3.2
Impact Factor
ISSN: 1449-1907
Int J Med Sci 2014; 11(9):918-924. doi:10.7150/ijms.8850 This issue Cite
Research Paper
Renal Transplantation: Relationship between Hospital/Surgeon Volume and Postoperative Severe Sepsis/Graft-Failure. A Nationwide Population-Based Study
1. Departments of Medical Research, Chi-Mei Medical Center, Tainan, Taiwan.
2. Departments of Anesthesiology, Chi-Mei Medical Center, Tainan, Taiwan.
3. Departments of Nephrology, Chi-Mei Medical Center, Tainan, Taiwan.
4. Department of Hospital and Health Care Administration, Chia Nan University of Pharmacy and Science, Tainan, Taiwan.
5. Department of Food Nutrition, Chung Hwa University of Medical Technology, Tainan, Taiwan.
6. Department of Health Care Management, National Taipei University of Nursing and Health Sciences, Taiwan.
* These authors made equal contributions to this paper.
Received 2014-2-16; Accepted 2014-6-10; Published 2014-6-24
Abstract
Background and objects: We explored the relationship between hospital/surgeon volume and postoperative severe sepsis/graft-failure (including death).
Methods: The Taiwan National Health Insurance Research Database claims data for all patients with end-stage renal disease patients who underwent kidney transplantation between January 1, 1999, and December 31, 2007, were reviewed. Surgeons and hospitals were categorized into two groups based on their patient volume. The two primary outcomes were severe sepsis and graft failure (including death). The logistical regressions were done to compute the Odds ratios (OR) of outcomes after adjusting for possible confounding factors. Kaplan-Meier analysis was used to calculate the cumulative survival rates of graft failure after kidney transplantation during follow-up (1999-2008).
Results: The risk of developing severe sepsis in a hospital in which surgeons do little renal transplantation was significant (odds ratio [OR]; p = 0.0115): 1.65 times (95% CI: 1.12-2.42) higher than for a hospital in which surgeons do many. The same trend was true for hospitals with a low volume of renal transplantations (OR = 2.39; 95% CI: 1.62-3.52; p < 0.0001). The likelihood of a graft failure (including death) within one year for the low-volume surgeon group was 3.1 times higher than for the high-volume surgeon group (p < 0.0001); the trend was similar for hospital volume. Female patients had a lower risk than did male patients, and patients ≥ 55 years old and those with a higher Charlson comorbidity index score, had a higher risk of severe sepsis.
Conclusions: We conclude that the risk of severe sepsis and graft failure (including death) is higher for patients treated in hospitals and by surgeons with a low volume of renal transplantations. Therefore, the health authorities should consider exporting best practices through educational outreach and regulation and then providing transparent information for public best interest.
Keywords: graft failure, population-based, renal transplantation, sepsis, volume-outcome relationship.
Introduction
Studies on surgeons and hospitals with higher caseloads provide evidence of better outcomes in major surgery, especially in cancer [1-3]; however, the association is controversial in different healthcare systems [4] and some types of surgery. Despite the benefits of volume, the controversy claimed it is due to the concentration of hospital care supply and also concerned medical skills loss in lower level hospital and patients will flow to higher level hospital.
Graft rejection and infection were the two major causes of death in renal transplant recipients from the 1970s until the mid-1980s [5]. Because of improved immunosuppressive protocols and surgical techniques, the incidence of graft rejection has impressively decreased [6]. Despite recent advances in management and therapy, postoperative infection remains high, around 40% [7-9], and exceeds acute rejection as the leading cause of hospitalization in renal transplant recipients with a functioning allograft [10]. Sepsis is a systemic and deleterious host response to infection, and severe sepsis is defined as sepsis with acute organ dysfunction, hypoperfusion (including oliguria, lactic acidosis, or encephalopathy), or hypotension [11, 12]. The incidence of severe sepsis after most elective operative procedures increased from 0.3% in 1997 to 0.9% in 2006 [13]. In the U.S., organ transplant recipients are at an especially high risk for developing severe sepsis [14]. It is the most common life-threatening complication of long-term immunosuppressive therapy and is the main reason for intensive care unit (ICU) admission of renal transplant recipients [8]. An ICU admission is associated with a decreased graft longevity rate and a higher post-transplantation mortality rate [8, 15]. Moreover, patients with severe sepsis might develop multiple septic episodes during the same hospitalization or after discharge [16, 17].
