3.2
Impact Factor
ISSN: 1449-1907
Int J Med Sci 2015; 12(7):559-565. doi:10.7150/ijms.11521 This issue Cite
Research Paper
Etomidate Anesthesia during ERCP Caused More Stable Haemodynamic Responses Compared with Propofol: A Randomized Clinical Trial
1. Department of Anesthesiology, Eastern Hepatobiliary Surgery Hospital, Second Military Medical University, Shanghai, China
2. Department of Anesthesiology, Shanghai first people's hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
*Contributed equally to this work
Received 2015-1-7; Accepted 2015-5-25; Published 2015-7-3
Abstract
Background: Propofol may result in hypotension and respiratory depression, while etomidate is considered to be a safe induction agent for haemodynamically unstable patients because of its low risk of hypotension. We hypothesized that etomidate anesthesia during ERCP caused more stable haemodynamic responses compared with propofol. The primary endpoint was to compare the haemodynamic effects of etomidate vs. propofol in ERCP cases. The secondary endpoint was overall survival.
Methods: A total of 80 patients undergoing ERCP were randomly assigned to an etomidate or propofol group. Patients in the etomidate group received etomidate induction and maintenance during ERCP, and patients in the propofol group received propofol induction and maintenance. Cardiovascular parameters and procedure-related time were measured and recorded during ERCP.
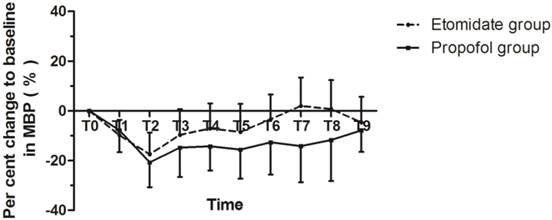
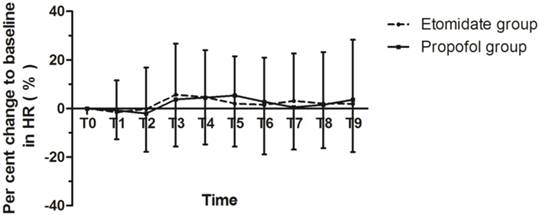
Results: The average percent change to baseline in MBP was -8.4±7.8 and -14.4±9.4 with P = 0.002, and in HR was 1.8±16.6 and 2.4±16.3 with P = 0.874 in the etomidate group and the propofol group, respectively. MBP values in the etomidate group decreased significantly less than those in the propofol group (P<0.05). The ERCP duration and recovery time in both groups was similar. There was no significant difference in the survival rates between groups ( p = 0.942).
Conclusions: Etomidate anesthesia during ERCP caused more stable haemodynamic responses compared with propofol.
Keywords: Etomidate anesthesia, propofol
Introduction
Over the last few years, there has been growing interest in the use of propofol in endoscopic procedures. However, propofol may result in hypotension, respiratory depression, and loss of protective reflexes. It is extremely important to ensure the patient's clinical stability during endoscopic procedures.[1] Most patients who need ERCP suffer from obstructive jaundice. Patients with obstructive jaundice are prone to develop hypotension and bradycardia during anesthesia induction and maintenance compared with nonjaundiced patients.[2-4] Etomidate is a nonbarbiturate hypnotic that induces anesthesia through GABA receptors in the central nervous system.[5] Etomidate is considered to be a safe induction agent for haemodynamically unstable patients because of its low risk of hypotension.[6, 7] Etomidate for procedural sedation has been used in emergency departments for many years.[8-10] Recently, it was reported that etomidate for sedation during colonoscopy resulted in more stable haemodynamic responses and shorter recovery and discharge times compared with propofol.[11] However, there are some conflicting results on the adverse effect of etomidate on adrenocortical suppression. One recent paper compared much larger numbers of patients given etomidate and propofol, and found that etomidate was associated with an increased risk of 30-day mortality, cardiovascular morbidity, and prolonged hospital stay [12], while another systematic review showed that etomidate suppressed adrenal function transiently without demonstrating a significant effect on mortality.[13]
In the present study, we hypothesized that etomidate anesthesia during ERCP causes more stable haemodynamic responses compared with propofol. The primary endpoint was to compare the haemodynamic effects of etomidate vs. propofol in ERCP cases. The secondary endpoint was overall survival. Overall survival was defined as the interval between treatment and death of any cause.
