Impact Factor ISSN: 1449-1907
Int J Med Sci 2020; 17(15):2257-2263. doi:10.7150/ijms.50007 This issue Cite
Research Paper
Prognosis models for severe and critical COVID-19 based on the Charlson and Elixhauser comorbidity indices
1. Department of Intensive Care Unit, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, China.
2. Department of Hematology, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, China.
3. Department of Endocrine and Metabolic Diseases, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou, Zhejiang, China.
Received 2020-6-27; Accepted 2020-8-10; Published 2020-8-25
Abstract
Background: Corona Virus Disease 2019 (COVID-19) has become a global pandemic. This study established prognostic scoring models based on comorbidities and other clinical information for severe and critical patients with COVID-19.
Material and Methods: We retrospectively collected data from 51 patients diagnosed as severe or critical COVID-19 who were admitted between January 29, 2020, and February 18, 2020. The Charlson (CCI), Elixhauser (ECI), and age- and smoking-adjusted Charlson (ASCCI) and Elixhauser (ASECI) comorbidity indices were used to evaluate the patient outcomes.
Results: The mean hospital length of stay (LOS) of the COVID-19 patients was 22.82 ± 12.32 days; 19 patients (37.3%) were hospitalized for more than 24 days. Multivariate analysis identified older age (OR 1.064, P = 0.018, 95%CI 1.011-1.121) and smoking (OR 3.696, P = 0.080, 95%CI 0.856-15.955) as positive predictors of a long LOS. There were significant trends for increasing hospital LOS with increasing CCI, ASCCI, and ASECI scores (OR 57.500, P = 0.001, 95%CI 5.687-581.399; OR 71.500, P = 0.001, 95%CI 5.689-898.642; and OR 19.556, P = 0.001, 95%CI 3.315-115.372, respectively). The result was similar for the outcome of critical illness (OR 21.333, P = 0.001, 95%CI 3.565-127.672; OR 13.000, P = 0.009, 95%CI 1.921-87.990; OR 11.333, P = 0.008, 95%CI 1.859-69.080, respectively).
Conclusions: This study established prognostic scoring models based on comorbidities and clinical information, which may help with the graded management of patients according to prognosis score and remind physicians to pay more attention to patients with high scores.
Keywords: Charlson comorbidity index, comorbidity, Corona Virus Disease, Elixhauser comorbidity index, length of stay, outcome
Introduction
In December 2019, several cases of unexplained pneumonia with a history of exposure to a South China seafood market were seen in hospitals in Wuhan, Hubei Province, China. The pathogen was quickly identified as a novel coronavirus [1] and the World Health Organization (WHO) officially named the disease Corona Virus Disease 2019 (COVID-19). The novel coronavirus, which is similar to severe acute respiratory syndrome coronavirus (SARS-CoV), was designated SARS-CoV-2 by the International Committee on Taxonomy of Viruses [2, 3]. On March 11, 2020, the WHO announced that COVID-19 had become a global pandemic. More than 200 countries/territories have reported laboratory-confirmed COVID-19 cases [4]. As of April 7, 2020, there have been 1,279,722 confirmed cases and 72,614 deaths globally [4].
Many clinical characteristics and outcomes of COVID-19 have been reported [5-7]. Although most patients have mild symptoms and favorable prognoses, older age is associated with poor prognosis for COVID-19 [7, 8]. Moreover, in a systematic review, Vardavas et al. showed that smoking was most likely to be associated with negative progression and adverse outcomes of COVID-19 [9]. In addition to older age and smoking, the COVID-19 patients with adverse clinical outcomes have a higher prevalence of comorbidities, such as chronic obstructive pulmonary disease, diabetes, hypertension, and malignancy [10]. It is important to evaluate the risk of adverse outcomes in COVID-19 patients by stratified analysis for comorbidities. However, it is difficult to integrate all of the comorbidity information simultaneously when evaluating clinical outcome. Therefore, several measures have been designed to evaluate the overall impact of comorbidities, including the Charlson (CCI) [11] and Elixhauser (ECI) comorbidity indices [12]. To our knowledge, no prognostic model based on comorbidities and clinical information has been reported for COVID-19 patients.
Most mild patients have good prognoses, although the outcomes of critically ill patients are unclear. Therefore, this study established prognostic scoring models based on comorbidities and clinical information, which may aid in evaluating the outcomes of and formulating medical strategies for severe and critical COVID-19 patients.