Multiple factors are associated with the development of severe sepsis after kidney transplantation, e.g., the intensity of exposure to potential pathogens (epidemiologic exposure) and the combined effect of all of the factors that contribute to a patient's susceptibility to infection [18]. Moreover, many factors contribute to the development of severe sepsis [18]. Preventing and managing severe sepsis places extraordinary demands, not only on surgeons, but also on other medical personnel, including anesthesiologists, diagnostic and interventional radiologists, critical care specialists, nursing, and nutritional support service workers.
The level of the surgeons' transplantation experience and the level of the hospital transplantation teams' quality of care may significantly contribute to reducing the incidence of severe sepsis post-transplantation. Multiple studies [7, 19, 20] have shown an association between hospital volume and surgical outcomes for organ transplantations. However, the effect of surgeon or hospital volume on severe sepsis is not yet clear despite its being a key factor associated with both graft longevity and patient survival. This study aims to explore the relationship between the surgeon or hospital volume with consideration of the postoperative severe sepsis or graft-failure (including death). Therefore, we investigated this question on a nationwide scale using claims data from the Taiwan National Health Insurance Database.
Study population and Methods
Database
The data for this study were obtained from Taiwan National Health Insurance Research Database (NHIRD) of the Taiwan National Health Research Institute. The NHIRD, which covers nearly all inpatient and outpatient medical benefit claims for the Taiwanese population of over 22 million (about 99% of Taiwan's population in 2008), is one of the most comprehensive nationwide population-based data sources currently available and has been used extensively in many epidemiological studies. The NHIRD provides encrypted patient identification numbers, gender, date of birth, dates of admission and discharge, the ICD-9-CM (International Classification of Diseases, Ninth Revision, Clinical Modification) codes of diagnoses (up to five) and procedures (up to five), details of prescriptions, and expenditure amounts. It also includes a registry of contracted medical facilities, and a registry of board-certified surgeons. With ethical approval from National Health Research Institute, we used data for the ambulatory care claims, all inpatient claims, and registry for patients with catastrophic illnesses for this study. All NHI datasets can be interlinked with each individual personal identification number.
Selection of patients and variables
All patients with end-stage renal disease (ESRD) who underwent kidney transplantation between January 1, 1999, and December 31, 2007, were identified by the ICD-9 CM procedure code 55.69. Patients with unknown gender or missing data were excluded. All patients were followed-up through December 31, 2008. Seventeen hundred seventy-nine kidney transplantations were done by 142 surgeons in 35 hospitals during this period.
Physicians and hospitals were categorized by their total patient volume by using their unique identifiers in the database. The sample of 1779 patients was divided into two groups: the low-volume group (first 1/3) and the medium- and high-volume group (second 1/3 [medium-volume] and third 1/3 [high-volume]), based on physician volume: ≤ 33 transplantations (low-volume), and > 33 transplantations (medium- and high-volume), and the hospital sample was divided into two groups: ≤ 95 transplantations (low-volume), and > 95 transplantations (medium- and high-volume).
The two primary outcomes were severe sepsis and graft failure (including death). The ICD-9-CM codes for the sepsis in this study used the definition from Angus et al [21], severe sepsis was defined as sepsis complicated by organ dysfunction. The key independent variables were the kidney transplantation volumes both for physicians and for hospitals. Other physician attributes included age (≤ 40, 40-49, and ≥ 50) and gender. The hospitals were grouped by public or private ownership. Patient characteristics included age (≤ 20, 21-55, and > 55), gender, and modified Charlson Comorbidity Index score [22], which was used to infer the health status of each patient; higher sums of weighted scores indicated higher disease severity.