Methods
This study was approved by the Committee on Ethics of Biomedicine Research, Eastern Hepatobiliary Surgery Hospital (EHBHKY-2013-002-003) prior to its start. The registration number of randomized clinical trials is ChiCTR-TRC-13003850 (The URL is http://www.chictr.org/cn/. The name of the principal investigator is Jinchao Song). A total of 80 ASA I-III patients undergoing ERCP, aged 18-70 years and weighing 45-90 kg, were enrolled in this study. Written consent was obtained from all subjects. Patients with known adrenocortical insufficiency, chronic sedative or opioid analgesic use, known allergy to the study drugs, heart failure (ejection fraction <40%), and/or severe respiratory disease (vital capacity and/or forced expiratory volume <50%) preoperatively were excluded from this study. The patients were computer-randomized into either the etomidate group or the propofol group. The patients, gastroenterologists, anesthesiologist assistant and nurses in the recovery room were blinded to the grouping. The anesthesiologist assistant observed and recorded vital signs through a local area network in the next room.
All patients were premedicated with an intramuscular injection of 100mg pethidine twenty minutes before entering the endoscopy room. BIS (BIS™ XP sensor), noninvasive blood pressure, heart rate (HR), ECG and peripheral oxygen saturation (SpO2) were monitored continuously throughout the study in the endoscopy room (Philips HP Viridia 24/26 M1205A). A 20-gauge intravenous cannula was placed in the peripheral vein for 0.9% normal saline infusion and drugs. The BIS sensor (BISTM XP) was applied according to the manufacturer's recommendations. Oxygen was administered at a rate of 5 L/min by nasal catheter during ERCP. All the patients underwent ERCP in the prone position without tracheal intubation. After recovery, 50-100mg of pethidine was given, if necessary.
Baseline values of mean arterial blood pressure (MBP) were measured at 5 minutes after the patient entered the endoscopy room. Then, all patients received 2-2.5 mg midazolam (IV). After 5 minutes, induction was started. In the etomidate group, etomidate was delivered at a rate of 30 μg•kg-1•min-1 by a Graseby 3500 syringe pump (SIMS Graseby Ltd., Herts, England) until the BIS was 50, then ERCP was started. Anesthesia was maintained with etomidate (8-12 μg•kg-1•min-1) during ERCP. In the propofol group, propofol was delivered at a rate of 0.3 mg•kg-1•min-1 until the BIS was 50, and anesthesia was maintained with propofol (0.12-0.18 mg•kg-1•min-1). BIS was used to monitor the depth of anesthesia during the ERCP operation, and controlled at about 50 by fine-adjusting anesthetic agents. Emergency equipment was available throughout the ERCP procedure. Dopamine (2-5 μg • kg-1 • min-1) or an appropriate dose of metaraminol was given once MBP fell below 60 mmHg, and 0.25mg atropine was given once HR fell below 50 bpm. Appropriate nitroglycerin was given once MBP rose above 120 mmHg, and appropriate esmolol was given once HR rose above 120 bpm. If spontaneous ventilation was insufficient (SpO2 < 92%), the anesthesiologist performed assisted mask ventilation as necessary. If myoclonus was observed in the etomidate group, 50-100mg propofol was given and propofol took the place of etomidate in anesthesia maintenance. ERCP procedures were performed in a standardized manner under the supervision of two experienced gastroenterologists.
MBP, HR, SpO2 were measured and recorded at the designated time points: T0 = baseline values, 5 min after entering the endoscopy room; T1 = 5 min after the patients received midazolam; T2= when BIS was 50 (after induction of etomidate or propofol); T3 = at scope intubation and T4-10 = by 5-min intervals during the ERCP. The induction time, duration of ERCP and recovery time were recorded.
