Impact Factor ISSN: 1449-1907
Int J Med Sci 2015; 12(5):407-415. doi:10.7150/ijms.11270 This issue Cite
Research Paper
Lentiviral Vectors Mediate Long-Term and High Efficiency Transgene Expression in HEK 293T cells
1. School of Biotechnology, Southern Medical University, Guangzhou, Guangdong, China
2. Department of Biomedical Engineering, The Johns Hopkins University School of Medicine, Baltimore, USA
3. Beijing Minhai Biotechnology CO., LTD, Beijing, China
4. Department of Pharmacology, University of Florida, Gainesville, Florida, USA.
5. Department of Physiology and Functional Genomics, University of Florida, Gainesville, Florida, USA.
#These authors contributed equally to this work.
Received 2014-12-8; Accepted 2015-2-20; Published 2015-5-15
Abstract
Objectives: Lentiviral vectors have been used successfully to rapidly produce decigram quantities of active recombinant proteins in mammalian cell lines. To optimize the protein production platform, the roles of Ubiquitous Chromatin Opening Element (UCOE), an insulator, and selected promoters were evaluated based on efficiency and stability of foreign gene expression mediated by lentiviral vectors.
Methods: Five lentiviral vectors, pFIN-EF1α-GFP-2A-mCherH-WPRE containing EF1α promoter and HS4 insulator, p'HR.cppt.3'1.2kb-UCOE-SFFV-eGFP containing SFFV promoter and UCOE, pTYF-CMV(β-globin intron)-eGFP containing CMV promoter and β-globin intron, pTYF-CMV-eGFP containing CMV promoter, and pTYF-EF1α-eGFP with EF1α promoter were packaged, titered, and then transduced into 293T cells (1000 viral genomes per cell). The transduced cells were passaged once every three days at a ratio of 1:10. Expression level and stability of the foreign gene, green fluorescence protein (GFP), was evaluated using fluorescent microscopy and flow cytometry. Furthermore, we constructed a hepatitis C virus (HCV) E1 recombinant lentiviral vector, pLV-CMV-E1, driven by the CMV promoter. This vector was packaged and transduced into 293T cells, and the recombinant cell lines with stable expression of E1 protein were established by limiting dilution.
Results: GFP expression in 293T cells transduced with the five lentiviral vectors peaked between passages 3 and 5 and persisted for more than 5 weeks. The expression was prolonged in the cells transduced with TYF-CMV (β-globin intron)-eGFP or TYF-CMV-eGFP, demonstrating less than a 50% decrease even at 9 weeks post transduction (p>0.05). The TYF-CMV-eGFP-transduced cells began with a higher level of GFP expression than other vectors did. The percentage of GFP positive cells for any of the five lentiviral vectors sustained over time. Moreover, the survival rates of all transfected cells exceeded 80% at both 5 and 9 weeks post transduction. Surprisingly, neither the HS4 insulator nor the UCOE sequence improved the GFP expression level or stability. Clonal cell lines with HCV E1 gene were generated from LV-CMV-E1 vector-infected 293T cells. A representative recombinant cell line maintained stable E1expression for at least 9 weeks without significant difference in morphology compared with untreated 293T cells.
Conclusion: The results suggest that all five vectors can stably transduce 293T cells, producing long term transgene expression with different efficiencies. However, neither the insulator nor the UCOE improved the GFP expression. The vectors containing the promoter CMV or CMV (β-globin intron) generated the highest gene expressions, manifesting as more favorable candidates for recombinant protein production in HEK293T cells.
Keywords: lentiviral vector, HEK 293 cells, protein production, UCOE, insulator, promoters, HCV E1.
Introduction
Currently, several protein production platforms such as bacterial, yeast, insect and mammalian cell culture expressing systems are available for quick manufacturing of large quantities of recombinant proteins. Bacterial systems are the oldest and most widely used expression platforms. Although endowed with advantages such as simplicity, speed and cost-effectiveness, they are not suitable for producing eukaryotic proteins due to issues of poor solubility-induced aggregation or misfolding or lack of proper post-translational modifications necessary for full biological activity [1, 2]. Yeast-based protein expression systems often achieve higher yield than bacterial systems, and are able to express complex proteins and perform certain post-translational modifications [3, 4]. Insect cell systems have become popular for expressing complex recombinant proteins while stop short of recapitulating complex mammalian N-glycans containing galactose or sialic acid residues [5, 6].
Mammalian cell line-based platforms seem to bias these problems and have produced many recombinant biomedical proteins with fully biological activity. But the methods can be tedious, time consuming (often taking months), and costly. Selection of stable and high expressing clones from transient transfections or lentiviral transductions is the key to the success of mammalian cell line-based protein production. Transient expression systems are expensive, often inefficient, and prone to loss of expression. In comparison, lentiviral vectors enjoy some advantages over the other vectors such as the ability to infect both dividing and non-dividing cells, irreversible integration in the genome, and a large genomic capacity [7-11].