Material and Methods
Study participants
The patient records used in this study were obtained from the First Affiliated Hospital of Wenzhou Medical University (Wenzhou, Zhejiang, China) after ethics committee approval. The requirement for individual patient consent was waived for this study because it did not affect clinical care and all health information was deidentified.
Patients diagnosed with COVID-19 using the criteria of the Diagnosis and Treatment of Novel Coronavirus Pneumonia of China [13] (trial version 5) who received medical treatment for severe illness at the First Affiliated Hospital of Wenzhou Medical University were enrolled. Fifty-one patients were admitted between January 29, 2020, and February 18, 2020. In addition to the epidemiological history and clinical features, all patients included in our study were confirmed by positive SARS-CoV-2 nucleic acids using real-time reverse-transcription polymerase chain reaction detection of throat swabs or lower respiratory tract specimens. According to the clinical classification of the National Health Committee of China [13], the COVID-19 patients were divided into four types: mild, typical, severe, and critical. The mild and typical cases were excluded from this study.
Severe illness met at least one of the following criteria: (1) respiratory rate ≥ 30/minute, (2) finger oxygen saturation ≤ 93% at rest, and (3) arterial partial pressure of oxygen/inspiratory oxygen fraction ≤ 300 mmHg [13]. Critical illness met at least one of the following criteria: (1) respiratory failure with mechanical ventilation, (2) shock, and (3) transferred to the intensive care unit due to multiple organ failure [13].
Data collection
The data extracted from the electronic medical records of the First Affiliated Hospital of Wenzhou Medical University comprised gender, age, smoking history, comorbidities, initial symptoms, respiratory therapy strategies, medications, laboratory data, and hospital length of stay (LOS). The baseline laboratory data were measured during the first 24 hours of admission. Clinical outcomes were followed up to March 16, 2020. Patients were allowed to leave the hospital only when they met the discharge standards of the National Health Committee of China [13].
Comorbidity assessment
Information on individual comorbidities before the diagnosis of COVID-19 was obtained via oral reports by the patients and their families. Comorbidities were assessed using the following four indexes: CCI, ECI, age- and smoking-adjusted Charlson comorbidity index (ASCCI), and age- and smoking-adjusted Elixhauser comorbidity index (ASECI). The CCI [11] and ECI [12, 14] are comorbidity scoring systems for 17 and 30 different medical conditions, respectively. Tables S1 and S2 give the details of the comorbidities the CCI and ECI are based on. The ASCCI and ASECI were built from the CCI and ECI after adding points for age and smoking.
Outcome variables
The primary outcome was hospital LOS, divided into short (≤ 24 days) and long (> 24 days) LOS. The secondary outcome was progression to critical illness.
Statistical analysis
Kolmogorov-Smirnov normality tests were used to evaluate the normality assumption for numerical variables. Normally distributed data were expressed as the mean ± standard deviation and non-normally distributed data as the median and inter-quartile range. Categorical variables were presented as a frequency with a percentage. Inter-group differences for the normally and non-normally distributed variables were compared using the unpaired Student's t-test and Wilcoxon rank-sum test, respectively. The Pearson χ2 test and Fisher's exact test were used to analyze categorical variables.
To assess the association of clinical variables with hospital LOS in the COVID-19 patients, we first screened gender, age, smoking history, white blood cell (WBC), lymphocyte, d-dimer, and interleukin-6 (IL-6), which are reported to be related to prognosis. To avoid over-fitting, we removed the variables that were not associated with the outcome via univariate analysis (P-value ≥ 0.1). Then, the significant variables (P-value < 0.1, i.e., former/current smoking and age) in the multivariate model were assigned the corresponding scores matching the CCI and ECI according to the regression coefficient. This gave two new scoring models (ASCCI and ASECI) for predicting outcomes.
The ability of the four models (CCI, ECI, ASCCI, and ASECI) to predict prognosis was examined by logistic regression. The results are presented as odds ratios (ORs) with 95% confidence intervals (CIs). Model discrimination was also assessed by calculating the receiver operating characteristic (ROC) curves and area under the receiver operating characteristic curve (AUROC). The sensitivity and specificity of the models were determined by ROC curve analysis. The DeLong test was used to evaluate differences in AUROC among the models.
