Impact Factor ISSN: 1449-1907
Int J Med Sci 2017; 14(3):224-230. doi:10.7150/ijms.17545 This issue Cite
Research Paper
Neuronal Effects of Sugammadex in combination with Rocuronium or Vecuronium
1. Department of Physiology, School of Medicine, University of Valencia, Valencia, Spain;
2. Hospital General de Castellon, Castellon, Spain.
Received 2016-9-12; Accepted 2016-12-28; Published 2017-2-23
Abstract
Rocuronium (ROC) and Vecuronium (VEC) are the most currently used steroidal non-depolarizing neuromuscular blocking (MNB) agents. Sugammadex (SUG) rapidly reverses steroidal NMB agents after anaesthesia. The present study was conducted in order to evaluate neuronal effects of SUG alone and in combination with both ROC and VEC. Using MTT, CASP-3 activity and Western-blot we determined the toxicity of SUG, ROC or VEC in neurons in primary culture. SUG induces apoptosis/necrosis in neurons in primary culture and increases cytochrome C (CytC), apoptosis-inducing factor (AIF), Smac/Diablo and Caspase 3 (CASP-3) protein expression. Our results also demonstrated that both ROC and VEC prevent these SUG effects. The protective role of both ROC and VEC could be explained by the fact that SUG encapsulates NMB drugs. In BBB impaired conditions it would be desirable to control SUG doses to prevent the excess of free SUG in plasma that may induce neuronal damage. A balance between SUG, ROC or VEC would be necessary to prevent the risk of cell damage.
Keywords: rocuronium, vecuronium, sugammadex, neuron, apoptosis.
Introduction
NMB drugs are used to obtain skeletal muscle relaxation in order to enable endotracheal intubation, facilitate mechanical ventilation and prevent patient from moving to assure optimal surgical conditions [1,2]. ROC and VEC are the most currently used steroidal non-depolarizing MNB agents [1,3,4,5] acting as competitive antagonists to acetylcholine (Ach) for binding to the nicotinic receptor at the neuromuscular junction [6,7,8]. After tracheal extubation, residual NMB may result in pulmonary complications, airway obstruction and increased morbidity. In this sense, pharmacological reversion of NMB is a useful tool to prevent post-anaesthetic complications [9,10]. Sugammadex (SUG) is a modified γ-cyclodextrin that rapidly reverses steroidal neuromuscular blockade after anaesthesia without inhibition of the enzyme acetylcholinesterase [11,12,2]. It is a drug with a doughnut-like molecular structure containing a lipophilic core and a hydrophilic periphery forming a complex with ROC or VEC that reduces their binding affinity to nicotinic receptors in the neuromuscular junctions [13]. SUG combined with ROC or VEC is well tolerated with no clinical evidence of residual neuromuscular blockade at recommended doses of SUG [14, 5].
In a previous study we demonstrated that sugammadex induces apoptosis/necrosis in neurons in primary culture and increases cytochrome C (CytC), apoptosis-inducing factor (AIF), Smac/Diablo and Caspase 3 (CASP-3) protein expression [15]. However, the combination of SUG with ROC or VEC has not been previously assessed. The present study was conducted in order to evaluate neuronal effects of SUG in combination with ROC or VEC
Material and Methods
Materials
All animals were handled according to the recommendations of the Bioethics Committee of the School of Medicine of the University of Valencia, Spain. Ethics committee specifically approved this study (number A1265026030697). SUG was acquired from Merck Millipore products (Madrid, Spain). Rocuronium Bromide and Vecuronium Bromide were obtained from Sigma Aldrich (Madrid, Spain). Appropriate concentrations of SUG (75 µg/ml), ROC (2 µg/ml) or VEC (0.33 µg/ml) were used for these experiments. Dulbecco's modified Eagle's medium (DMEM) and foetal bovine serum (FBS) were obtained from Gibco life technology (Gibco Invitrogen Corporation, Barcelona, Spain). 3-(4,5-dimethyl-2-thiazolyl)-2,5-dipheniyl-2H-tetrazolium bromide (MTT) was purchased from Sigma Chemical Co. (St Louis, MO). Western blot enhanced chemo-luminescent detection system (ECL) was obtained from Amersham Bioscience (Amersham Biosciences, Barcelona, Spain). Monoclonal anti-glial fibrillary acidic protein (anti-GFAP) antibody (1:500) was acquired from Sigma-Aldrich Biotech (Madrid, Spain). Monoclonal anti-MAP-2 protein (anti-MAP-2) antibody (1:500) and monoclonal anti-cytochrome C protein (anti-CytC) antibody (1:500) and anti-α-tubulin were acquired from Santa Cruz Biotechnology (Barcelona, Spain). Anti-apoptosis-inducing factor (anti-AIF) (1:500) and anti-Smac/Diablo (1:500) were obtained from Sigma-Aldrich Biotech. (Barcelona, Spain). All other reagents were of analytical or culture grade purity.