Here, we evaluated five lentiviral vectors containing GFP gene and a combination of different promoters, HS4 insulator, or UCOE upon transducing the HEK 293T cells in terms of the stability and the efficiency of GFP expressing using fluorescent microscopy and flow cytometry. The results showed that all the five vectors induced long-term GFP expression with different efficiencies. Neither the insulator nor UCOE affected GFP expression, but the vectors containing the CMV promoter (with or without beta-globin intron) yielded high and prolonged transgene expression, demonstrating the potential as a practical protein production vector. Furthermore, we used a lentiviral vector LV-CMV-E1 containing hepatitis C virus (HCV) E1 gene to transduce 293T cells and established the recombinant cell lines with sustaining stable expression of E1 protein by limiting dilution.
Materials and Methods
Plasmids
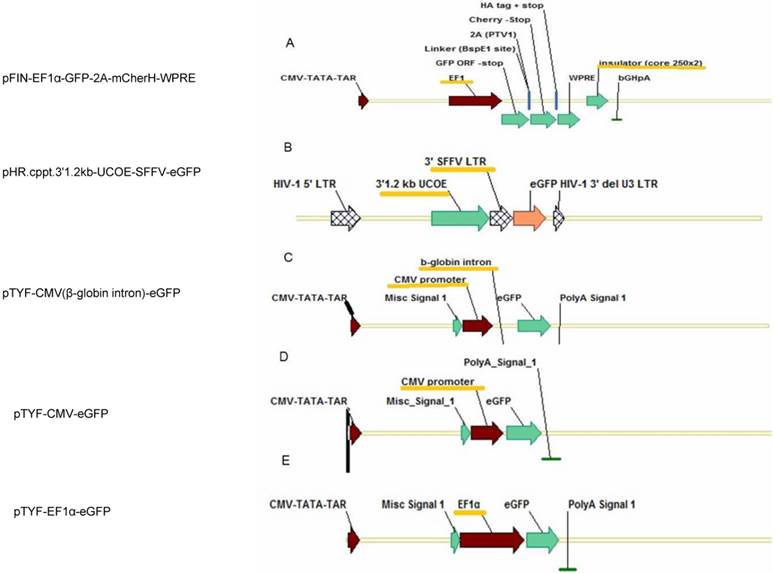
Five lentiviral vectors were used in this study (Figure 1). pFIN-EF1α-GFP-2A-mCherH-WPRE containing EF1α promoter and HS4 insulator was kindly provided by Dr. Susan Semple-Rowland (Department of Neuroscience, University of Florida) [12]. p'HR.cppt.3'1.2kb-UCOE-SFFV-eGFP containing SFFV promoter and UCOE was constructed previously in the Dr. Adrian Thrasher's laboratory (Institute of Child Health, University College London, UK) [13]. pTYF-CMV (β-globin intron)-eGFP containing CMV promoter and β-globin intron, pTYF-CMV-eGFP only containing CMV promoter, and pTYF-EF1α-eGFP with EF1α promoter were constructed in this lab. The packaging plasmids pMD2.G and psPAX2 were obtained from Dr. Junming Yue (Department of Pathology, University of Tennessee Health Science Center).
Schemes of the lentiviral transfer vectors used in the study. (A) pFIN-EF1α-GFP-2A-mCherH-WPRE containing EF1α promoter and HS4 insulator, (B) p'HR.cppt.3'1.2kb-UCOE-SFFV-eGFP containing SFFV promoter and UCOE, (C) pTYF-CMV(β-globin intron)-eGFP containing CMV promoter and β-globin intron, (D) pTYF-CMV-eGFP containing CMV promoter, and (E) pTYF-EF1α-eGFP with EF1α promoter. Abbreviations: EF1 α - elongation factor 1 α promoter; SFFV, spleen focus forming virus (SFFV) promoter; GFP - green fluorescent protein; mCher - cherry fluorescent protein; 2A - porcine teschovirus (pTV1) 2A-like cleavage peptide; bGH-bovine growth hormone derived polyadenylation signal.
Cell Cultures
Human embryonic kidney ( HEK) 293T cells (ATCC, Manassas, VA) were cultured in Dulbecco's modified Eagle's medium (DMEM) (Hyclone, Logan, UT) supplemented with 10% fetal bovine serum (FBS) (Hyclone), 100 units/ml penicillin, 100 mg/ml streptomycin (Invitrogen, Carlsbad, CA, USA) and maintain in 5% CO2 at 37℃.