A two-sided P-value < 0.05 was regarded as representing statistical significance. Additionally, we used a wider P-value (< 0.1) to filter potentially relevant variables in the univariate analysis. Statistical analyses were performed using SPSS software 20.0 (SPSS, Chicago, IL, USA) and MedCalc software 19.0.5 (MedCalc Software, Ostend, Belgium).
Results
Clinical characteristics
The clinical characteristics of 51 patients with COVID-19 were summarized in Table 1. There were significant differences in scores of CCI, ECI, ASCCI and ASECI between short-term and long-term LOS group. One patient was classified in long-term LOS group even though she died 14 days after admission. The mean LOS of the COVID-19 patients was 22.82 ± 12.32 days, and 19 patients (37.3%) were > 24 days. The detailed distribution of hospital LOS was presented in Figure S1.
Clinical characteristics of the severe and critical patients with COVID-19
| Characteristics | Total | Short-term LOS (≤ 24 days) | Long-term LOS (> 24 days) |
|---|---|---|---|
| (n = 51) | (n = 32) | (n = 19) | |
| Gender (men/women) | 36/15 | 22/10 | 14/5 |
| Age (years) | 57.37 ± 14.98 | 53.09 ± 13.63 | 64.58 ± 14.69** |
| < 40, n (%) | 3 (5.9) | 3 (9.4) | 0 (0) |
| ≥ 40, < 50, n (%) | 12 (23.5) | 10 (31.3) | 2 (10.5) |
| ≥ 50, < 60, n (%) | 14 (27.5) | 7 (21.9) | 7 (36.8) |
| ≥ 60, < 70, n (%) | 10 (19.6) | 9 (28.1) | 1 (5.3) |
| ≥ 70, < 80, n (%) | 9 (17.6) | 3 (9.4) | 6 (31.6) |
| ≥ 80, < 90, n (%) | 2 (3.9) | 0 (0) | 2 (10.5) |
| ≥ 90, n (%) | 1 (2.0) | 0 (0) | 1 (5.3) |
| Smoking history | |||
| Former/current, n (%) | 40 (78.4) | 28 (87.5) | 12 (63.2) |
| Never, n (%) | 11 (21.6) | 4 (12.5) | 7 (36.8) |
| Initial symptoms | |||
| Fever, n (%) | 48 (94.1) | 30 (93.8) | 18 (94.7) |
| Chill, n (%) | 16 (31.4) | 14 (43.8) | 2 (10.5)* |
| Pharyngodynia, n (%) | 5 (9.8) | 2 (6.2) | 3 (15.8) |
| Cough, n (%) | 38 (74.5) | 22 (68.8) | 16 (84.2) |
| Sputum, n (%) | 22 (43.1) | 12 (37.5) | 10 (52.6) |
| Fatigue, n (%) | 7 (13.7) | 6 (18.8) | 1 (5.3) |
| Headache/dizziness, n (%) | 2 (3.9) | 2 (6.2) | 0 (0) |
| Myalgia, n (%) | 5 (9.8) | 4 (12.5) | 1 (5.3) |
| Conjunctival congestion, n (%) | 0 (0) | 0 (0) | 0 (0) |
| Diarrhea, n (%) | 2 (3.9) | 2 (6.2) | 0 (0) |
| Chest distress/dyspnea, n (%) | 10 (19.6) | 5 (15.6) | 5 (26.3) |
| Respiratory therapies | |||
| High-flow oxygen therapy, n (%) | 21 (41.2) | 5 (15.6) | 16 (84.2)** |
| Noninvasive ventilation, n (%) | 15 (29.4) | 2 (6.2) | 13(68.4)** |
| Invasive ventilation, n (%) | 9 (17.6) | 0 (0) | 9 (47.4)** |
| ECMO, n (%) | 6 (11.8) | 0 (0) | 6 (31.6)** |
| Medication | |||
| Antiviral therapy, n (%) | 51 (100) | 32 (100) | 19 (100) |
| Antibiotic therapy, n (%) | 46 (90.2) | 27 (84.4) | 19 (100) |
| Glucocorticoid therapy, n (%) | 27 (52.9) | 13 (40.6) | 14 (73.7)* |
| Intravenous immunoglobulin therapy, n (%) | 21 (41.2) | 12 (37.5) | 9 (47.4) |
| Thymosin therapy, n (%) | 24 (47.1) | 11 (34.4) | 13 (68.4)* |
| Laboratory data | |||
| WBC (109/L) | 7.78 (5.31-11.32) | 6.78 (4.84-10.88) | 9.95 (7.44-12.36)* |
| Lymphocyte (109/L) | 0.81 (0.54-1.11) | 0.81 (0.54-1.01) | 0.81 (0.48-1.21) |
| D-dimer (mg/L) | 0.85 (0.60-1.24) | 0.84 (0.55-1.17) | 0.91 (0.68-1.58) |