The patient's satisfaction with the procedure (1 = unacceptable, 2 = extremely uncomfortable, 3 = slightly uncomfortable, 4 = no discomfort) was assessed 1 hour after ERCP. The gastroenterologist's satisfaction was assessed immediately after ERCP as: 1, poor; 2, fair; 3, good; 4, excellent.[14]
Clinical signs of adrenocortical suppression such as hypotension and arrhythmia were monitored carefully after ERCP since etomidate may transiently cause adrenal suppression. After ruling out the general reason of hypotension and arrhythmia, we may consider adrenocortical suppression in etomidate group. Hydrocortisone (200-300 mg per day) was given if necessary.
The primary endpoint was the average percent change to baseline in mean arterial pressure and heart rate. Percent change = (MBPT1-10 - MBPT0)/ MBPT0* 100. The secondary endpoint was overall survival. Overall survival was defined as the interval between treatment and death of any cause.
The group sample size was calculated based on the result of a pilot study, in which we found that the average percent change to baseline in mean arterial pressure was -7.5±8.3 (n = 12) in the etomidate group and -13.5±10.3 (n = 12) in the propofol group. 38 samples for each group met the requirement of α = 0.05 and power = 0.8.[15]
All data in the text and tables are expressed as mean±SD, number (n) or percentage. Continuous outcomes with normal distribution were analyzed with independent 2-sample t-test. The count data were compared using the χ2 test or Fisher's exact test or Continuity correction where appropriate. Reported P value was 2-sided, with P < 0.05 considered statistically significant. The Kaplan-Meier estimate was used in survival analysis. All analyses were conducted using SPSS 17.0 (SPSS Inc., Chicago, IL). Figures were made using GraphPad Prism 5.
Results
The study was completed without any significant clinical complication. There was no statistical significance between characteristics of patients such as gender, age, body height, weight or diagnosis (Table 1).
The average percent change to baseline MBP was -8.4±7.8 and -14.4±9.4 with P = 0.002, and average percent change to baseline HR was 1.8±16.6 and 2.4±16.3 with P = 0.874 in the etomidate group and the propofol group, respectively. MBP values in the etomidate group decreased significantly less than those in the propofol group (P<0.05). Figure 1, 2 shows the time course of percent change to baseline in mean arterial pressure and heart rate. Figure 3 shows the SpO2% levels over the designated time points.
Induction time was longer in the etomidate group (P<0.05), but there was no statistical significance between groups for ERCP duration and recovery time (P>0.05) (Table 2). There was no statistical significance between groups for patient satisfaction and gastroenterologist satisfaction (P>0.05) (Table 2). The mean hospital stay was 8.7 days in the etomidate group vs. 8.6 days in the propofol group (n.s.).
All adverse events during and after ERCP are presented in Table 3. There was significantly more injection site pain in the propofol group (P<0.05).
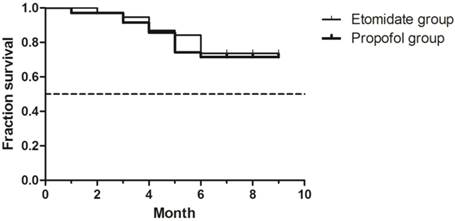
There was no significant difference in the survival rates between groups ( p = 0.942).
Patient Characteristics and Pre-operative Laboratory Values.
| Etomidate group (n=40) | Propofol group (n=40) | P | |
|---|---|---|---|
| Gender, M/F | 28/12 | 28/12 | - |
| Age, yr | 55.8±10.6 | 52.4±11.4 | 0.172 |
| Body height, cm | 164.8±8.6 | 164.1±8.3 | 0.762 |
| Weight, kg | 62.4±11.4 | 63.5±11.8 | 0.709 |
| Bilirubin, μmol/L | 118.2±117.8 | 108.2±142.2 | 0.748 |
| WBC, ×109/L | 5.2±1.7 | 5.9±2.0 | 0.126 |
| ASA (Ⅰ/Ⅱ/Ⅲ) | 18/15/7 | 17/16/7 | - |
| Benign disease, n | 22 | 23 | - |
| Malign disease, n | 18 | 17 | - |
Data are expressed as Mean±SD or number of patients. ASA, American Society of Anesthesiologists. Malign disease includes mainly Carcinoma of head of pancreas, Gallbladder Carcinoma, Hilar bile duct cholangiocarcinomas, Carcinoma in the middle and distal bile duct, and Intrahepatic bile duct cholangiocarcinomas. Benign disease includes mainly Intrahepatic bile duct stone, Choledocholithiasis, and Common bile duct stricture.