Recombinant lentivirus packaged and titered
The lentiviral vectors were packaged adopting the three-plasmid packaging system [14]. On day one, a total of 6x106 293T cells were seeded in a 100 mm dish. On day 2, a transfection mix was made as the following: a solution of 500uL was first prepared consisting of 1.25 μg of shuttle plasmid pMD2.G, 3.75 μg of packaging plasmid psPAX2, 5 μg of transfer expression plasmid DNA, and 125 μl of 2 mM CaCl2 in deionized distilled water; CaCl2/DNA was then added dropwise while vortexing to equal volume of 2xHBS for a total of 1mL. This mix was added to the dish, and the cells maintained in 5% CO2 at 37℃. The GFP expression was observed by fluorescent microscopy after 24h, and the packaged recombinant lentiviruses were harvested from the supernatant of cell cultures at 48h post transfection. The lentivirus RNA was prepared using the AxyPrepTM Body Fluid viral DNA/RNA prepmini Kit (Axygen, CHN) and treated by DNase I (Promega, US) digestion. Reverse transcription was performed afterwards using iScript™ cDNA Synthesis Kit (Biorad, US). Viral load of recombinant lentivirus was quantified in copies/ml by real-time PCR (Takara Biotechnology Co., Ltd, Dalian, China) based on a standard curve generated from plasmid pTYF-EF1a-GFP using the following primers: Primer LV-F: 5′-TAAAGCTTGCCTTGAGTGCT-3′, Primer LV-R 5′-GTCTGAGGGATCTCTAGTTACCAG-3′.
Cell transduction in vitro and detection of GFP expression
A total of 4x104 293T cells /well were prepared in a 24-wells plate. On the following day, the cells in each well were transduced with packaged recombinant lentivirus at an MOI of 1000 (1000 viral genomes per cell) in DMEM medium containing 10% FBS with 6-8 μg /ml hexadimethrine bromide (Polybrene, Sigma, Germany). After 24h, transduction media was replaced with fresh DMEM with 10% FBS and incubated for 3-5 days at 37℃ and 5% CO2. The transduced cells were passaged once every three days at a ratio of 1:10. The fluorescence from the eGFP was examined under an Olympus Model BX41 fluorescent microscope (Olympus, Tokyo, Japan) and provided a marker for evaluating transgene expression of the transduced cells. The transduction efficiency of lentivirus-GFP and mean fluorescence intensities were measured by a FACS Calibur flow cytometer (Becton Dickinson, MA, USA) at 5 and 9 weeks post transduction.
Construction of HCV E1 lentiviral vector and generation of E1-expressing clonal cell lines
The HCV E1 gene was cloned into an optimal lentiviral vector pLV-CMV-eGFP in place of the eGFP gene, resulting in pLV-CMV-E1. Lentivirus was produced and titered as described above. 293T cells were transduced with LV-CMV-E1 at MOI=100 and incubated at 37℃, in 8% CO2 for 3-5 days. The transduced cells were then clonally expanded by limiting dilution. The cells were plated in three plates at a density of 0.8 cell/well in 100μl of Dulbecco's modified Eagle's medium containing 10% FBS. Two to three weeks later, clones in good condition were picked and cultured. The cells were passaged once every three days at a ratio of 1:10. Gene expression of the transduced cells was evaluated by Western Blot using an anti-HCV polyclonal antibody, which was produced in our laboratory.
Statistical Analysis
All data were presented as the mean ±SD. SPSS20.0 software was used for data analysis. The differences in mean values of the efficiencies and fluorescence intensities between different vectors were analyzed by one-way ANONA test. p<0.05 was considered to be statistically significant.
Results
Generation of recombinant lentiviral vectors
The five lentiviral vectors: pFIN-EF1α-GFP-2A-mCherH-WPRE, p'HR.cppt.3'1.2kb-UCOE-SFFV-eGFP, pTYF-CMV (β-globin intron)-EGFP, pTYF-CMV-eGFP, and pTYF-EF1α-eGFP were packaged with packaging plasmids pMD2.G and psPAX2. The viral loads of the vectors as quantified by real-time PCR from the supernatants of transfected cells were 2.8x108, 3.7x108, 2.5x108, 1.0x109, 2.5x108 copies/ml, respectively.
Lentiviral vectors TYF-CMV (β-globin intron)-eGFP and TYF-CMV-eGFP mediated high level of GFP expression in HEK 293T cells
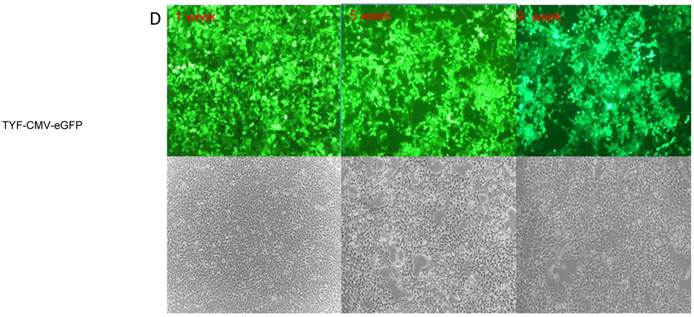
To evaluate the lentiviral vectors for the production of recombinant protein in HEK 293T cells, we compared transduction efficiencies and expression strengths of the five tested vectors (Table 1, Figures 2 & 3). We found that vectors containing the CMV promoter (with or without β-globin intron) resulted in the highest initial levels of GFP expression in HEK-293T cells (p<0.01). The TYF-CMV-eGFP vector displayed a similar extent of GFP expression as compared to TYF-CMV (β-globin intron)-eGFP at 5 weeks post transduction (p>0.1), but less than the latter at 9 weeks (p<0.05).