| IL-6 (pg/mL) | 12.71 (4.04-74.65) | 8.35 (3.88-47.84) | 34.00 (4.42-103.34) |
| CCI | 0.00 (0.00-1.00) | 0.00 (0.00-1.00) | 2.00 (1.00-3.00)** |
| ECI | 0.00 (0.00-9.00) | 0.00 (0.00-0.00) | 9.00 (0.00-13.00)** |
| ASCCI | 2.29 ± 1.78 | 1.42 ± 0.88 | 3.76 ± 1.95** |
| ASECI | 2.00 (0.50-11.00) | 1.25 (0.50-2.00) | 12.00 (2.50-15.00)** |
| Critical cases, n (%) | 13 (25.5) | 1 (3.1) | 12 (63.2)** |
Data were expressed as mean ± standard deviation, median (inter-quartile range) or frequency (percentage). *, P < 0.05; **, P < 0.01. ASCCI, age and smoking-adjusted Charlson comorbidity index; ASECI, age and smoking-adjusted Elixhauser comorbidity index; CCI, Charlson comorbidity index; COVID-19, Corona Virus Disease 2019; ECI, Elixhauser comorbidity index; ECMO, extracorporeal membrane oxygenation; IL-6, interleukin-6; LOS, length of stay; WBC, white blood cell.
Analysis of clinical variables
We analysed the relationships between 7 clinical variables (gender, age, smoking history, WBC, lymphocyte, d-dimer and IL-6) and hospital LOS. As shown in Table S3, statistical differences between age (OR = 1.065, P = 0.012, 95%CI = 1.014-1.119), smoking history (OR = 4.083, P = 0.049, 95%CI = 1.005-16.597), IL-6 (OR = 1.011, P = 0.080, 95%CI = 0.999-1.022) and hospital LOS was identified via univariate analysis. Then, IL-6 was removed from the final multivariable model (forward LR method) because P-value was ≥ 0.1. The final independent variables of the multivariable model were presented in Table 2. Lastly, the corresponding scores were assigned to former/current smoking group and different age groups according to regression coefficient.
The final parameters of the multivariable model
| Independent variables | Points | Regression coefficient | OR | P-value | 95%CI |
|---|---|---|---|---|---|
| Age (years) | 0.62 | 1.064 | 0.018 | 1.011-1.121 | |
| < 40 | 0 | ||||
| ≥ 40, < 50 | 0.5 | ||||
| ≥ 50, < 60 | 1.0 | ||||
| ≥ 60, < 70 | 1.5 | ||||
| ≥ 70, < 80 | 2 | ||||
| ≥ 80, < 90 | 2.5 | ||||
| ≥ 90 | 3 | ||||
| Smoking history | 1.307 | 3.696 | 0.080 | 0.856-15.955 | |
| Never | 0 | ||||
| Former/current | 1 | ||||
| Constant | - | -4.539 | - | 0.006 | - |
The significant P-value was indicated in bold. CI, confidence interval; OR, odds ratio.
Four models of prognostic evaluation based on comorbidities
The detailed distribution of comorbidities based on the CCI and ECI was presented in Tables S1 and S2. In the CCI model, a total of 27 patients (52.9%) had no comorbidity, and the most common comorbid condition was mild liver disease (11 patients, 21.6%). As to the ECI model, a total of 16 patients (31.4%) had no comorbidity, and the most common comorbid condition was hypertension (19 patients, 37.3%).
As shown in Table 3, there were significant trends for increasing hospital LOS with increasing scores of CCI, ASCCI and ASECI (OR = 57.500, P = 0.001, 95%CI = 5.687-581.399, OR = 71.500, P = 0.001, 95%CI = 5.689-898.642, OR = 19.556, P = 0.001, 95%CI = 3.315-115.372, respectively). A similar result can be drawn from the outcome of critical illness (OR = 21.333, P = 0.001, 95%CI = 3.565-127.672, OR = 13.000, P = 0.009, 95%CI = 1.921-87.990, OR = 11.333, P = 0.008, 95%CI = 1.859-69.080, respectively).
