in Cypher siRNA treated H9c2 cells compared to controls. As expected, Western blotting displayed the similar results that the protein expression levels of Bax was upregulated in Cypher siRNA treated H9c2 cells (Fig. 4A and 4B). Furthermore, a significant decreased level of antiapoptotic protein p21 in Cypher siRNA treated H9c2 cells was detected (Fig. 4C and 4D). Increased apoptosis in Cypher knockdown H9c2 cells was further evident from increased ratio of cleaved caspase 3 to GAPDH. Thus, knockdown of Cypher had a promote ability on the mitochondria-dependent pathway activity to induce apoptosis in cardiomyocytes.
Effects of Cypher knockdown on the mRNA expression of apoptosis-related proteins in H9c2 cells. The mRNA levels of Cypher, bcl-xl, bcl-2, Bax, Bim, Bak, PUMA, Fas, p53, and NF-κB were detected by real-time polymerase chain reaction procedure. The relative levels of above mentioned mRNA were calculated by the values of ΔCt by normalizing with that of GAPDH. *P < 0.05 versus control. Experiments were repeated three times.
Effects of Cypher knockdown on apoptosis regulatory protein expression in H9c2 cells. (A-L) Representative western blot bands and statistical results of protein expression. *P < 0.05 versus control cells. Experiments were repeated three times.
MAPK and Akt participated in downregulation of Cypher induced apoptosis in H9c2 cardiomyocytes
Mitogen-activated protein kinases (MAPK) signal transduction pathways are involved in the regulation of apoptosis in cardiac myocytes [18]. Thus, we investigated whether these pathways were modified in Cypher knockdown H9c2 cells and western blotting was used to detect the phosphorylation levels of c-Jun NH2-terminal kinase (JNK), extracellular regulated protein kinases 1/2 (Erk1/2) and p38 MAPK, which are key mediators of cardiac apoptosis. As shown in Fig.4G-J, a significantly increased phosphorylated levels of p38 MAPK and decreased phosphorylation of Erk1/2 in Cypher siRNA treated H9c2 cells were detected by western blotting compared with those in the control, whereas there was no increase in the activity of JNK kinases (data not shown). And there was no significant change in the total expression levels of JNK, p38 MAPK and Erk after Cypher knockdown. We next examined whether the survival protein p-Akt was involved in apoptosis as determined by immunoblotting for phosphorylation of either Thr308 or Ser473. As shown in Fig.4K and 4L, Cypher siRNA treatment decreased phosphorylated Akt Ser473 expression, indicating that apoptotic process in Cypher siRNA treated H9c2 cells involved Akt activation. Collectively, these results suggested that knockdown of Cypher aggravated cardiomyocytes apoptosis possibly by regulation Akt/p38 MAPK signaling pathway.
SC79 reversed the apoptotic detrimental effects induced Cypher-deficiency
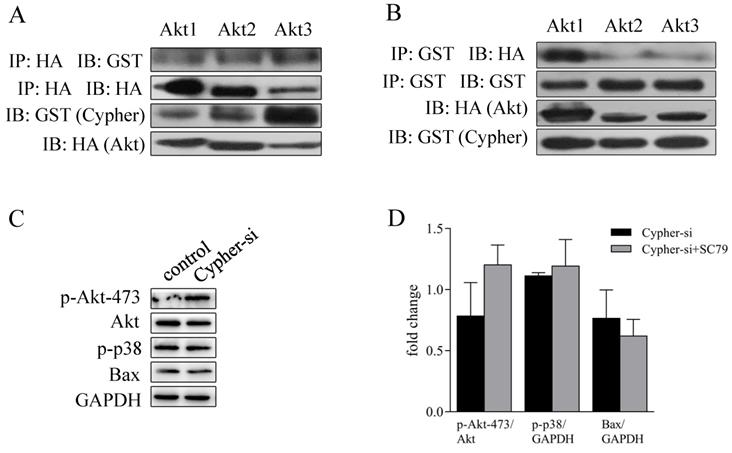
Interestingly, coimmunoprecipitation studies with Akt antibodies and Cypher demonstrated their colocalization (Fig. 5A and 5B). Then, we focused our attention on the rescue function of Akt activator on knockdown Cypher to reduce cells apoptosis. Finally, H9c2 cells in the presence of Cypher siRNA were then pre-incubated with 10 μM SC79 (a popular used agonist of Akt) for 30 min prior to harvest for western blotting analysis. The effect of the activator was confirmed by significantly increased Akt phosphorylation. Moreover, the increased expressions of apoptotic proteins Bax and p-p38 MAPK caused by Cypher suppression were markedly reversed by Akt activation concurrently. Taken together, these results confirmed that Akt/p38 MAPK constitute an axis regulating apoptosis in the Cypher-deficiency induced cardiomyocyte injury.