The GFP positive efficiency, MFI and cell Survival rates of HEK 293T cells transduced with lentiviral vectors
| vector | Week | GFP positive efficiency (%) | Survival rates (%) | GFP MFI |
|---|---|---|---|---|
| FIN-EF1α-GFP-2A-mCherH-WPRE | 5w | 22.7±3.3 | 90.4±3.4 | 6391.7±1030.4 |
| 9w | 27.6±6.9 | 96.5±3.1 | 1985.7±67.4 | |
| HR.cppt.3'1.2kb-UCOE-SFFV-eGFP | 5w | 5.8±0.4 | 90.2±4.0 | 1436.0±1.4 |
| 9w | 5.9±0.2 | 82.1±1.3 | 386.5±5.0 | |
| TYF-CMV(β-globin intron)-eGFP | 5w | 54.1±3.7 | 83.4±3.1 | 21845.7±1959.0 |
| 9w | 76.5±2.0 | 97.5±7.2 | 12814.7±1703.6 | |
| TYF-CMV-eGFP | 5w | 57.6±7.8 | 86.2±3.8 | 26596.7±3900 |
| 9w | 91.7±1.7 | 87.5±3.5 | 21192±882.7 | |
| TYF-EF1α-eGFP | 5w | 38.7±1.1 | 85.2±1.9 | 9467.7±134.8 |
| 9w | 38.2±3.9 | 87.8±2.3 | 1664.7±113.4 |
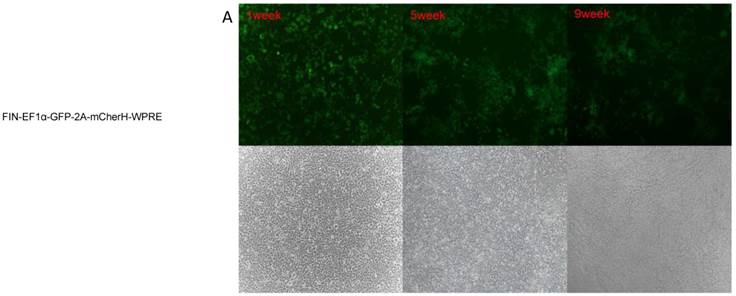
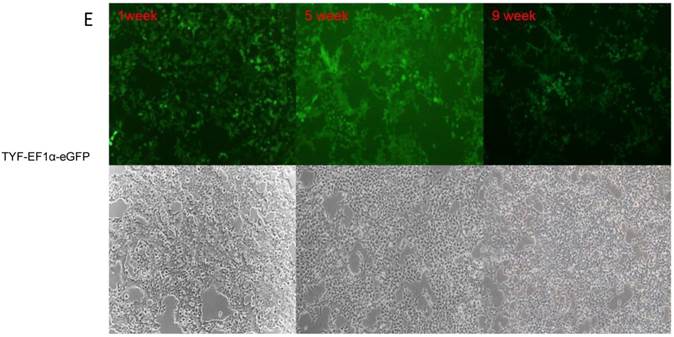
Lentiviral-mediated transduction of GFP into HEK 293T cells. HEK 293T cells were transduced with lentiviral vectors FIN-EF1α-GFP-2A-mCherH-WPRE (A), HR.cppt.3'1.2kb-UCOE-SFFV-eGFP (B) , TYF-CMV (β-globin intron)-eGFP (C), TYF-CMV-eGFP (D) or TYF-EF1α-eGFP (E) as described in the method. The images were captured by a fluorescent (upper panels) or bright light (lower panels) microscope at 1 week, 5 weeks and 9 weeks post transduction using same exposure time.


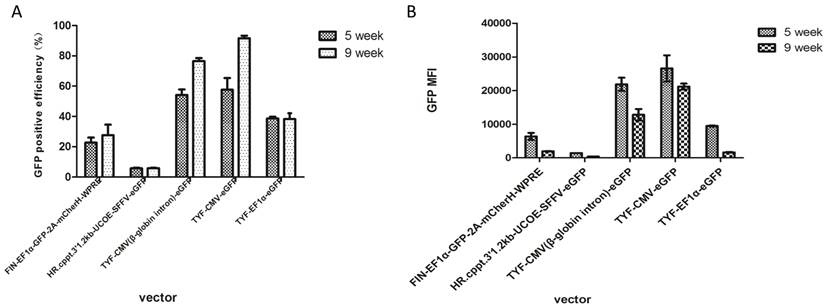
Comparison of different lentiviral vectors containing different elements on GFP positive efficiency and GFP MFI. Flow cytometry was performed at 5 weeks and 9 weeks after HEK 293T cells were transduced with lentiviral vectors. (A) GFP positive efficiency at 5 weeks post transduction. (B) The GFP MFI at 5 weeks and 9 weeks post transduction.