Procedure-related time, satisfaction. and hospital stay
| Etomidate group (n = 40) | Propofol group (n = 40) | P | |
|---|---|---|---|
| Induction time (min) | 5.6±0.8 | 5.2±0.9 | 0.037 |
| Duration of ERCP (min) | 20.9±8.4 | 20.4±9.2 | 0.800 |
| Recovery time (min) | 14.5±9.3 | 15.2±6.1 | 0.702 |
| Patient satisfaction | 3.8±0.4 | 3.8±0.3 | 0.419 |
| Gastroenterologist satisfaction | 3.8±0.4 | 3.8±0.4 | 1.000 |
| Hospital stay (days) | 8.7±3.6 | 8.6±3.3 | 0.856 |
Data are expressed as Mean±SD. Induction time (the time from starting propofol/etomidate to BIS = 50), Duration of ERCP (the time from scope intubation to scope withdrawal), and Recovery time (the time from stopping the drugs to full recovery (modified Aldrete score of 10)).
Adverse events
| Etomidate group n (%) | Propofol group n (%) | P | ||
|---|---|---|---|---|
| During ERCP | Desaturation | 0 | 0 | - |
| Apnoea | 0 | 0 | - | |
| Hypotension | 1 (2.5) | 1 (2.5) | 1.000 | |
| Bradycardia | 0 | 0 | - | |
| Hypertension | 2 (5) | 1 (2.5) | 1.000 | |
| Tachycardias | 4 (10) | 6 (15) | 0.499 | |
| Nausea-vomiting | 1 (2.5) | 1 (2.5) | 1.000 | |
| Myoclonus | 1 (2.5) | 0 | 1.000 | |
| Injection site pain | 0 | 6 (15) | 0.034 | |
| After ERCP | Pancreatitis | 0 | 0 | - |
| Cholangitis | 1 (2.5) | 2 (5) | 1.000 | |
| Sepsis | 0 | 0 | - | |
| Adrenal crisis | 0 | 0 | - |
Data are expressed as percentage. Desaturation (oxygen saturation <90% for >10 seconds); Apnoea (cessation of respiratory activity for over 10 seconds); Hypotension (MBP < 60 mmHg or decreases more than 25% from the baseline); Bradycardia (HR <50 bpm); Tachycardias was defined as HR >120 bpm in this study. Hypertension was defined as MBP > 120 mmHg.
The time course of percent change to baseline in mean arterial pressure. T0 = baseline values; T1 = at 5 min after the patients received midazolam; T2= when BIS was 50 after induction; T3 = at scope intubation; T4-10 = by 5-min intervals during the ERCP.
The time course of percent change to baseline in heart rate. T0 = baseline values; T1 = at 5 min after the patients received midazolam; T2= when BIS was 50 after induction; T3 = at scope intubation; T4-10 = by 5-min intervals during the ERCP.
The SpO2% levels over the designated time points. T0 = baseline values; T1 = at 5 min after the patients received midazolam; T2= when BIS was 50 after induction; T3 = at scope intubation; T4-10 = by 5-min intervals during the ERCP.
Survival analysis. Overall survival was defined as the interval between treatment and death of any cause.
Discussion
In the present study, we investigated the influence of etomidate and propofol on haemodynamics in patients who underwent ERCP. The results showed that etomidate anesthesia during ERCP caused more stable haemodynamic responses compared with propofol.