SC79 inhibited Cypher-siRNA-induced apoptosis of H9c2 cells by activating Akt. (A) HEK293T cells were transfected with GST-Cypher and HA-Akt plasmids. The cells were then lysed. Lysates from HEK293T co-immunoprecipitated (IP) with IgG or antibody to HA, followed by immunoblot (IB) analyses with GST antibody. (B) HEK293T cells were transfected with GST-Cypher and HA-Akt plasmids. The cells were then lysed. Lysates from HEK293T IP with IgG or antibody to GST, followed by IB analyses with HA antibody. H9c2 cells were pre-cultured in serum-free medium in the presence of Cypher-siRNA for 72 hrs, and then stimulated further with 10 µM SC79 for an additional 30 mins. The cells were then lysed, and Western blot analysis was performed. (C) Western band images are representative of three independent experiments. (D) The quantitative analysis of p-Akt-473 using total Akt as normalization, while p-p38 and Bax using GADPH as normalization. Experiments were repeated three times.
Discussion
DCM, the most primary myocardial disorder, is a major cause of chronic heart failure [19]. Various etiologies cause DCM, while the gene mutations paly significant effect on the pathogenesis of DCM [20]. Mutations in the sarcomeric and cytoskeletal genes, such as titin, desmin, lamin A/C, and α-actin, accounted for the majority of genetic causes [7]. And defects in cytoskeleton damaged cardiac structural integrity which seemed to be a prevalent mechanism of DCM.
Accumulating data indicated the involvement of Cypher/ZASP, one of the cytoskeletal proteins, in the pathogenesis of DCM [8-13]. Cypher knockout mice induced a severe form of DCM by disorganizing both sarcomere and cytoskeleton [10]. And ablation of c-Jun exhibited similar alterations, such as sarcomere disarrangement, cytoskeleton disorganization, resulting in DCM [21]. DCM-associated BAG3 mutations impaired Z-disc assembly and increased the stress-induced apoptosis in cultured cardiomyocytes [22,23]. And cardiac tissue from a DCM patient harboring a nonsynonymous mutation in α-actin gene also displayed sarcomeric disarray, and an increase in cardiomyocytes apoptosis [24].
Similarly, Cypher/ZASP not only maintains cardiac structure as scaffold of the cell, but also plays an important role in signal transduction [3]. Cypher/ZASP interacts with signaling molecules like PKA, p53, PKCs, and calcineurin to form multiprotein complexes at sarcomeric Z-lines [13-15]. However, the specific mechanisms remain unexplored. Since adult myocytes are undifferentiated cells, cardiomyocyte apoptosis has become a major mechanism underlying various cardiovascular diseases, including ischemia/reperfusion injury (I/R), doxorubicin (Dox) induced cardiotoxicity, ischemic heart disease, cardiomyopathy, and ultimately heart failure [25,26]. The proposed role for apoptosis in DCM has been supported by accumulating evidence in both human and animal models. Apoptosis rate in DCM patients was 0.08% to 0.25%, much higher than that of the control group (0.001% to 0.002%) [27]. And previous studies in both cTnT R141W and LMNA E82K transgenic mice, mouse models of DCM, showed increased apoptosis in the heart [28-30]. C-Jun knockout mice also underwent myocardial apoptosis [21]. Desmin-deficient mice displayed myocyte apoptosis finally resulting in DCM [31]. DCM mutations stress myocytes severely and trigger myocardial apoptosis reducing heart function. We therefore hypothesized that the apoptotic pathway may contribute to the development of DCM induced by Cypher deficient.