Lentiviral vectors generated stable long-term foreign gene expression in 293T cells
Stability of recombinant cell lines is essential to recombinant protein production. In this study, transduced 293T cells were passaged and observed under a fluorescent microscope for consistent exposure times for 9 weeks following transduction. GFP expressions reached the highest levels between passages 3 to 5 and lasted for more than 5 weeks with all five vectors. (Figure 2). Similarly, transduction efficiencies in all five lentivirus-transduced cells were stable beyond 5 weeks. The GFP expression in 293T cells transduced with TYF-CMV (β-globin intron)-eGFP or TYF-CMV-eGFP persisted, demonstrating less than 50% decrease at 9 weeks relative to that at 5 weeks (p>0.05). Furthermore, the survival rates of all transduced cells were more than 80% at 5 or 9 week time point (Table 1). The mean fluorescent intensities and transduction efficacies of the vectors were measured at either time point by flow cytometry and analyzed by SPSS 20.0 (Table 1, Figure 3). At five weeks, the mean positive rates of GFP expression were 22.7%, 5.8%, 54.1%, 57.6% and 38.7%, respectively; while the mean fluorescent signal intensities (MFI) were 6392, 1436, 21846, 26597, and 9468, respectively. At nine weeks, the mean positive rates of GFP were 27.6%, 5.9%, 76.5%, 91.7% and 38.2%, respectively, and the MFIs were 1986, 387, 12815, 21192 and 1665, respectively.
Effects of UCOE or HS4 insulator on the protein expressing yield and stability in lentiviral vectors-transduced 293T cells
We investigated the role of HS4 insulator and UCOE in protein production in lentiviral vector-transduced 293T cells. Unexpectedly, they did not enhance GFP expression level or stability (Table 1, Figure 2&3). FIN-EF1α-GFP-2A-mCherH-WPRE, containing the HS4 insulator, exhibited a similar GFP expression transduction efficiency at 5 and 9 weeks post transduction when compared with that of TYF-EF1α-eGFP (p>0.1). The vector 'HR.cppt.3'1.2kb-UCOE-SFFV-GFP, containing a UCOE, elicited much lower transduction efficiency and GFP expression than the other vectors.
Clonal cell lines of HCV E1 sustained stable expression of E1
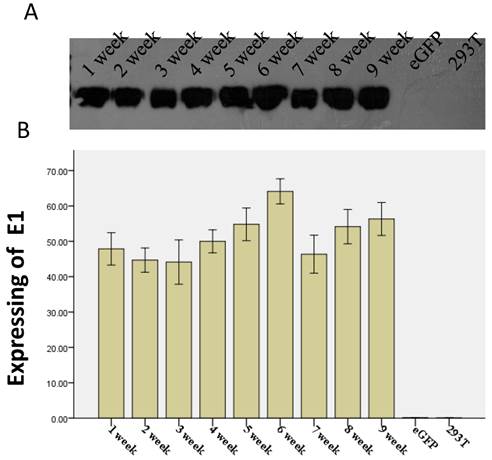
The coding sequence of HCV E1 was successfully cloned into the optimized pLV-CMV-eGFP vector, resulting in the pLV-CMV-E1 vector. The 293T cells were transduced with LV-CMV-E1 and clonal cell lines of HCV E1 were generated by limiting dilution. A representative recombinant cell line maintained robust E1 expression for at least 9 weeks without significant difference in morphology, compared with untreated 293T cells (Figure 4).
The sustaining expression of HCV E1 in clonal 293T cell lines established using the LV-CMV-E1 vector. Five microliters of medium, collected from a representative cell clone of HCV E1 during 9 weeks, were separated on SDS-PAGE and analyzed by western blotting with an anti-HCV polyclonal antibody. (A) A representative western blot showing sustaining expressing of HCV E1 from 1 week to 9 weeks. (B) Bar graphs showing the expression of HCV E1 levels in each time point. Data are means ±S.E.M. from 3 experiments. Y-axis is HCV E1 protein levels expressed as relative optical density values in arbitrary units. eGFP: the 293T cell transduced by LV-CMV-eGFP; 293T: untreated 293T cells.
Discussion
Recombinant therapeutic proteins produced in mammalian cells represent a major class of biopharmaceuticals. Establishing stable and high-production recombinant cell lines remains among the top priorities for protein manufacturers [15]. Decigram quantities of highly purified, recombinant proteins can be made by lentiviral vectors according to many reports [11, 14, 16-18]. To aid in the optimization of the lentiviral vectors for recombinant protein production specifically in the HEK 293T cell line that has been used extensively as an expression tool for recombinant proteins, we evaluated 5 unique combinations of various promoters, an insulator, and an UCOE with respect to transduction efficiency and expression strength. The results indicated that all five vectors are capable of stably transducing 293T cells and producing lasting expression of recombinant proteins with different efficiencies. The CMV promoter (with or without β-globin intron) resulted in the highest initial level of GFP expression amongst the vectors, indicating its ability for rapid and high-level transgenic expression in 293T cell line. This finding pinpoints to the importance of a proper choice of promoter in achieving high yield of transgene production in recombinant cell lines [19].