Four models of prognostic evaluation based on comorbidities for the severe and critical patients with COVID-19
| Models | LOS (≤ 24 days or > 24 days) | Critical illness (yes or no) | ||||
|---|---|---|---|---|---|---|
| OR | P-value | 95%CI | OR | P-value | 95%CI | |
| CCI model | ||||||
| = 0 (n = 27) | Reference | Reference | ||||
| = 1 (n = 13) | 3.594 | 0.104 | 0.769-16.787 | 1.455 | 0.703 | 0.212-9.984 |
| ≥ 2 (n = 11) | 57.500 | 0.001 | 5.687-581.399 | 21.333 | 0.001 | 3.565-127.672 |
| ECI model | ||||||
| < 0 (n = 5) | Reference | Reference | ||||
| = 0 (n = 28) | 0.250 | 0.191 | 0.031-1.999 | 0.480 | 0.565 | 0.040-5.831 |
| > 0 (n = 18) | 3.900 | 0.196 | 0.494-30.758 | 4.000 | 0.253 | 0.371-43.139 |
| ASCCI model | ||||||
| ≤ 1 (n = 15) | Reference | Reference | ||||
| > 1, ≤ 3 (n = 24) | 2.167 | 0.387 | 0.376-12.495 | 0.929 | 0.940 | 0.136-6.323 |
| > 3 (n = 12) | 71.500 | 0.001 | 5.689-898.642 | 13.000 | 0.009 | 1.921-87.990 |
| ASECI model | ||||||
| ≤ 1 (n = 19) | Reference | Reference | ||||
| > 1, ≤ 5 (n = 18) | 2.051 | 0.381 | 0.411-10.238 | 1.700 | 0.588 | 0.249-11.586 |
| > 5 (n = 14) | 19.556 | 0.001 | 3.315-115.372 | 11.333 | 0.008 | 1.859-69.080 |
The significant P-value was indicated in bold. ASCCI, age and smoking-adjusted Charlson comorbidity index; ASECI, age and smoking-adjusted Elixhauser comorbidity index; CCI, Charlson comorbidity index; CI, confidence interval; COVID-19, Corona Virus Disease 2019; ECI, Elixhauser comorbidity index; LOS, length of stay; OR, odds ratio.
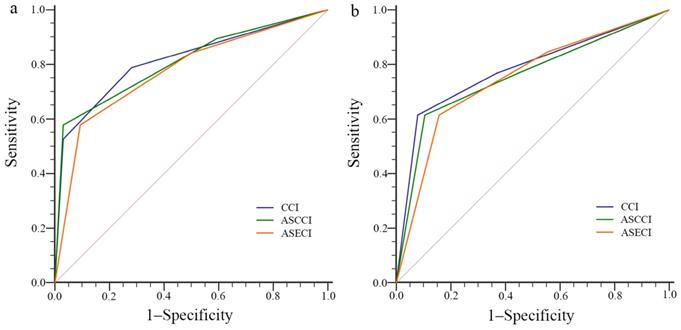
Comparisons of the performance among the different models, based on the sensitivity, specificity, ROC curves and AUROC, were summarized in Table 4 and Figure 1a, b. All the three models showed good performance, however, there were numerical differences but no statistical differences in the AUROC values among the three models via DeLong test.
Performance comparisons among the different models
| Models | Sensitivity | Specificity | AUROC | SE | 95%CI | P-value |
|---|---|---|---|---|---|---|
| For LOS | ||||||
| CCI model | 79.0 | 71.9 | 0.816 | 0.063 | 0.682-0.910 | < 0.0001 |
| ASCCI model | 57.9 | 96.9 | 0.808 | 0.063 | 0.674-0.905 | < 0.0001 |
| ASECI model | 57.9 | 90.6 | 0.776 | 0.068 | 0.638-0.881 | < 0.0001 |
| For critical illness | ||||||
| CCI model | 61.5 | 92.1 | 0.783 | 0.081 | 0.646-0.886 | 0.0005 |
| ASCCI model | 61.5 | 89.5 | 0.758 | 0.085 | 0.618-0.867 | 0.0023 |
| ASECI model | 61.5 | 84.2 | 0.750 | 0.080 | 0.609-0.861 | 0.0019 |
The values of sensitivity and specificity were expressed as percentage (%). ASCCI, age and smoking-adjusted Charlson comorbidity index; ASECI, age and smoking-adjusted Elixhauser comorbidity index; AUROC, area under receiver operating characteristic curve; CCI, Charlson comorbidity index; CI, confidence interval; LOS, length of stay; SE, standard error.