Neurons in primary culture
Primary cultures of cortical neurons were prepared from the cerebral cortex of 14-15-day old rat foetuses (5 female rats to obtain the foetuses). The cerebral cortex, obtained under toxin-free sterile conditions, was dissected and dissociated mechanically by pipetting 10 times with DMEM (10 ml for the cortex obtained from 12-14 foetuses). The cell suspension was filtered through a nylon mesh with a pore size of 90 μm and plated (5 x 104 cells/cm2) on poly-lysine-coated dishes. After the cell attachment (1 h), the plating medium was changed to DMEM pH 7.4, supplemented with 10% foetal bovine serum (FBS), 10 mM HEPES, 40 mM NaHCO3, 100 units/ml penicillin, and 100 mg/ml streptomycin. One hour is enough to stick neurons and to avoid astrocytes sticking on the plate.
After 4 days, the plating medium was changed with another medium prepared as described above. At seven days of culture, 20% FBS was changed to a 10% solution. Cultures were grown in a humidified atmosphere of 5% CO2 and 95% O2, at 37oC, for 3 days, and then exposed to cytosine β-D arabinofuranoside (10 μM) for 24 h to prevent proliferation of no neuronal cells. Possible contamination by astrocytes was assessed by immuno-fluorescence using monoclonal anti-glial fibrillary acidic protein (GFAP) (astrocytes marker: Sigma-Aldrich. 1:500) antibody (clone G-A-5) and polyclonal anti-microtubule-associated protein 2 (MAP-2) (neuronal marker: Sigma-Aldrich. 1:500). Under these conditions approximately 99% ± 3% of all cultured cells were neurons [15]. All animal work was performed according to minimize animal suffering and to reduce the total number of animals used.
MTT assay
Cellular viability of the cultures was determined by MTT assay [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] [16]. Cells were plated in a 96 well culture plate and incubated with SUG, ROC, VEC, SUG+ROC and SUG+VEC during 24 h. After cell treatments, the medium was removed and the cortical cells were incubated with red free medium and MTT solution [0.5 mg/ml, prepared in a phosphate buffer saline (PBS) solution] for 4 h at 37ºC. Finally the medium was removed and formazan particles were dissolved in dimethyl sulfoxide (DMSO). Cell viability was normalized relative to control cells whose viability was noted as 100% when treated for the indicated periods with the corresponding vehicles. Relative amount of MTT reduction, was determined by spectrophotometry at 570 nm.
Lactate Dehydrogenase (LDH) Assay
To evaluate plasma membrane integrity, LDH release was determined by monitoring the leakage of the cytosolic LDH to the extracellular medium. LDH was measured spectrophotometrically at 340 nm, following the rate of conversion of reduced nicotinamide adenine dinucleotide to oxidized nicotinamide adenine dinucleotide.
Optic microscopy
Neuron monolayers growing on culture dishes were used for optic microscopy studies. Photographs were made at the seventh day of culture. SUG, ROC and VEC were added 24 h before those images were obtained.