Chromatin insulators separate active transcriptional domains in the genome to allow differential regulation of genes and prevent the spread of heterochromatin towards active transcriptional units [20, 21]. The role of insulator DNAs is to isolate genetic loci by blocking interactions between cis regulatory elements and inappropriate target promoters in neighboring transcription units [22, 23]. Insulators disrupt the communication between a regulatory sequence such as an enhancer or a silencer, and a promoter [24]. Such a function has been employed to potentially promote transgene expression in cell-line-based recombinant protein production systems. For example, the full-length chicken hypersensitive site-4 (cHS4) insulator element was previously shown to protect viral vectors against chromosomal position effects [25-28] and augment foreign gene expression in viral vectors-transduced cells [29-31]. Other studies demonstrated that 250-bp “core” element from within 5′HS4 is sufficient to protect against silencing of transgenes caused by chromosomal position effect (CPE). Two or more copies of the cHS4 core should have the similar protection activity as the full-length cHS4 in plasmid transfections [32, 33] , but they are unstable in lentiviral vectors. The full activity of insulator can be restored when the 250-bp core is combined with the 3' 400bp cHS4 sequence, which also has an unique and significant insulator activity [34] In the current study, the HS4 insulator did not impact GFP expression level or stability as compared to 293T cells transduced with the lentiviral vector FIN-EF1α-GFP-2A-mCherH-WPRE that contains two copies of the cHS4 5' 250-bp core, or the TYF-EF1α-EGFP vector that does not have an insulator. These negative outcomes suggest the limitation of cHS4 5' 250-bp core in the lentiviral vector on improving foreign gene expression in transduced 293T cells. On the other hand, the '650 bp' cHS4 sequence consists of a 3' 400 bp core combined with the 5' 250 bp core may provide a workable solution because the 3' 400 bp is supposedly to interact with the 5' 250 bp to provide effective insulation and improve the lentiviral vector-mediated foreign gene expression in 293T cells. This aspect warrants new investigations [37].
Ubiquitous chromatin opening elements (UCOEs) have been reported to drive high levels of protein expression over extended periods of time in in vitro transfection settings [18, 35, 36]. UCOEs have been incorporated into lentiviral vectors in order to prolong the expression of integrated transgenes in gene therapy experiments [13, 37]. In Freestyle 293-F cells, the inclusion of the minimized UCOEs in the transduction vectors critically helped maintain the stability of decigram levels of protein expression (eg. murine Siderocalin) [18]. In this study, the GFP expression was unaffected by the use of UCOE in the lentiviral vector in HEK 293T cells. In fact, the protein yield with the vector including the UCOE and SFFV promoter was significantly lower than that of with the other vectors. This indicates that UCOEs may function differently in different types of cells.
In this report, we used the vector LV-CMV-E1 containing HCV E1 to transduce 293T cells and established the recombinant cell lines by limiting dilution in less than 20 days. The recombinant cells could stably produce recombinant proteins. Moreover, in comparison with transient transfection, where production occurred for a short period of 2-6 days [38], our results showed that the expression level of E1 was stable for at least 9 weeks. This stems most likely from the fact that the optimized vector derived from lentivirus are relatively resistant to silencing.
In summary, the five lentiviral vectors evaluated in this study stably transduced 293T cells and resulted in long term transgene expression. The promoters served as the determining factor for the efficacy of the vectors. The vectors containing the promoter CMV or CMV (β-globin intron) generated the highest gene expressions amongst the group, while the insulator and UCOE did not improve GFP expression. These findings provide valuable insights that will guide our effort in optimizing lentiviral vectors for recombinant protein production specifically in HEK 293T cells. This lentiviral protein expressing system is simple and highly efficient and may be adapted for bio-therapy protein manufacturing and other biotechnology applications.
Acknowledgements
We thank Dr. Susan Semple-Rowland (Department of Neuroscience, University of Florida) and Dr. Adrian Thrasher (Institute of Child Health, University College London, UK) for providing the vector pFIN-EF1α-GFP-2A-mCherH-WPRE and p'HR.cppt.3'1.2kb-UCOE-SFFV-eGFP. We acknowledge Wenmin Li, Xiusheng Chen, Yang Xu, Si Chen, Shengyao Wang, Xin Nie, and Ao Hu for their technical assistance and help with Western blot and real-time PCR.