ROC curve of the different models for the outcomes of (a) hospital LOS and (b) critical illness. ASCCI, age and smoking-adjusted Charlson comorbidity index; ASECI, age and smoking-adjusted Elixhauser comorbidity index; CCI, Charlson comorbidity index; LOS, length of stay; ROC, receiver operating characteristic.
Discussion
This study found significant associations of the hospital LOS with clinical characteristics, including age, smoking history, IL-6, and comorbidities. Furthermore, we developed prognostic scoring models based on existing comorbidity indices to evaluate the outcomes of severe and critical COVID-19. The CCI, ASCCI, and ASECI models performed well and helped clinical-decision making.
In just a few months, the number of confirmed cases of COVID-19 and deaths worldwide has risen rapidly [4]. Although the overall mortality rate is lower than those of SARS-CoV and Middle East respiratory syndrome coronavirus [15], severe and critical COVID-19 patients still have poor outcomes and high mortality [15, 16]. It is important to identify effective indicators or scoring models that predict their outcomes.
Several studies have confirmed that older age and smoking status are associated with negative progression and poor outcomes of COVID-19 [7-9], which was consistent with our findings. Moreover, Li et al. found that males were likely to have more complicated clinical conditions and worse in-hospital outcomes than females [17]. In addition to demographic characteristics, several laboratory indicators have been reported to be closely related to the prognosis of COVID-19. Gao et al. reported that IL-6 and d-dimer were closely related to the occurrence of severe COVID-19 in adults, and their combined detection had the highest specificity and sensitivity for early prediction of the severity of COVID-19 [18]. Zhou et al. believed that d-dimer > 1 μg/mL could help clinicians to identify patients with poor prognosis at an early stage [8]. Qu et al. showed that the platelet-to-lymphocyte ratio of patients reflected the degree of cytokine storm, and might be a new predictor of the prognosis of COVID-19 [19]. However, no significant correlations of hospital LOS with gender, IL-6 and d-dimer were found in our study.
Examining the impact of comorbidities on the outcome of COVID-19, population analyses of the COVID-19 patients with cancer [20] and diabetes [21] found that the patients with either were more likely to have rapid progression and poor outcomes. Furthermore, Guan et al. evaluated the risk of a serious adverse outcome in patients with COVID-19 by stratification according to the number and type of comorbidities, identifying sub-populations with poorer prognoses [10]. However, no scoring system integrating all comorbidities has been established for evaluating clinical outcomes in COVID-19. Therefore, we evaluated a system for scoring comorbidities to evaluate their impact on the prognosis of severe and critical COVID-19, comparing the CCI, ECI, ASCCI, and ASECI comorbidity models to determine which one is the best outcome predictor.
The CCI includes 17 comorbidities and was first developed to predict 1-year mortality using data for one hospital and was validated in a cohort of 685 breast cancer patients from another hospital [11]. The CCI is the most widely used comorbidity index and has long proven useful [22-24]. Modification of the CCI, after adjusting for other significant covariates such as age could improve the predictive ability of the model [25]. The ECI includes 30 comorbid conditions and is used to predict in-hospital mortality [12]. Simard et al. established a new index combining the CCI and ECI that could predict the 30-day mortality in the general population [26]. However, the application of CCI and ECI to acute infectious diseases is still in its infancy. Therefore, this paper is an exploratory study.
Our study has several limitations. First, it was a single-center, retrospective study with a small sample size, so confounding factors and selection bias are inevitable. Second, the generalization ability of the models was not validated externally. Further studies need to validate these models using new data from different medical centers. Third, the original weights of CCI and ECI were derived using inpatient data from a hospital and they were not COVID-19-specific. Therefore, it is necessary to construct COVID-19-specific weights for a future comorbidity scoring model. Fourth, under-reporting of comorbidities should not be ignored as a major limitation, as it may lead to biased results. However, significant under-reporting was unlikely because our findings were largely consistent with previous studies [5, 6, 10].