Apoptosis, or programmed cell death, is systematically regulated by numerous genes involving the pro- and anti-apoptotic families [32]. The mitochondrial pathway, the major apoptosis-inducing pathway, is initiated by releasing cytochrome c (Cyt c) into the cellular cytoplasm to form the apoptosome resulting in activating downstream effector caspase-3 [33]. The balance between pro-apoptotic (Bax) and anti-apoptotic (bcl-2) protein of the Bcl-2 family, both residing in the outer mitochondrial membrane, plays a crucial role on the process of apoptosis by regulating Cyt c release [34]. We then first studied whether the Cypher deficiency in cardiomyocytes exerted a direct apoptotic effect, by performing TUNEL staining, flow cytometric test and measuring caspase-3 activity. As illustrated in Figure 6, we observed that knockdown of Cypher significantly decreased cell viability, reduced p21 expression, increased cardiomyocytes apoptosis, the bcl-2/Bax ratio and caspase-3 expression. Furthermore, TUNEL staining in Cypher knockout mice myocardium showed the similar apoptotic effect in vivo.
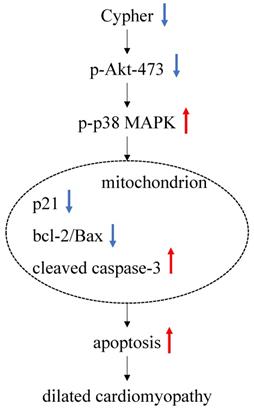
Schematic of the working signal pathway. In Cypher deficient cardiomyocytes, phosphor Akt-473 is downregulated and leads to a significant increased p-p38 MAPK. There it is assumed to activate mitochondria pathway, manifested specifically by decreasing the expression of p21 and the ratio of bcl-2 to Bax, while increasing the expression of cleaved caspase-3. These lead to an increase of apoptosis, thus ultimately leading to dilated cardiomyopathy.
Many stimuli regulate cell apoptosis through the Akt pathway [35]. Serine/threonine protein kinase B (PKB)/Akt is involved in the regulation of many process including cell survival, proliferation, metabolism, and angiogenesis [36]. Especially, Akt serves as a key mediator in cardiomyocyte apoptosis [37]. For example, activation of the Akt has been shown to protect cardiomyocytes from I/R or post-myocardial infarction injury through inhibiting myocyte apoptosis [38,39]. Previous studies revealed that Akt was inhibited in Dox-treated hearts, whereas enhanced Akt activation prevented cardiac apoptosis and dysfunction in response to Dox [40]. Studies in animal models of DCM revealed the Akt acted as an antiapoptotic effector in heart. Resveratrol improves cardiac function in the ischemic heart diseases and DCM through activating Akt-dependent pathway [41]. Dhcr24 protect against DCM also through up-regulation of activated Akt [42]. We then investigated whether Cypher-deficiency induced cardiac apoptosis was associated with changes in the Akt survival pathway. In the present study, we found a reduction in phosphorylation in Ser473 of Akt in Cypher knockdown H9c2 cells the same as a notably reduced level of p-Akt was observed in the cTnT R141W mice [29]. Previous study showed that phosphorylation Akt at Ser473 was necessary for its full activation [43].
To explore the underlying mechanism, the level of the cell stress signaling Erk, p38 MAPK and JNK were evaluated. Our data showed a significantly decreased level of p-Erk but increased level of p-p38 MAPK in Cypher knockdown H9c2 cells compared to the control group. Finally, to confirm whether the apoptotic effect in H9c2 cells inducing by Cypher deficiency was related to Akt pathway, the Akt activator SC79 was used. We found that SC79 administration protected against apoptosis in H9c2 cells treated with Cypher siRNA by enhancing Akt phosphorylation, inhibiting p38 MAPK phosphorylation and reducing pro-apoptotic protein Bax expression. Thus, there was clear evidence that Akt pathway plays an essential role in apoptotic effects inducing by Cypher deficiency.
Akt and MAPK are two main signaling pathways which participate in cardiomyocyte apoptosis [18,44]. The mammalian MAPK family, including Erk1/2, p38 MAPK, and JNK have been identified so far. Increased p38 MAPK activity promotes cardiac apoptosis, which could be suppressed by Akt activation [18]. Specifically, p38 MAPK signaling pathway is activated in H2O2-, I/R-, Dox-induced myocardial apoptosis [45-47]. Enhanced p38 MAPK activity results in cardiac remodeling and ventricular dilation, leading to severe cardiomyopathy at last. In the contrary to the pro-apoptotic effect of p38 MAPK, Erk1/2 signaling elicits protection in cells to maintain cardiac function by blocking caspase-3 activation [48].