This work was supported by National 863 High Technique Development Project of China Grant 2012AA02A403 (H.L.), the Industrial High-tech Fields of Science and Technology Plan Projects of Guangdong Province grant 2013B010404026, National Natural Science Foundation of China Grant 81072113 (H.L.) and 81401920 (H.D) and 2013 Specialized Research Fund for the Doctoral Program of Higher Education (SRFDP) 20134433120020 (H.D).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Hannig G, Makrides SC. Strategies for optimizing heterologous protein expression in Escherichia coli. Trends Biotechnol. 1998;16(2):54-60
2. Baneyx F, Mujacic M. Recombinant protein folding and misfolding in Escherichia coli. Nature biotechnology. 2004;22(11):1399-1408
3. Daly R, Hearn MT. Expression of heterologous proteins in Pichia pastoris: a useful experimental tool in protein engineering and production. Journal of molecular recognition: JMR. 2005;18(2):119-138
4. Caterina Holz, Bianka Prinz, Natalia Bolotina, Volker Sievert, Konrad Büssow, Bernd Simon, Ulf Stahl, Lang C. Establishing the yeast Saccharomyces cerevisiae as a system for expression of human proteins on a proteome-scale. J Struct Funct Genomics. 2003;4(2-3):97-108
5. Kost TA, Condreay JP, Jarvis DL. Baculovirus as versatile vectors for protein expression in insect and mammalian cells. Nature biotechnology. 2005;23(5):567-575
6. Chang G-D, Chen C-J, Lin C-Y, Chen H-C, Chen H. Improvement of glycosylation in insect cells with mammalian glycosyltransferases. Journal of biotechnology. 2003;102(1):61-71
7. Gaillet B, Gilbert R, Amziani R, Guilbault C, Gadoury C, Caron AW, Mullick A, Garnier A, Massie B. High-level recombinant protein production in CHO cells using an adenoviral vector and the cumate gene-switch. Biotechnology progress. 2007;23(1):200-209
8. Oberbek A, Matasci M, Hacker DL, Wurm FM. Generation of stable, high-producing CHO cell lines by lentiviral vector-mediated gene transfer in serum-free suspension culture. Biotechnology and bioengineering. 2011;108(3):600-610
9. Klimatcheva E, Planelles V, Day SL, Fulreader F, Renda MJ, Rosenblatt J. Defective lentiviral vectors are efficiently trafficked by HIV-1 and inhibit its replication. Molecular therapy: the journal of the American Society of Gene Therapy. 2001;3(6):928-939
10. Ansorge S, Lanthier S, Transfiguracion J, Durocher Y, Henry O, Kamen A. Development of a scalable process for high-yield lentiviral vector production by transient transfection of HEK293 suspension cultures. The journal of gene medicine. 2009;11(10):868-876
11. Gaillet B, Gilbert R, Broussau S, Pilotte A, Malenfant F, Mullick A, Garnier A, Massie B. High-level recombinant protein production in CHO cells using lentiviral vectors and the cumate gene-switch. Biotechnology and bioengineering. 2010;106(2):203-215
12. Verrier JD, Madorsky I, Coggin WE, Geesey M, Hochman M, Walling E, Daroszewski D, Eccles KS, Ludlow R, Semple-Rowland SL. Bicistronic lentiviruses containing a viral 2A cleavage sequence reliably co-express two proteins and restore vision to an animal model of LCA1. PloS one. 2011;6(5):e20553
13. Zhang F, Frost AR, Blundell MP, Bales O, Antoniou MN, Thrasher AJ. A ubiquitous chromatin opening element (UCOE) confers resistance to DNA methylation-mediated silencing of lentiviral vectors. Molecular therapy: the journal of the American Society of Gene Therapy. 2010;18(9):1640-1649
14. Durocher Y. High-level and high-throughput recombinant protein production by transient transfection of suspension-growing human 293-EBNA1 cells. Nucleic Acids Research. 2002;30(2):9e-9
15. Matasci M, Baldi L, Wurm FM. Recombinant therapeutic protein production in cultivated mammalian cells: current status and future prospects. Drug Discovery Today: Technologies. 2008;5:2-3
16. da Rosa NG, Swiech K, Picanco-Castro V, Russo-Carbolante EM, Soares Neto MA, de Castilho-Fernandes A, Faca VM, Fontes AM, Covas DT. SK-HEP cells and lentiviral vector for production of human recombinant factor VIII. Biotechnology letters. 2012;34(8):1435-1443
17. Spencer HT, Denning G, Gautney RE, Dropulic B, Roy AJ, Baranyi L, Gangadharan B, Parker ET, Lollar P, Doering CB. Lentiviral vector platform for production of bioengineered recombinant coagulation factor VIII. Molecular therapy: the journal of the American Society of Gene Therapy. 2011;19(2):302-309
18. Bandaranayake AD, Correnti C, Ryu BY, Brault M, Strong RK, Rawlings DJ. Daedalus: a robust, turnkey platform for rapid production of decigram quantities of active recombinant proteins in human cell lines using novel lentiviral vectors. Nucleic acids research. 2011;39(21):e143
19. Zhang L, Yin S, Tan W, Xiao D, Weng Y, Wang W, Li T, Shi J, Shuai L, Li H. et al. Recombinant interferon-gamma lentivirus co-infection inhibits adenovirus replication ex vivo. PloS one. 2012;7(8):e42455