Conclusions
Older age, smoking, and a high comorbidity score were most likely to be associated with poor prognoses for severe and critical COVID-19 cases. We established prognostic scoring models based on comorbidities and clinical information that might help the graded management of patients with different prognosis scores and remind physicians to pay more attention to patients with high risk scores.
Supplementary Material
Supplementary figure and tables.
Abbreviations
ASCCI, age and smoking-adjusted Charlson comorbidity index; ASECI, age and smoking-adjusted Elixhauser comorbidity index; AUROC, area under the receiver operating characteristic curve; CCI, Charlson comorbidity index; CI, confidence intervals; COVID-19, Corona Virus Disease 2019; ECI, Elixhauser comorbidity index; IL-6, interleukin-6; LOS, length of stay; OR, odds ratios; ROC, receiver operating characteristic; SARS-CoV, severe acute respiratory syndrome coronavirus; WBC, white blood cell; WHO, World Health Organization.
Acknowledgements
Ethics approval and informed consent
This study was approved by the Ethical Committee of the First Affiliated Hospital of Wenzhou Medical University. The requirement for individual patient consent was waived for this study because it did not impact clinical care and all health information was deidentified.
Data availability
The data analysed during the current study are available from the corresponding author on reasonable request.
Funding
This work was supported by a grant from Study on early warning and treatment of severe patients with novel coronavirus pneumonia in Wenzhou (NO. ZY202001-2), National Natural Science Foundation of China (NO. 81873949, 81671968), Major project co-founded by Zhejiang Province and Ministry of Science and Technology (NO. WKJ-ZJ-1909), and Major Science and Technology Project of Wenzhou Science and Technology Bureau (NO. 2018ZY002).
Author Contributions
JYP and WZ conceived and designed this study; XYQ and XH helped with the collection and assembly of data. All authors contributed toward data analysis, drafting and critically revising the paper and agree to be accountable for all aspects of the work. All authors read and approved the final manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W, China Novel Coronavirus Investigating, Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020;382(8):727-733
2. Ren LL, Wang YM, Wu ZQ, Xiang ZC, Guo L, Xu T, Jiang YZ, Xiong Y, Li YJ, Li XW, Li H, Fan GH, Gu XY, Xiao Y, Gao H, Xu JY, Yang F, Wang XM, Wu C, Chen L, Liu YW, Liu B, Yang J, Wang XR, Dong J, Li L, Huang CL, Zhao JP, Hu Y, Cheng ZS, Liu LL, Qian ZH, Qin C, Jin Q, Cao B, Wang JW. Identification of a novel coronavirus causing severe pneumonia in human: a descriptive study. Chin Med J (Engl). 2020 10.1097/CM9.0000000000000722
3. Gorbalenya AE, Baker SC, Baric RS, Baric RS, de Groot RJ, Drosten C, Gulyaeva AA, Haagmans BL, Lauber C, Leontovich AM, Neuman BW, Penzar D, Perlman S, Poon LL, Samborskiy D, Sidorov LA, Sola L, Ziebuhr J. Severe acute respiratory syndrome-related coronavirus: The species and its viruses-a statement of the Coronavirus Study Group. BioRxiv. 2020
4. World Health Organization. Novel Coronavirus (2019-nCoV) situation reports. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/.
5. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497-506
6. Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020 e201585
7. Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, Huang H, Zhang L, Zhou X, Du C, Zhang Y, Song J, Wang S, Chao Y, Yang Z, Xu J, Zhou X, Chen D, Xiong W, Xu L, Zhou F, Jiang J, Bai C, Zheng J, Song Y. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med. 2020 e200994
8. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054-1062
9. Vardavas CI, Nikitara K. COVID-19 and smoking: A systematic review of the evidence. Tob Induc Dis. 2020;18:20
10. Guan WJ, Liang WH, Zhao Y, Liang HR, Chen ZS, Li YM, Liu XQ, Chen RC, Tang CL, Wang T, Ou CQ, Li L, Chen PY, Sang L, Wang W, Li JF, Li CC, Ou LM, Cheng B, Xiong S, Ni ZY, Xiang J, Hu Y, Liu L, Shan H, Lei CL, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Cheng LL, Ye F, Li SY, Zheng JP, Zhang NF, Zhong NS, He JX; China Medical Treatment Expert Group for COVID-19. Comorbidity and its impact on 1590 patients with Covid-19 in China: A Nationwide Analysis. Eur Respir J. 2020 2000547
11. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373-83
12. Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care. 1998;36(1):8-27
13. National Health Commission of the People's Republic of China. Diagnosis and treatment protocols of pneumonia caused by a novel coronavirus (trial version 5). http://www.nhc.gov.cn/yzygj/s7653p/202002/3b09b894ac9b4204a79db5b8912d4440.shtml.