Statistical Analysis
Descriptive statistical analyses using Pearson χ2 tests were done to compare the characteristics of patients, physicians, and hospitals with physician volume and hospital volume. The association between medical costs and physician volume, medical costs and hospital volume, length of stay in hospital and physician volume, and length of stay in hospital and hospital volume were determined using Students' t-test. Unconditional logistic regression analyses were used assess the crude odds ratio of (1) severe sepsis and (2) graft failure (including death) at one year between the physician volume and hospital volume groups. Moreover, multivariate logistic regression using the generalized estimated equation method (GEE), which clusters hospital volume, was used to obtain the adjusted odds ratio (AOR) of graft failure (including death) at one year between the physician volume groups. GEE method accounts for the fact that patients within the same hospital volume group may be more similar to each other than they are to patients in other hospital volume groups because of specific physician and treatment practices.
Kaplan-Meier analysis was used to calculate the cumulative survival rates of graft failure (including death) after kidney transplantation in a 10-year (1999-2008) follow-up period, and the log-rank test was used to test the differences between the survival curves. Survival time was measured from the date of kidney transplantation until the day of graft failure or death or until the end of the study. SAS 9.3.1 (SAS Institute, Cary, NC) was used for all statistical analyses. The alpha value indicated significance at the 0.05 level.
Results
The mean medical cost and length of stay (hospitalization) was significantly less in the high physician-volume groups than in the low physician-volume groups (new Taiwan dollars (NT$) 195,223 vs. NT$257,495; 17.56 vs. 19.84 days, respectively; P < 0.0001). There was no significant difference in the gender of the patients, but significantly (P < 0.0001) more than 80% of the patients were in the 21-55 years old age group in both the high- and low-volume groups. Interestingly, in the low-volume group, a higher proportion of patients had more comorbidities (CCI > 2). In addition, physician attributes (gender and age), were both significantly different (P < 0.0001) between the high- and low-volume physicians (Table 1).
The distributions of patient attributes were similar when stratifying by hospital volume. Mean medical cost was significantly lower in the high-volume hospital groups than in the low-volume hospital groups (NT$205,784 vs. NT$238,175, respectively; P < 0.0001), but mean length of stay was not significantly different (Table 2).
The distributions of basic characteristics by physician volume with medical cost and length of stay.
| Physician Volume | |||
|---|---|---|---|
| Low (1-33) | High (> 33) | P-value | |
| Total number of physicians | 128 | 14 | |
| Total number of patients | 619 (34.79) | 1160 (65.21) | |
| Mean medical cost (NT$) | 257,495 ± 204,344 | 195,223 ± 126,356 | < 0.0001 |
| Mean length of stay (days) | 19.84 ± 19.64 | 17.56 ± 10.04 | 0.0013 |
| Patient attributes | |||
| Gender | |||
| Male | 308 (49.76) | 557 (48.02) | |
| Female | 311 (50.24) | 603 (51.98) | 0.4842 |
| Age (years) | |||
| ≤ 20 | 46 (7.43) | 57 (4.91) | 0.0411 |
| 21-55 | 519 (83.84) | 976 (84.14) | |
| > 55 | 54 (8.72) | 127 (10.95) | |
| CCI score | |||
| 0 | 380 (61.39) | 819 (70.61) | 0.0002 |
| 1 | 157 (25.36) | 239 (20.60) | |
| > 2 | 82 (13.25) | 102 (8.79) | |
| Physician attributes | |||
| Gender | |||
| Male | 593 (95.80) | 1160 (100.00) | < 0.0001 |
| Female | 26 (4.20) | 0 | |
| Age (years) | |||
| ≤ 40 | 206 (33.28) | 326 (28.10) | 0.0003 |
| 40-49 | 281 (45.40) | 486 (41.90) | |
| ≥ 50 | 132 (21.32) | 348 (30.00) | |
NT$, new Taiwan dollars; CCI, Charlson Comorbidity Index.