In our endoscopy center, as a rule, the patients underwent ERCP in the prone position without tracheal intubation. It is known that the prone position may lead to inhibition of breathing because of airway obstruction. To reduce the incidence of respiratory depression caused by opioid agents, patients received pethidine pretreatment (100mg i.m.) instead of intravenous opioids. The absorption of intramuscular injection of drugs may be irregular and a confounding factor to the hemodynamic stability. Patients in both groups received pethidine pretreatment, therefore, the analgesia level could be comparable between two groups. In the present study, no patient experienced desaturation or apnoea, and the incidence of respiratory depression was much lower than in the other reports.[11] There are at least two factors that may help explain this. First, the low incidence of respiratory depression primarily due to the normal BMI in the studied Chinese patients. Secondly, patients with known severe respiratory disease (vital capacity and/or forced expiratory volume <50%) were excluded from this study. There were not enough data in obese patients and in patients with severe respiratory disease. Therefore, we must be careful of hypoxia during ERCP under the general anesthesia without tracheal intubation in these patients. New techniques, such as supraglottic jet oxygenation and ventilation, can be used to minimize hypoxia/hypercapnia during ERCP under infusion of etomidate or propofol in these patients.[16]
It has been known that sedation and anesthesia are routinely required during ERCP [17-19], because ERCP is a complex endoscopic procedure requiring a high level of patient cooperation. Various types of sedative and analgesic techniques have been used during ERCP procedure. It is generally accepted among gastroenterologists that propofol is a good hypnotic with rapid onset, rapid recovery, and minimal side effects.[17,18,20] It was reported that a target-controlled infusion system for administration of propofol provided safe and effective sedation during ERCP.[21] In a pilot study, it was shown that patient-maintained sedation with TCI propofol was safe and fully effective in 16 patients.[22] However, it was reported in a guideline of sedation and anesthesia in GI endoscopy that transient hypotension occurs in 4% to 7% of cases using propofol sedation and transient hypoxia occurs in 3% to 7% of cases.[23]
Etomidate is a nonbarbiturate hypnotic that induces anesthesia through GABA receptors in the central nervous system.[5] It has a rapid onset of action (≤1 minute) and a short duration of action (3-5 minutes). According to Miller's Anesthesia, “The properties of etomidate include haemodynamic stability, minimal respiratory depression, cerebral protection, and pharmacokinetics enabling rapid recovery after a single dose”.[24] Etomidate's haemodynamic stability may be due to its unique lack of effect on the sympathetic nervous system and on baroreceptor function.[25]
Most patients who need ERCP suffer from obstructive jaundice. In the present study, the baseline of bilirubin was 118.2±117.8μmol/L in the etomidate group and 108.2±142.2 μmol/L in the propofol group. Patients with obstructive jaundice are more prone to develop hypotension and bradycardia during anesthesia induction and maintenance compared with non-jaundiced patients.[2,3] It was reported that patients with obstructive jaundice had decreased sensitivity in both the sympathetic and vagal components of the baroreflex.[4] Reich et al. suggested that “To avoid severe hypotension, alternatives to propofol anesthetic induction (e.g., etomidate) should be considered in patients older than 50 yr of age with ASA physical status ≥III.”[26]
Myoclonus was a common side effect of etomidate for procedural sedation, which occurred in 20% to 45% of the patients in the Falk review.[8] Miner et al. noted a 20% incidence of myoclonus in their randomized clinical trial comparing etomidate with propofol.[10] In the present study, only one patient in the etomidate group experienced myoclonus, and required a brief period of mask ventilation and 50mg propofol (i.v.). The incidence of myoclonus in the present study was far lower than in other studies, which may be due to midazolam (2-2.5 mg i.v.) pretreatment in all patients before induction. Midazolam pretreatment reduces etomidate-induced myoclonic movements.[27,28] Furthermore, in the present study, etomidate was delivered at a rate of 30 μg•kg-1•min-1 by a Graseby 3500 syringe pump. This relatively “slow” delivering speed may also reduce the incidence of myoclonus.[29]
It was reported mean aortic and left ventricular end-diastolic pressure decreased 5 and 15 min after midazolam, 0.2 mg/kg iv. [30] Marty et al. noted that midazolam (0.3 mg/kg) used for induction of anesthesia resulted in a transient depression of baroreflex function and a sustained decrease of sympathetic tone. [31] However, Lim et al. found that co-induction with midazolam and propofol could prevent a marked BP decrease at tracheal intubation for induction in aged patients. [32] In the present study, the blood pressure on T1 decreased compared with T0, which may be partly attributable to small dose of midazolam (2-2.5 mg i.v.). However, because patients in both groups received the same dose of midazolam, the same effects would be expected in both groups.