Caspase 3 activity assay
Sub-cellular fractioning of cortical neurons was carried out to segregate cytosol and membrane sections. Briefly, cells in the supernatant were collected by centrifugation and attached cells scraped in 5 mM EDTA in PBS were pelleted by a 10 min, 4°C centrifugation at 1000 x g. Pellets were re-suspended in 200 μl of fractionation buffer (0.25 M sucrose, 1 mM Mg chloride, 2 mM EGTA, and 25 mM HEPES, pH 7.4) and lysed by three cycles of flash freezing in liquid nitrogen. Lysates were then centrifuged at 100000 × g for 30 min at 4oC. Supernatants (cytosol fraction) were separated and the pellet (membrane fraction) was dissolved again in 200 μl of fractionation buffer containing 5% Triton X-100 and briefly sonicated. Whole-cell lysates were obtained by harvesting or homogenizing the samples in sub-cellular fractioning buffer containing 2.5% Triton X-100, followed by a brief sonication. This activity was measured in cytosolic fractions by using a highly sensitive colorimetric substrate, N-acetyl-Asp-Glu-Val-Asp p-nitroanilide (Ac-DEVD-pNA) following the manufacturer's instructions (CalBiochem, La Jolla, CA). Enzyme activity (pmol/min) was calculated, using the manufacturer's formulae.
Western-blots
Attached cells (neurons in primary culture) were collected and harvested with pH 7.4 ice-cold buffer (0.125 M Tris-HCl, pH 6.8, 2% SDS, 19% glycerol, 1 mM phenylmethylsulfonyl fluoride, 1μg/ml leupeptin, 1μg/ml aprotinin, and 1μg/ml pepstatin), and disrupted by repeated cycles of freezing and thawing, and then boiled for 5 min at 90º. Cell extracts were homogenized and centrifuged (10000 x g for 10 min at 4ºC). The resulting supernatant was used for western-blot analysis. Protein concentration was determined using a modified Lowry method [17]. Proteins were separated by SDS-PAGE gels and transferred to nitrocellulose membranes using standard techniques. Membranes were blocked with 5% dried milk in TBS containing 0.05% Tween-20 and then incubated with the corresponding antibodies following the manufacturer's recommendations. The blots were washed three times, 15 minutes each cycle, with phosphate-buffered saline containing 0.2% Tween 20 and then incubated for 1 h with a secondary horseradish peroxidase-linked anti-rabbit or anti-mouse IgG antibody. ECL was used for antibody detection. Auto-radiographic signals were assessed using a Bio-Rad scanning densitometer.
Statistical significance
Statistical analyses were performed using the t-Student test and one way ANOVA with the program GraphPad Prism 7. p ≤ 0.05 was considered statistically significant.
Results
SUG, ROC, VEC and cell viability
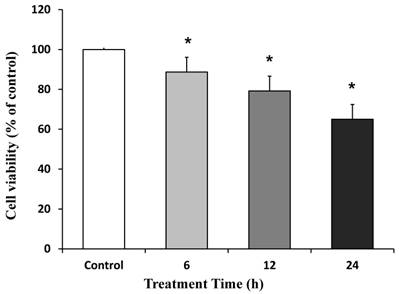
The role of SUG, ROC and VEC on cell viability was studied using MTT conversion assays. Figure 1 shows that incubation with SUG (75 µg/ml) at different treatment times (6, 12 and 24 h) induced a significant decrease in neuron viability at any time tested compared with control cells. We choose for future experiments 24 h treatment time, because it represents the biggest decrease data compared to control neurons.
Neuronal cell death induced by SUG (75 µg/ml). Time dependent neurotoxicity of SUG was determined by the MTT assay. Data are means ± SD for 4 independent experiments. *p ≤ 0.05 comparing SUG vs control (no additions).
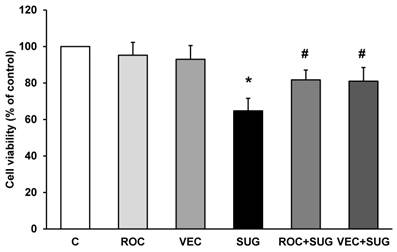
In Figure 2, we show that ROC or VEC did not change cell viability compared to control cells. On the other hand, both ROC and VEC prevented the decrease in cell viability induced by SUG (Figure 2).
Determination of cell viability using ROC (2 µg/ml), VEC (0.33 µg/ml), SUG (75 µg/ml), ROC+SUG (2 µg/ml and 75 µg/ml respectively), and VEC+SUG (0.33 µg/ml and 75 µg/ml respectively) by the MTT assay. *p ≤ 0.05 vs control. #p ≤ 0.05 vs SUG. Data are means ± SD for 5 independent experiments.