20. Felsenfeld G, Burgess-Beusse B, Farrell C, Gaszner M, Ghirlando R, Huang S, Jin C, Litt M, Magdinier F, Mutskov V. et al. Chromatin boundaries and chromatin domains. Cold Spring Harbor symposia on quantitative biology. 2004;69:245-250
21. Neff T, Shotkoski F, Stamatoyannopoulos G. Stem cell gene therapy, position effects and chromatin insulators. Stem cells. 1997;15(Suppl 1):265-271
22. Chung JH, Whiteley M, Felsenfeld G. A 5′ element of the chicken β-globin domain serves as an insulator in human erythroid cells and protects against position effect in Drosophila. Cell. 1993;74(3):505-514
23. Kellum R, Schedl P. A position-effect assay for boundaries of higher order chromosomal domains. Cell. 1991;64(5):941-950
24. Bushey AM, Dorman ER, Corces VG. Chromatin insulators: regulatory mechanisms and epigenetic inheritance. Molecular cell. 2008;32(1):1-9
25. Arumugam PI, Scholes J, Perelman N, Xia P, Yee JK, Malik P. Improved human beta-globin expression from self-inactivating lentiviral vectors carrying the chicken hypersensitive site-4 (cHS4) insulator element. Molecular therapy: the journal of the American Society of Gene Therapy. 2007;15(10):1863-1871
26. Emery DW, Yannaki E, Tubb J, Nishino T, Li Q, Stamatoyannopoulos G. Development of virus vectors for gene therapy of beta chain hemoglobinopathies: flanking with a chromatin insulator reduces gamma-globin gene silencing in vivo. Blood. 2002;100(6):2012-2019
27. Rivella S, Callegari JA, May C, Tan CW, Sadelain M. The cHS4 Insulator Increases the Probability of Retroviral Expression at Random Chromosomal Integration Sites. Journal of virology. 2000;74(10):4679-4687
28. Yannaki E, Tubb J, Aker M, Stamatoyannopoulos G, Emery DW. Topological constraints governing the use of the chicken HS4 chromatin insulator in oncoretrovirus vectors. Molecular therapy: the journal of the American Society of Gene Therapy. 2002;5(5 Pt 1):589-598
29. Steinwaerder DS, Lieber A. Insulation from viral transcriptional regulatory elements improves inducible transgene expression from adenovirus vectors in vitro and in vivo. Gene therapy. 2000;7(7):556-567
30. Inoue T, Yamaza H, Sakai Y, Mizuno S, Ohno M, Hamasaki N, Fukumaki Y. Position-independent human beta-globin gene expression mediated by a recombinant adeno-associated virus vector carrying the chicken beta-globin insulator. Journal of human genetics. 1999;44(3):152-162
31. Ramezani A, Hawley TS, Hawley RG. Performance- and safety-enhanced lentiviral vectors containing the human interferon-beta scaffold attachment region and the chicken beta-globin insulator. Blood. 2003;101(12):4717-4724
32. Recillas-Targa F, Pikaart MJ, Burgess-Beusse B, Bell AC, Litt MD, West AG, Gaszner M, Felsenfeld G. Position-effect protection and enhancer blocking by the chicken beta-globin insulator are separable activities. Proceedings of the National Academy of Sciences of the United States of America. 2002;99(10):6883-6888
33. H. J, Chung, Adam C. Bell, Felsenfeld aG: Characterization of the chicken betaglobin insulator. Proc Natl Acad Sci. 1997;94:575-580
34. Arumugam PI, Urbinati F, Velu CS, Higashimoto T, Grimes HL, Malik P. The 3' region of the chicken hypersensitive site-4 insulator has properties similar to its core and is required for full insulator activity. PloS one. 2009;4(9):e6995
35. Antoniou M, Harland L, Mustoe T, Williams S, Holdstock J, Yague E, Mulcahy T, Griffiths M, Edwards S, Ioannou PA. et al. Transgenes encompassing dual-promoter CpG islands from the human TBP and HNRPA2B1 loci are resistant to heterochromatin-mediated silencing. Genomics. 2003;82(3):269-279
36. Williams S, Mustoe T, Mulcahy T, Griffiths M, Simpson D, Antoniou M, Irvine A, Mountain A, Crombie R. CpG-island fragments from the HNRPA2B1/CBX3 genomic locus reduce silencing and enhance transgene expression from the hCMV promoter/enhancer in mammalian cells. BMC biotechnology. 2005;5:17
37. Zhang F, Thornhill SI, Howe SJ, Ulaganathan M, Schambach A, Sinclair J, Kinnon C, Gaspar HB, Antoniou M, Thrasher AJ. Lentiviral vectors containing an enhancer-less ubiquitously acting chromatin opening element (UCOE) provide highly reproducible and stable transgene expression in hematopoietic cells. Blood. 2007;110(5):1448-1457
38. Subramanian S, Srienc F. Quantitative analysis of transient gene expression in mammalian cells using the green fluorescent protein. Journal of biotechnology. 1996;49(1-3):137-151
Author contact
Corresponding author: Haifa Zheng, Ph.D., Beijing Minhai Biotechnology CO.LTD, No.1 Simiao Road, Biotechnology and Pharmaceuticals Industrial Base, Daxing District, Beijing 102600, China. Phone: 86-10-59613588; E-mail: zhenghaifacom. Hongwei Li, Ph.D., School of Biotechnology, Southern Medical University, 1023 South Shatai Road, Guangzhou, Guangdong 510515, China. Phone: 86-20-61648555; Fax: 86-20-61648555; E-mail: hongwei1com

 Global reach, higher impact
Global reach, higher impact