14. van Walraven C, Austin PC, Jennings A, Quan H, Forster AJ. A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care. 2009;47(6):626-633
15. Wang C, Horby PW, Hayden FG, Gao GF. A novel coronavirus outbreak of global health concern. Lancet. 2020;395(10223):470-473
16. Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. Zhonghua Liu Xing Bing Xue Za Zhi. 2020;41(2):145-151.
17. Li J, Zhang Y, Wang F, Liu B, Li H, Tang G, Chang Z, Liu A, Fu C, Gao J, Li J. Sex differences in clinical findings among patients with coronavirus disease 2019 (COVID-19) and severe condition. MedRxiv. 2020
18. Gao Y, Li T, Han M, Li X, Wu D, Xu Y, Zhu Y, Liu Y, Wang X, Wang L. Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19. J Med Virol. 2020 10.1002/jmv.25770
19. Qu R, Ling Y, Zhang YH, Wei LY, Chen X, Li XM, Liu XY, Liu HM, Guo Z, Ren H, Wang Q. Platelet-to-lymphocyte ratio is associated with prognosis in patients with coronavirus disease-19. J Med Virol. 2020 10.1002/jmv.25767
20. Liang W, Guan W, Chen R, Wang W, Li J, Xu K, Li C, Ai Q, Lu W, Liang H, Li S, He J. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21(3):335-337
21. Guo W, Li M, Dong Y, Zhou H, Zhang Z, Tian C, Qin R, Wang H, Shen Y, Du K, Zhao L, Fan H, Luo S, Hu D. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev. 2020 e3319
22. Jiménez Caballero PE, López Espuela F, Portilla Cuenca JC, Ramírez Moreno JM, Pedrera Zamorano JD, Casado Naranjo I. Charlson comorbidity index in ischemic stroke and intracerebral hemorrhage as predictor of mortality and functional outcome after 6 months. J Stroke Cerebrovasc Dis. 2013;22(7):e214-e218
23. Mayr R, May M, Burger M, Martini T, Pycha A, Dechet C, Lodde M, Comploj E, Wieland WF, Denzinger S, Otto W, Aziz A, Fritsche HM, Gierth M. The Charlson comorbidity index predicts survival after disease recurrence in patients following radical cystectomy for urothelial carcinoma of the bladder. Urol Int. 2014;93(3):303-310
24. Klabunde CN, Legler JM, Warren JL, Baldwin LM, Schrag D. A refined comorbidity measurement algorithm for claims-based studies of breast, prostate, colorectal, and lung cancer patients. Ann Epidemiol. 2007;17(8):584-590
25. Yang CC, Fong Y, Lin LC, Que J, Ting WC, Chang CL, Wu HM, Ho CH, Wang JJ, Huang CI. The age-adjusted Charlson comorbidity index is a better predictor of survival in operated lung cancer patients than the Charlson and Elixhauser comorbidity indices. Eur J Cardiothorac Surg. 2018;53(1):235-240
26. Simard M, Sirois C, Candas B. Validation of the Combined Comorbidity Index of Charlson and Elixhauser to Predict 30-Day Mortality Across ICD-9 and ICD-10. Med Care. 2018;56(5):441-447
Author contact
Corresponding authors: Jingye Pan, Department of Intensive Care Unit, The First Affiliated Hospital of Wenzhou Medical University, Nan Bai Xiang Street, Ouhai District, Wenzhou, Zhejiang 325000, China. Phone: 86-57788078262; Email: wyyyicucom. Yingru Lu, Department of Intensive Care Unit, The First Affiliated Hospital of Wenzhou Medical University, Nan Bai Xiang Street, Ouhai District, Wenzhou, Zhejiang 325000, China. Phone: 86-13857768811; Email: yingrulu66com.

 Global reach, higher impact
Global reach, higher impact