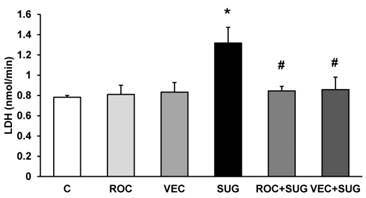
ROC and VEC did not increase LDH release whereas neurons incubated with SUG for 24 h showed a significant LDH leakage of 61.2% compared to control cells. The combination of SUG with ROC or VEC did not increase LDH release, indicating that ROC and VEC prevent cell death induced by SUG (Figure 3).
LDH assay using ROC (2 µg/ml), VEC (0.33 µg/ml), SUG (75 µg/ml), ROC+SUG (2 µg/ml and 75 µg/ml respectively), and VEC+SUG (0.33 µg/ml and 75 µg/ml respectively). *p ≤ 0.05 vs control. #p ≤ 0.05 vs SUG. Data are means ± SD for 4 independent experiments.
Optic microscopy after SUG, ROC and VEC treatment
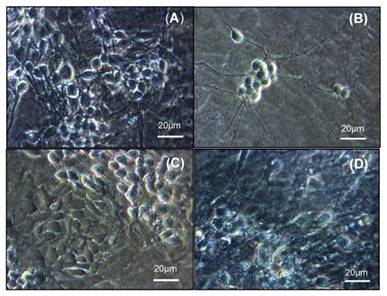
Using optic microscopy technique, we detected a decrease in cell number after 24 h of SUG treatment (Figure 4B) compared to control neurons (Figure 4A). The presence of ROC or VEC prevented the decrease in the number of neurons induced by SUG, Figure 4C and D.
Optic microscopic image of cultured neurons incubated without drugs (A), with SUG (75 µg/ml) (B), ROC (2 µg/ml) + SUG (75 µg/ml) (C) and VEC (0.33 µg/ml) + SUG (75 µg/ml) (D).
Caspase 3 activity
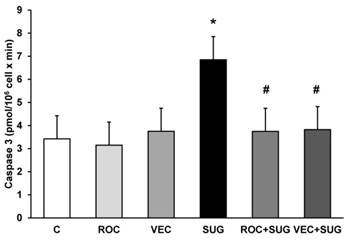
Neither ROC nor VEC altered the caspase 3 activity. The presence of SUG (75 µg/ml) for 24 h increased caspase 3 activity from 3.5 (control cells) to 6.89 pmol/106 cells x min, indicating significant increase of apoptosis after SUG addition. In the culture medium ROC or VEC avoided caspase-3 activity increase produced by SUG (Figure 5), which indicated a prevention of apoptosis when either ROC or VEC were present in the culture.
Determination of caspase 3 activity without (C) and with ROC (2µg/ml), VEC (0.33 µg/ml), SUG (75 µg/ml), ROC+SUG (2 µg/ml and 75 µg/ml respectively), and VEC+SUG (0.33 µg/ml and 75 µg/ml respectively). *p ≤ 0.05 vs control. #p ≤ 0.05 vs SUG. Data are means ± SD for 4 independent experiments.
Proteins related to cell-death molecular signals
Using western-blot technique shown in Figures 6, 7 and 8 different protein levels related to cell-death molecular signals were determined. ROC and VEC did not affect cytochrome c expression in neurons in primary culture. Nevertheless, SUG produced an increase of cytochrome c expression compared with control neurons (Figure 6). SUG incubated with either ROC or VEC avoided cytochrome c overexpression induced by SUG.
Quantitative analysis of cytochrome c. Results are expressed as relative changes in arbitrary units normalized against an internal standard (α-tubulin). Data are means ± SD (error bars) (n=5). *p ≤ 0.05 vs control; #p ≤ 0.05 vs SUG.