In conclusion, the results of present study suggest that Cypher deficiency induces apoptosis by inhibiting Akt signaling and increasing p38 MAPK phosphorylation, which ultimately leads to DCM.
Acknowledgements
This work was supported by the Fundamental Research Funds for the Zhejiang Province Universities (2019XZZX003-15), Key Research and Development plan of Zhejiang Province (2020C03017), National key R & D plan of the Ministry of science and technology (2018YFE0119000).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Lopez-Ayala JM, Ortiz-Genga M, Gomez-Milanes I, Lopez-Cuenca D, Ruiz-Espejo F, Sanchez-Munoz JJ. et al. A mutation in the Z-line Cypher/ZASP protein is associated with arrhythmogenic right ventricular cardiomyopathy. Clin Genet. 2015;88(2):172-176
2. Peter AK, Cheng H, Ross RS, Knowlton KU, Chen J. The costamere bridges sarcomeres to the sarcolemma in striated muscle. Prog Pediatr Cardiol. 2011;31(2):83-88
3. Zheng M, Cheng H, Banerjee I, Chen J. ALP/Enigma PDZ-LIM domain proteins in the heart. J Mol Cell Biol. 2010;2(2):96-102
4. Levitas A, Konstantino Y, Muhammad E, Zaid Afawi, Jean Marc Weinstein, Guy Amit. et al. D117N in Cypher/ZASP may not be a causative mutation for dilated cardiomyopathy and ventricular arrhythmias. Eur J Hum Genet. 2016;24(5):666-671
5. Arimura T, Hayashi T, Terada H, Lee SY, Zhou Q, Takahashi M. et al. A Cypher/ZASP mutation associated with dilated cardiomyopathy alters the binding affinity to protein kinase C. J Biol Chem. 2004;279(8):6746-6752
6. Halliday BP, Cleland JGF, Goldberger JJ, Prasad SK. Personalizing Risk Stratification for Sudden Death in Dilated Cardiomyopathy: The Past, Present, and Future. Circulation. 2017;136(2):215-231
7. Weintraub RG, Semsarian C, Macdonald P. Dilated cardiomyopathy. Lancet. 2017;390(10092):400-414
8. Vatta M, Mohapatra B, Jimenez S, Sanchez X, Faulkner G, Perles Z. et al. Mutations in Cypher/ZASP in patients with dilated cardiomyopathy and left ventricular non-compaction. J Am Coll Cardiol. 2003;42(11):2014-2027
9. van der Meer DL, Marques IJ, Leito JT, Jaya Besser, Bakkers J, Schoonheere E. et al. Zebrafish cypher is important for somite formation and heart development. Dev Biol. 2006;299(2):356-372
10. Zheng M, Cheng H, Li X, Zhang JL, Cui L, Ouyang K. et al. Cardiac-specific ablation of Cypher leads to a severe form of dilated cardiomyopathy with premature death. Hum Mol Genet. 2009;18(4):701-713
11. Li Z, Ai T, Samani K, Xi YT, Tzeng HP, Xie MX. et al. A ZASP missense mutation, S196L, leads to cytoskeletal and electrical abnormalities in a mouse model of cardiomyopathy. Circ Arrhythm Electrophysiol. 2010;3(6):646-656
12. Frey N, Olson EN. Calsarcin-3, a novel skeletal muscle-specific member of the calsarcin family, interacts with multiple Z-disc proteins. J Biol Chem. 2002;277(16):13998-14004
13. Lin C, Guo X, Lange S, Liu J, Ouyang K, Yin X. et al. Cypher/ZASP is a novel A-kinase anchoring protein. J Biol Chem. 2013;288(41):29403-29413
14. Martinelli VC, Kyle WB, Kojic S, Vitulo N, Li ZH, Belgrano A. et al. ZASP interacts with the mechanosensing protein Ankrd2 and p53 in the signalling network of striated muscle. PLoS One. 2014;9(3):e92259
15. Lee MH, Siddoway B, Kaeser GE, Segota I, Rivera R, Romanow WJ. et al. Somatic APP gene recombination in Alzheimer's disease and normal neurons. Nature. 2018;563(7733):639-645