Other side effects of etomidate are nausea and vomiting.[33] Vinson's study reported that 4% (5 of 134 patients) of patients experienced nausea and vomiting,[9] but in the present study, only one patient in each group experienced these side effects.
Our study had two limitations. The first is that we did not measure plasma cortisol and adrenocorticotropic hormone levels. It has been well known that adrenocortical suppression is one of the most important adverse effects of etomidate.[34] A recent paper compared much larger numbers of patients given etomidate and propofol (2616 patients were given etomidate, and 28,532 were given propofol), they found that etomidate was associated with an increased risk of 30-day mortality, cardiovascular morbidity, and prolonged hospital stay. They thought that etomidate should be used judiciously, considering that improved haemodynamic stability may be accompanied by substantially worse longer-term outcomes.[12] However, in a systematic review and meta-analysis of randomized controlled trials and observational studies, it was reported that single-dose etomidate does not increase mortality in patients with sepsis.[35] In the present study, no patient experienced adrenal crisis. There was no significant difference between groups for pancreatitis, cholangitis or sepsis after ERCP. Etomidate did not prolong hospital stay. Furthermore, there was no significant difference in the survival distributions between groups. Etomidate did not worsen longer-term outcomes. Differences in patient characteristics might contribute partly to the different results between our study and Ryu Komatsu's study: ASA I-III patients were enrolled in this study, while ASA III-IV patients were evaluated in Komatsu's study. Our data indicate that etomidate can safely be used in ASA I-III patients during ERCP.
The second limitation is that we did not measure respiratory rate, as the prone position makes it difficult to accurately count respiratory rate both artificially and automatically. In the present study, the effect on respiratory function was judged only by SpO2, which may lead to missing subclinical respiratory depression.
In conclusion, our study demonstrated that etomidate anesthesia during ERCP caused more stable haemodynamic responses compared with propofol. Etomidate is an alternative to propofol during ERCP.
Acknowledgements
The authors thank Bing Hu, MD, PhD and Fenghai Yu, MD, PhD (Endoscopy center, Eastern Hepatobiliary Surgery Hospital) for some suggestions. The authors thank Olivia Hurwitz (Department of Anesthesiology, Yale University School of Medicine) for the article revise.
Funding
This study was supported by a grant from the National Natural Science Foundation of China: (NSFC, No.81371511 and No.31171013) and B. Braun Fund for Anesthesia Scientific Research (BBF2011-007).
Conflicts of Interest
The authors declare no conflicts of interest. The study complies with current ethical consideration.