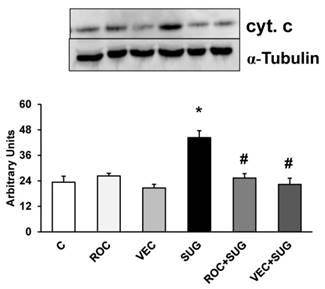
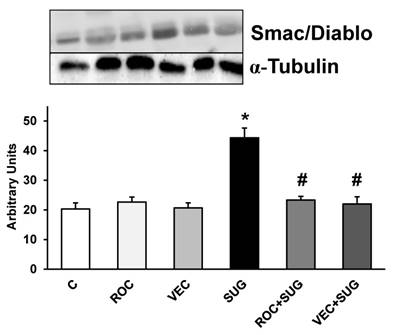
Figure 7 shows Smac/Diablo expression in neurons in primary culture. ROC and VEC did not modify Smac/Diablo expression whereas SUG increased this protein. The presence of either ROC or VEC prevented Smac/Diablo overexpression induced by SUG.
Results (Smac/Diablo) of quantitative analysis of immunoblots are means ± SD (error bars) (n=4) expressed as relative changes in arbitrary units normalized against an internal standard (α-tubulin). *p ≤ 0.05 vs control; #p ≤ 0.05 vs SUG.
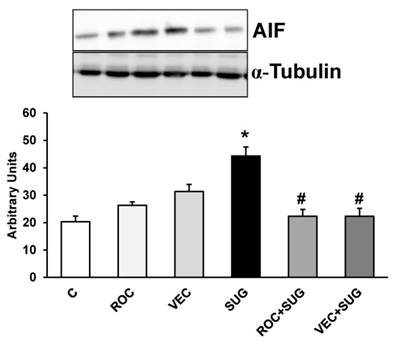
We demonstrated in Figure 8 the increase in AIF protein expression after SUG addition compared to control cells that was prevented by co-incubation with either ROC or VEC. Neither ROC nor VEC alone managed to modify AIF expression.
Results (AIF) of quantitative analysis of immunoblots are means ± SD (error bars) (n=5) expressed as relative changes in arbitrary units normalized against an internal standard (α-tubulin). *p ≤ 0.05 vs control; #p ≤ 0.05 vs SUG.
Discussion
We previously demonstrated that SUG at clinically significant doses, induces neuronal death by apoptosis/necrosis, increasing CytC, AIF, Smac/Diablo expression and CASP-3 activity [15]. In the present work, we show how ROC and VEC did not induce any direct effect in neurons in primary culture, but prevented toxic neuronal effects induced by SUG.
ROC and VEC act as antagonists of the cholinergic receptor at neuromuscular junction and this effect is reversed by SUG [11]. The side chains of SUG bind to steroidal NMB [18,19]. The union is non-covalent with Van-der-Waals interactions playing a minor role, and positively charged groups of NMB electrostatically bind negatively charged end groups of SUG [18]. The encapsulation by SUG, forming a 1:1 complex with free intravascular steroidal NMB drugs, rapidly reduces the concentration of both ROC and VEC in the neuromuscular junction, resulting in an earlier reversal of the neuromuscular blockade [5]. In a study in healthy volunteers, the simultaneous administration of SUG with ROC or VEC evidenced a faster decrease in plasmatic concentrations of NMB agents than those of SUG facilitating free SUG side effects [14]. This could imply that, in the clinical setting, if SUG is administered at high doses, the unbound SUG molecules will remain free, increasing the possibility of inducing toxic effects [5]. The affinity of SUG for ROC is 2.5 times higher than VEC [18]. In this sense, the rapid onset of action and rapid reversal of effects by SUG makes ROC preferable to VEC [19,20].
Both ROC and VEC have hepatic and renal metabolism [21] and the rate of elimination of NMB depends on their half-life, being shorter for VEC [22]. SUG is eliminated unaltered via urine [23,24] and the inactive complex ROC-SUG or VEC-SUG is rapidly cleared through the kidney, according to the pharmacokinetic properties of SUG [25,26,5]. Therefore, when renal or hepatic function is impaired, ROC, VEC and SUG half-life and plasma concentration can be increased [22].