16. Sun Z, Han J, Zhao W, Zhang Y, Wang S, Ye L. et al. TRPV1 activation exacerbates hypoxia/reoxygenation-induced apoptosis in H9C2 cells via calcium overload and mitochondrial dysfunction. Int J Mol Sci. 2014;15(10):18362-18380
17. Zheng X, Zhang W, Hu X. Different concentrations of lipopolysaccharide regulate barrier function through the PI3K/Akt signalling pathway in human pulmonary microvascular endothelial cells. Sci Rep. 2018;8(1):9963
18. Al-Damry NT, Attia HA, Al-Rasheed NM, Al-Rasheed NM, Mohamad RA, Al-Amin MA. et al. Sitagliptin attenuates myocardial apoptosis via activating LKB-1/AMPK/Akt pathway and suppressing the activity of GSK-3β and p38α/MAPK in a rat model of diabetic cardiomyopathy. Biomed Pharmacother. 2018;107:347-358
19. Japp AG, Gulati A, Cook SA, Cowie MR, Prasad SK. The Diagnosis and Evaluation of Dilated Cardiomyopathy. J Am Coll Cardiol. 2016;67(25):2996-3010
20. McNally EM, Mestroni L. Dilated Cardiomyopathy: Genetic Determinants and Mechanisms. Circ Res. 2017;121(7):731-748
21. Windak R, Müller J, Felley A, Wagner EF, Pedrazzini T, Sumara G. et al. The AP-1 transcription factor c-Jun prevents stress-imposed maladaptive remodeling of the heart. PLoS One. 2013;8(9):e73294
22. Liu L, Sun K, Zhang X, Tang Y, Xu D. Advances in the role and mechanism of BAG3 in dilated cardiomyopathy. Heart Fail Rev. 2019 10.1007/s10741-019-09899-7
23. Arimura T, Ishikawa T, Nunoda S, Kawai S, Kimura A. Dilated cardiomyopathy-associated BAG3 mutations impair Z-disc assembly and enhance sensitivity to apoptosis in cardiomyocytes. Hum Mutat. 2011;32(12):1481-1491
24. Frank D, Yusuf Rangrez A, Friedrich C, Dittmann S, Stallmeyer B, Yadav P. et al. Cardiac α-Actin (ACTC1) Gene Mutation Causes Atrial-Septal Defects Associated With Late-Onset Dilated Cardiomyopathy. Circ Genom Precis Med. 2019;12(8):e002491
25. Lei Y, Li X, Yuan F, Liu L, Zhang J, Yang Y. et al. Toll-like receptor 4 ablation rescues against paraquat-triggered myocardial dysfunction: Role of ER stress and apoptosis. Environ Toxicol. 2017;32(2):656-668
26. Kültürsay H, Kayikçioğlu M. Apoptozis ve Kardiyovasküler Hastaliklar. Apoptosis and cardiovascular disease. Anadolu Kardiyol Derg. 2002;2(4):323-329
27. Ashrafian H, Watkins H. Reviews of translational medicine and genomics in cardiovascular disease: new disease taxonomy and therapeutic implications cardiomyopathies: therapeutics based on molecular phenotype. J Am Coll Cardiol. 2007;49(12):1251-1264
28. Zhao H, Wang W, Zhang J, Liang T, Fan GP, Wang ZW. et al. Inhibition of osteopontin reduce the cardiac myofibrosis in dilated cardiomyopathy via focal adhesion kinase mediated signaling pathway. Am J Transl Res. 2016;8(9):3645-3655
29. Lu D, Ma Y, Zhang W, Bao D, Dong W, Lian H. et al. Knockdown of cytochrome P450 2E1 inhibits oxidative stress and apoptosis in the cTnT(R141W) dilated cardiomyopathy transgenic mice. Hypertension. 2012;60(1):81-89
30. Lu D, Lian H, Zhang X, Shao H, Huang L, Qin C. et al. LMNA E82K mutation activates FAS and mitochondrial pathways of apoptosis in heart tissue specific transgenic mice. PLoS One. 2010;5(12):e15167
31. Chandar S, Yeo LS, Leimena C, Tan JC, Xiao XH, Nikolova-Krstevski V. et al. Effects of mechanical stress and carvedilol in lamin A/C-deficient dilated cardiomyopathy. Circ Res. 2010;106(3):573-582
32. Savitskaya MA, Onishchenko GE. Mechanisms of Apoptosis. Biochemistry (Mosc). 2015;80(11):1393-1405