References
1. Faigel DO, Baron TH, Goldstein JL, Hirota WK, Jacobson BC, Johanson JF, Leighton JA, Mallery JS, Peterson KA, Waring JP, Fanelli RD, Wheeler-Harbaugh J. Guidelines for the use of deep sedation and anesthesia for GI endoscopy. Gastrointest Endosc. 2002;56:613-7
2. Yang LQ, Song JC, Irwin MG, Song JG, Sun YM, Yu WF. A clinical prospective comparison of anesthetics sensitivity and hemodynamic effect among patients with or without obstructive jaundice. Acta anaesthesiologica Scandinavica. 2010;54:871-7
3. Song JG, Cao YF, Yang LQ, Yu WF, Li Q, Song JC, Fu XY, Fu Q. Awakening concentration of desflurane is decreased in patients with obstructive jaundice. Anesthesiology. 2005;102:562-5
4. Song JG, Cao YF, Sun YM, Ge YH, Xu XW, Yang LQ, Liu ZQ, Song SL, Yu WF. Baroreflex sensitivity is impaired in patients with obstructive jaundice. Anesthesiology. 2009;111:561-5
5. Davis PJ, Cook DR. Clinical pharmacokinetics of the newer intravenous anaesthetic agents. Clin Pharmacokinet. 1986;11:18-35
6. Möller Petrun A, Kamenik M. Bispectral index-guided induction of general anaesthesia in patients undergoing major abdominal surgery using propofol or etomidate: a double-blind, randomized, clinical trial. Br J Anaesth. 2013;110:388-96
7. Bendel S, Ruokonen E, Pölönen P, Uusaro A. Propofol causes more hypotension than etomidate in patients with severe aortic stenosis: a double-blind, randomized study comparing propofol and etomidate. Acta Anaesthesiol Scand. 2007;51:284-9
8. Falk J, Zed PJ. Etomidate for procedural sedation in the emergency department [review article]. Ann Pharmacother. 2004;38:1272-7
9. Vinson DR, Bradbury DR. Etomidate for procedural sedation in emergency medicine. Ann Emerg Med. 2002;39:592-8
10. Miner JR, Danahy M, Moch A, Biros M. Randomized clinical trial of etomidate versus propofol for procedural sedation in the emergency department. Ann Emerg Med. 2007;49:15-22
11. Toklu S, Iyilikci L, Gonen C, Ciftci L, Gunenc F, Sahin E, Gokel E. Comparison of etomidate-remifentanil and propofol-remifentanil sedation in patients scheduled for colonoscopy. Eur J Anaesthesiol. 2009;26:370-6
12. Komatsu R, You J, Mascha EJ, Sessler DI, Kasuya Y, Turan A. Anesthetic induction with etomidate, rather than propofol, is associated with increased 30-day mortality and cardiovascular morbidity after noncardiac surgery. Anesth Analg. 2013;117:1329-37
13. Hohl CM, Kelly-Smith CH, Yeung TC, Sweet DD, Doyle-Waters MM, Schulzer M. The effect of a bolus dose of etomidate on cortisol levels, mortality, and health services utilization: a systematic review. Ann Emerg Med. 2010;56:105-13
14. Manolaraki MM, Theodoropoulou A, Stroumpos C, Vardas E, Oustamanolakis P, Gritzali A, Chlouverakis G, Paspatis GA. Remifentanil compared with midazolam and pethidine sedation during colonoscopy: a prospective, randomized study. Dig Dis Sci. 2008;53:34-40
15. Lerman J. Study design in clinical research: sample size estimation and power analysis. Can J Anaesth. 1996;43:184-91
16. Levitt C, Wei H. Supraglotic pulsatile jet oxygenation and ventilation during deep propofol sedation for upper gastrointestinal endoscopy in a morbidly obese patient. J Clin Anesth. 2014;26:157-9
17. Angsuwatcharakon P, Rerknimitr R, Ridtitid W, Kongkam P, Poonyathawon S, Ponauthai Y, Sumdin S, Kullavanijaya P. Cocktail sedation containing propofol versus conventional sedation for ERCP: a prospective, randomized controlled study. BMC Anesthesiol. 2012;9:12-20
18. Jung M, Hofmann C, Kiesslich R, Brackertz A. Improved sedation in diagnostic and therapeutic ERCP: propofol is an alternative to midazolam. Endoscopy. 2000;32:233-8
19. Wehrmann T, Kokabpick S, Lembcke B, Caspary WF, Seifert H. Efficacy and safety of intravenous propofol sedation during routine ERCP: a prospective, controlled study. Gastrointest Endosc. 1999;49:677-83