Most frequently reported side effects of SUG administration are pain, nausea, coughing, headache or vomiting [2,10], being the hypersensitivity reactions, ranging from erythema to anaphylactic shock, are the most alarming among them [27,28,29]. Furthermore, allergy to NMB has been considered the first cause of perioperative anaphylaxis [30,31]. ROC could induce anaphylactic shock after induction of anaesthesia [32], and patients with a positive history of antibiotic hypersensitivity have a higher risk to develop intraoperative anaphylaxis [33]. ROC also has the potential to interact with other drugs, including aminoglycosides [34]. The incidence of VEC-induced anaphylactic reactions is lower than those induced by ROC [28]. Although SUG itself could induce hypersensitivity reactions, it was used to stop anaphylaxis induction by ROC, based on SUG design to encapsulate ROC [35]. In previous studies, SUG has proven capable of reversing the increase in mast-cell number and degranulation caused by ROC [36].
ROC, VEC or SUG cannot pass through a healthy and mature blood-brain barrier (BBB) [37,38]. However, there is evidence that patients with an impaired BBB function treated with either of these compounds, present autonomic dysfunction, seizures [39] and neuronal cell death [15]. Furthermore, ROC, but not VEC, has been detected in cerebrospinal fluid in patients with subarachnoid haemorrhage who underwent neurosurgery [39,40,41]. This finding could be attributed to the fact that ROC is more hydrosoluble than VEC, so it may easily arrives to central nervous system when BBB is damaged [42].
In summary, these reports highlight that clinical conditions which compromise the BBB integrity, such as Alzheimer´s disease, traumatic brain injury, brain ischemia or haemorrhage, meningitis, or immature nervous system, are susceptible of neurotoxicity induced by these drugs. [43,44,45].
Aging is associated with a decline in physiological function, and this includes drug metabolism [46]. In this sense, it has been reported that recovery periods from ROC and VEC were prolonged in elderly patients [47]. Moreover, when SUG was used for the reversion of the neuromuscular blockade induced by ROC, the recovery, although rapid, was slightly slower in the elderly patients [48,49]. Also other authors suggest that SUG, especially for ROC, fails to return to baseline values of EMG in elderly people [40]. Furthermore, there is no data about SUG effects in pregnant women, but in animal studies demonstrated the presence of SUG levels in breast milk [50]. SUG is not recommended for patients under 2 years old because of the lack of available data in this population but the risk of neuronal toxicity should be taken into account for future research [43].
In conclusion, the present study demonstrates that ROC or VEC prevents the increase of CytC, AIF, Smac/Diablo expression and CASP-3 activity induced by recommended doses of SUG in neurons in primary culture. Moreover, both ROC and VEC avoid cell death (apoptosis/necrosis) induced by SUG. This study suggests that it would be desirable to titrate SUG to the minimal effective dose in order to prevent excess of free SUG in plasma that could induce neuronal damage, especially in situations of BBB function impairment.
Acknowledgements
The authors thank to Generalitat Valenciana and University of Valencia for the financial support (AP 073/09).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Claudius C, Garvey LH, Viby-Mogensen J. The undesirable effects of neuromuscular blocking drugs. Anaesthesia. 2009Mar;64(Suppl 1):S10-21 doi: 10.1111/j.1365-2044.2008.05866.x
2. Kovac AL. Sugammadex: the first selective binding reversal agent for neuromuscular block. J Clin Anesth. 2009;21:444-453
3. Wang H, Liang QS, Cheng LR. et al. Magnesium sulfate enhances non-depolarizing muscle relaxant vecuronium action at adult muscle-type nicotinic acetylcholine receptor in vitro. Acta Pharmacol Sin. 2011;32:1454-1459