33. Kalpage HA, Wan J, Morse PT, Zurek MP, Turner AA, Khobeir A. et al. Cytochrome c phosphorylation: Control of mitochondrial electron transport chain flux and apoptosis. Int J Biochem Cell Biol. 2020;121:105704
34. Edlich F. BCL-2 proteins and apoptosis: Recent insights and unknowns. Biochem Biophys Res Commun. 2018;500(1):26-34
35. Nitulescu GM, Van De Venter M, Nitulescu G, Ungurianu A, Juzenas P, Peng Q. et al. The Akt pathway in oncology therapy and beyond (Review). Int J Oncol. 2018;53(6):2319-2331
36. Jafari M, Ghadami E, Dadkhah T, Akhavan-Niaki H. PI3k/AKT signaling pathway: Erythropoiesis and beyond. J Cell Physiol. 2019;234(3):2373-2385
37. Yao H, Han X, Han X. The cardioprotection of the insulin-mediated PI3K/Akt/mTOR signaling pathway. Am J Cardiovasc Drugs. 2014;14(6):433-442
38. Miao W, Yan Y, Bao TH, Jia WJ, Yang F, Wang Y. et al. Ischemic postconditioning exerts neuroprotective effect through negatively regulating PI3K/Akt2 signaling pathway by microRNA-124. Biomed Pharmacother. 2020;126:109786
39. Li R, Liu R, Yan F, Zhuang X, Shi H, Gao X. Inhibition of TRPA1 Promotes Cardiac Repair in Mice after Myocardial Infarction. J Cardiovasc Pharmacol. 2019 10.1097/FJC.0000000000000783
40. Sahu R, Dua TK, Das S, De Feo V, Dewanjee S. Wheat phenolics suppress doxorubicin-induced cardiotoxicity via inhibition of oxidative stress, MAP kinase activation, NF-κB pathway, PI3K/Akt/mTOR impairment, and cardiac apoptosis. Food Chem Toxicol. 2019;125:503-519
41. Zhang CJ, Huang Y, Lu JD, Lin J, Ge ZR, Huang H. Upregulated microRNA-132 rescues cardiac fibrosis and restores cardiocyte proliferation in dilated cardiomyopathy through the phosphatase and tensin homolog-mediated PI3K/Akt signal transduction pathway. J Cell Biochem. 2018 10.1002/jcb.27081
42. Dong W, Guan FF, Zhang X, Gao S, Liu N, Chen W. et al. Dhcr24 activates the PI3K/Akt/HKII pathway and protects against dilated cardiomyopathy in mice. Animal Model Exp Med. 2018;1(1):40-52
43. Matsui T, Rosenzweig A. Convergent signal transduction pathways controlling cardiomyocyte survival and function: the role of PI 3-kinase and Akt. J Mol Cell Cardiol. 2005;38(1):63-71
44. Duan J, Guan Y, Mu F, Guo C, Zhang EH, Yin Y. et al. Protective effect of butin against ischemia/reperfusion-induced myocardial injury in diabetic mice: involvement of the AMPK/GSK-3β/Nrf2 signaling pathway. Sci Rep. 2017;7:41491
45. Park ES, Kang JC, Jang YC, Park JS, Jang SY, Kim DE. et al. Cardioprotective effects of rhamnetin in H9c2 cardiomyoblast cells under H₂O₂-induced apoptosis. J Ethnopharmacol. 2014;153(3):552-560
46. Hang P, Zhao J, Sun L, Li M, Han Y, Du Z. et al. Brain-derived neurotrophic factor attenuates doxorubicin-induced cardiac dysfunction through activating Akt signalling in rats. J Cell Mol Med. 2017;21(4):685-696
47. Liu J, Li Y, Tang Y, Cheng J, Wang J, Li J. et al. Rhein protects the myocardiac cells against hypoxia/reoxygention-induced injury by suppressing GSK3β activity. Phytomedicine. 2018;51:1-6
48. Lee MJ, Chen HM, Tzang BS, Lin CW, Wang CJ, Liu JY. et al. Ocimum gratissimum Aqueous Extract Protects H9c2 Myocardiac Cells from H(2)O(2)-Induced Cell Apoptosis through Akt Signalling. Evid Based Complement Alternat Med. 2011;2011:578060
Author contact
Corresponding author: Xiaogang Guo PhD, Department of Cardiology, The First Affiliated Hospital, Zhejiang University School of Medicine, 79 Qingchun Road, Hangzhou, Zhejiang 310003, China. E-mail: gxg22222edu.cn.

 Global reach, higher impact
Global reach, higher impact