20. Kongkam P, Rerknimitr R, Punyathavorn S, Sitthi-Amorn C, Ponauthai Y, Prempracha N, Kullavanijaya P. Propofol infusion versus intermittent meperidine and midazolam injection for conscious sedation in ERCP. J Gastrointestin Liver Dis. 2008;17:291-7
21. Fanti L, Agostoni M, Casati A, Guslandi M, Giollo P, Torri G, Testoni PA. Target-controlled propofol infusion during monitored anesthesia in patients undergoing ERCP. Gastrointest Endosc. 2004;60:361-6
22. Gillham MJ, Hutchinson RC, Carter R, Kenny GN. Patient-maintained sedation for ERCP with a target-controlled infusion of propofol: a pilot study. Gastrointest Endosc. 2001;54:14-7
23. Standards of Practice Committee of the American Society for Gastrointestinal Endoscopy, Lichtenstein DR, Jagannath S, Baron TH, Anderson MA, Banerjee S, Dominitz JA, Fanelli RD, Gan SI, Harrison ME, Ikenberry SO, Shen B, Stewart L, Khan K, Vargo JJ. Sedation and anesthesia in GI endoscopy [guideline]. Gastrointest Endosc. 2008;68:815-26
24. Reves JG. Intravenous Anesthetics. In Miller Rd, ed. Miller's Anesthesia. 7th ed. Philadelphia: Elsevier Churchill Livingstone. 2009 Chapter 26
25. Ebert TJ, Muzi M, Berens R, Goff D, Kampine JP. Sympathetic responses to induction of anesthesia in humans with propofol or etomidate. Anesthesiology. 1992;76:725-33
26. Reich DL, Hossain S, Krol M, Baez B, Patel P, Bernstein A, Bodian CA. Predictors of hypotension after induction of general anesthesia. Anesth Analg. 2005;101:622-8
27. Schwarzkopf KR, Hueter L, Simon M, Fritz HG. Midazolam pretreatment reduces etomidate-induced myoclonic movements. Anaesth Intensive Care. 2003;31:18-20
28. Hüter L, Schreiber T, Gugel M, Schwarzkopf K. Low-dose intravenous midazolam reduces etomidate-induced myoclonus: a prospective, randomized study in patients undergoing elective cardioversion. Anesth Analg. 2007;105:1298-302
29. Song JC, Sun YM, Zhang MZ, Yang LQ, Tao TZ, Yu WF. The etomidate requirement is decreased in patients with obstructive jaundice. Anesth Analg. 2011;113:1028-32
30. Marty J, Nitenberg A, Blanchet F, Zouioueche S, Desmonts JM. Effects of midazolam on the coronary circulation in patients with coronary artery disease. Anesthesiology. 1986;64:206-10
31. Marty J, Gauzit R, Lefevre P, Couderc E, Farinotti R, Henzel C, Desmonts JM. Effects of diazepam and midazolam on baroreflex control of heart rate and on sympathetic activity in humans. Anesth Analg. 1986;65:113-9
32. Lim YS, Kang DH, Kim SH, Jang TH, Kim KH, Ryu SJ, Yu SB, Kim DS. The cardiovascular effects of midazolam co-induction to propofol for induction in aged patients. Korean J Anesthesiol. 2012;62:536-42
33. Ruth WJ, Burton JH, Bock AJ. Intravenous etomidate for procedural sedation in emergency department patients. Acad Emerg Med. 2001;8:13-8
34. Wagner RL, White PF, Kan PB, Rosenthal MH, Feldman D. Inhibition of adrenal steroidogenesis by the anesthetic etomidate. N Engl J Med. 1984;310:1415-21
35. Gu WJ, Wang F, Tang L, Liu JC. Single-dose etomidate does not increase mortality in patients with sepsis: a systematic review and meta-analysis of randomized controlled trials and observational studies. Chest. 2015;147:335-46
Author contact
![]() Corresponding author: Wei-Feng Yu, Department of Anesthesiology, Eastern Hepatobiliary Surgery Hospital, Second Military Medical University, Changhai Rd., No. 225, Shanghai, China. E-mail: ywf808com. Tel and fax: +86 21 81875231
Corresponding author: Wei-Feng Yu, Department of Anesthesiology, Eastern Hepatobiliary Surgery Hospital, Second Military Medical University, Changhai Rd., No. 225, Shanghai, China. E-mail: ywf808com. Tel and fax: +86 21 81875231

 Global reach, higher impact
Global reach, higher impact