4. Vorce SP, Mallak CT, Jacobs A. Quantitative analysis of the aminosteroidal non-depolarizing neuromuscular blocking agent vecuronium by LC-ESI-MS: A Postmortem Investigation. J Anal Toxicol. 2008;32:422-427
5. Yang LP, Keam SJ. Sugammadex: a review of its use in anaesthetic practice. Drugs. 2009;69:919-942
6. Bowman WC. Neuromuscular block. British Journal of Pharmacology. 2006;147(Suppl 1):S277-286
7. Lien CA. Development and potential clinical impairment of ultra-short-acting neuromuscular blocking agents. Br J Anaesth. 2011;107(Suppl 1):S60-71
8. Martyn JA, Fagerlund MJ, Eriksson LI. Basic principles of neuromuscular transmission. Anaesthesia. 2009;64(Suppl 1):S1-9
9. Jones RK, Caldwell JE, Brull SJ. et al. Reversal of profound rocuronium-induced blockade with sugammadex: A randomized comparison with neostigmine. Anesthesiology. 2008;109:816-824
10. Lemmens HJ, El-Orbany MI, Berry J. et al. Reversal of profound vecuronium-induced neuromuscular block under sevoflurane anesthesia: sugammadex versus neostigmine. BMC Anesthesiol. 2010;1:10-15
11. Adam JM, Bennett DJ, Bom A. et al. Cyclodextrin-dereved host molecules as reversal agents for the neuromuscular blocker rocuronium bromide: synthesis and structure-activity relationships. J Med Chem. 2002;45:1806-1816
12. Naguib M. Sugammadex: another milestone in clinical neuromuscular pharmacology. Anesth. Analg. 2007;104:575-581
13. Suy K, Morias K, Cammu G. et al. Effective reversal of moderate rocuronium- or vecuronium-induced neuromuscular block with sugammadex, a selective relaxant binding agent. Anesthesiology. 2007;106:283-288
14. Cammu G, De Kam PJ, Demeyer I. et al. Safety and tolerability of single intravenous doses of sugammadex administered simultaneously with rocuronium or vecuronium in healthy volunteers. Br J Anaesth. 2008;100:373-379
15. Palanca JM, Aguirre-Rueda D, Granell MV. et al. Sugammadex, a neuromuscular blockade reversal agent, causes neuronal apoptosis in primary cultures. Int J Med Sci. 2013;10:1278-1285
16. Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55-63
17. Peterson GL. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem. 1977;83:346-356
18. Akha AS, Rosa J, Jahr JS. et al. Sugammadex: cyclodextrins, development of selective binding agents, pharmacology, clinical development, and future directions. Anesthesiol Clin. 2010;28:691-708
19. Bom A, Hope F, Rutherford S. et al. Preclinical pharmacology of sugammadex. J Crit Care. 2009;24:29-35
20. Haerter F, Eikermann M. Reversing neuromuscular blockade: inhibitors of the acetylcholinesterase versus the encapsulating agents SUG and calibration. Expert Opin Pharmacother. 2016;17:819-833
21. Proost JH, Eriksson LI, Mirakhur RK. et al. Urinary, biliary and faecal excretion of rocuronium in humans. Br J Anaesth. 2000;85:717-723
22. Cammu G, Van Vlem B, van den Heuvel M. et al. Dialysability of sugammadex and its complex with rocuronium in intensive care patients with severe renal impairment. Br J Anaesth. 2012;109:382-390
23. Nag K, Singh DR, Shetti AN. et al. Sugammadex: A revolutionary drug in neuromuscular pharmacology. Anesth Essays Res. 2013;7:302-306
24. de Souza CM, Tardelli MA, Tedesco H. et al. Efficacy and safety of sugammadex in the reversal of deep neuromuscular blockade induced by rocuronium in patients with end-stage renal disease: A comparative prospective clinical trial. Eur J Anaesthesiol. 2015;32:681-686
25. Peeters P, Passier P, Smeets J. et al. Sugammadex is cleared rapidly and primarily unchanged via renal excretion. Biopharm Drug Dispos. 2011;32:159-167
26. Ren W, Jahr JS. Reversal of neuromuscular block with a selective relaxant binding agent (SRBA). Am J Ther. 2009;16:295-299
27. Jeyadoss J, Kuruppu P, Nanjappa N. et al. Sugammadex hypersensitivity-a case of anaphylaxis. Anaesth Intensive Care. 2014;42:89-92
28. Sadleir PH, Russell T, Clarke RC. et al. Intraoperative anaphylaxis to sugammadex and a protocol for intradermal skin testing. Anaesth Intensive Care. 2014;42:93-96
29. Soria A, Motamed C, Gaouar H. et al. Severe reaction following sugammadex injection: hypersensitivity? J Investig Allergol Clin Immunol. 2012;22:382
30. Chiriac AM, Demoly P. Allergy to neuromuscular blocking agents. Presse Med. 2016;45:768-773
31. Takazawa T, Mitsuhata H, Mertes PM. Sugammadex and rocuronium-induced anaphylaxis. J Anesth. 2016;30:290-297
32. Schulberg EM, Webb AR, Kolawole H. Early skin and challenge testing after rocuronium anaphylaxis. Anaesth Intensive Care. 2016;44:425-427
33. Hagau N, Gherman N, Cocis M. et al. Antibiotic-induced immediate type hypersensitivity is a risk factor for positive allergy skin tests for neuromuscular blocking agents. Allergol Int. 2016;65:52-55
34. Hasfurther DL, Bailey PL. Failure of neuromuscular blockade reversal after rocuronium in a patient who received oral neomycin. Can J Anaesth. 1996;43:617-620
35. Barbosa FT, da Cunha RM. Case of anaphylaxis induced by rocuronium treated with sugammadex. Rev Bras Anestesiol. 2012;62:538-542
36. Tomak Y, Yilmaz A, Bostan H. et al. Effects of sugammadex and rocuronium mast cell number and degranulation in rat liver. Anaesthesia. 2012;67:1101-1104
37. Reynolds LM, Lau M, Brown R. et al. Bioavailability of intramuscular rocuronium in infants and children. Anesthesiology. 1997;87:1096-1105
38. Huang S, Kim JK, Atochin DN. et al. Cerebral blood volume affects blood-brain barrier integrity in an acute transient stroke model. J Cereb Blood Flow Metab. 2013;33:898-905
39. Cardone C, Szenohradszky J, Yost S. et al. Activation of brain acetylcholine receptors by neuromuscular blocking drugs. A possible mechanism of neurotoxicity. Anesthesiology. 1994;80:1155-1161
40. Fuchs-Buder T, Strowitzki M, Rentsch K. et al. Concentration of rocuronium in cerebrospinal fluid of patients undergoing cerebral aneurysm clipping. Br J Anaesth. 2004;92:419-421
41. Tassonyi E, Fathi M, Hughes GJ. et al. Cerebrospinal fluid concentrations of atracurium, laudanosine and VEC following clinical subarachnoid haemorrhage. Acta Anaesthesiol Scand. 2002;46:1236-1241
42. Roy JJ, Varin F. Physicochemical properties of neuromuscular blocking agents and their impact on the pharmacokinetic-pharmacodynamic relationship. Br J Anaesth. 2004;93:241-248
43. Langley RJ, McFadzean J, McCormack J. The presumed central nervous system effects of rocuronium in a neonate and its reversal with sugammadex. Paediatr Anaesth. 2016;26:109-111
44. Schmidt S, Saunders AM, De La Paz MA. et al. Association of the apolipoprotein E gene with age-related macular degeneration: possible effect modification by family history, age, and gender. Mol Vis. 2000;31:287-293
45. Rahe-Meyer N, Fennema H, Schulman S. et al. Effect of reversal of neuromuscular blockade with sugammadex versus usual care on bleeding risk in a randomized study of surgical patients. Anesthesiology. 2014;121:969-977
46. Priebe HJ. The aged cardiovascular risk patient. Br J Anaesth. 2000;85:763-778
47. McDonagh DL, Benedict PE, Kovac AL. et al. Efficacy, safety, and pharmacokinetics of sugammadex for the reversal of rocuronium-induced neuromuscular blockade in elderly patients. Anesthesiology. 2011;114:318-329
48. Leysen J, Bridts CH, De Clerck LS. et al. Rocuronium-induced anaphylaxis is probably not mitigated by sugammadex:evidence from an in vitro experiment. Anaesthesia. 2011;66:526-527
49. Shin S, Han DW, Lee HS. et al. Elderly patients require higher doses of Sugammadex for rapid recovery from deep neuromuscular block. Basic Clin Pharmacol Toxicol. 2016Jun;118:462-467
50. Merck & Co Inc. BRIDION: EPAR-product Information Annex I: Summary of product characteristics European Medicines Agency; London, UK. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_Product_Information/human/000885/WC500052310.pdf.
Author contact
Corresponding author: Soraya L. Valles. Department of Physiology, School of Medicine, University of Valencia. Blasco Ibañez 15, Valencia, Spain. Tel: +34 963 983 813; Fax: +34 963 864 646. Email: lilian.valleses.

 Global reach, higher impact
Global reach, higher impact