The distributions of basic characteristics by hospital volume with medical cost and length of stay.
| Hospital Volume | |||
|---|---|---|---|
| Low (≤ 95) | High (> 95) | P-value | |
| Total number of hospitals | 30 | 5 | |
| Total number of patients | 610 (34.29) | 1169 (65.71) | |
| Mean medical cost (NT$) | 238,175 ± 162,801 | 205,784 ± 158,420 | < 0.0001 |
| Mean length of stay(days) | 18.58 ± 13.80 | 18.23 ± 14.35 | 0.626 |
| Patient attributes | |||
| Gender | |||
| Male | 307 (50.33) | 611 (52.27) | |
| Female | 303 (49.67) | 558 (47.73) | 0.2986 |
| Age (years) | |||
| ≤ 20 | 26 (4.26) | 77 (6.59) | 0.0561 |
| 21-55 | 529 (86.72) | 966 (82.63) | |
| > 55 | 55 (9.02) | 126 (10.78) | |
| CCI score | |||
| 0 | 394 (64.59) | 805 (68.86) | 0.1816 |
| 1 | 149 (24.43) | 247 (21.13) | |
| > 2 | 67 (10.98) | 117 (10.01) | |
| Hospital attributes | |||
| Hospital ownership | |||
| Public | 57 (9.34) | 937 (80.15) | < 0.0001 |
| Private | 553 (90.66) | 232 (19.85) | |
NT$, new Taiwan dollars; CCI, Charlson Comorbidity Index.
One-year severe-sepsis rate with odds ratio across physician and hospital caseload-volume groups.
| Risk Factor | None | Severe Sepsis | Odds Ratio (95% CI) | P-value |
|---|---|---|---|---|
| Severe sepsis in hospital | ||||
| Physician volume | ||||
| Low | 568 (91.76) | 51 (8.24) | 1.65 (1.12-2.42) | 0.0115 |
| High | 1100 (94.83) | 60 (5.17) | 1.00 | |
| Hospital volume | ||||
| Low | 550 (90.16) | 60 (9.84) | 2.39 (1.62-3.52) | < 0.0001 |
| High | 1118 (95.64) | 51 (4.36) | 1.00 | |
| Severe sepsis within one year | ||||
| Physician volume | ||||
| Low | 542 (87.56) | 77 (12.44) | 1.61 (1.17-2.22) | 0.0033 |
| High | 1066 (91.90) | 94 (8.10) | 1.00 | |
| Hospital volume | ||||
| Low | 525 (86.07) | 85 (13.93) | 2.04 (1.48-2.80) | < 0.0001 |
| High | 1083 (92.64) | 86 (7.36) | 1.00 | |
CI, confidence interval.
One-year graft-failure (including death) rate with odds ratios across physician and hospital caseload-volume groups
| None | Death | Odds Ratio | P-value | |
|---|---|---|---|---|
| Death or graft failure within one year | ||||
| Physician Volume | ||||
| Low | 584 (94.35) | 35 (5.65) | 3.10 (1.80-5.33) | < 0.0001 |
| High | 1138 (98.10) | 22 (1.90) | 1.00 | |
| Hospital Volume | ||||
| Low | 575 (94.26) | 35 (5.74) | 3.17 (1.85-5.46) | < 0.0001 |
| High | 1147 (98.12) | 22 (1.88) | 1.00 | |
Of the patients with a kidney transplantation who developed severe sepsis, about 8.24% were in the low-volume (OR: 1.65; 95% CI: 1.12-2.42; P = 0.0115) physician group versus about 5.17% in the high-volume physician group (Table 3). Moreover, about 9.84% were in the low-volume hospital group (OR: 2.39; 95% CI: 1.62-3.52; P = 0.0001) versus about 4.36% in the high-volume hospital group. These findings remained significant even when we estimated the risk of developing severe sepsis within one year. The likelihood of death or graft failure within one year in the physician low-volume group was 3.1 times higher than in the high-volume group (95% CI: 1.80-5.33; P < 0.0001) and 3.17 times higher in the hospital low-volume (95% CI: 1.85-5.46; P < 0.0001) (Table 4).
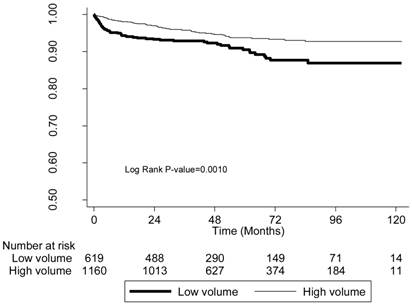
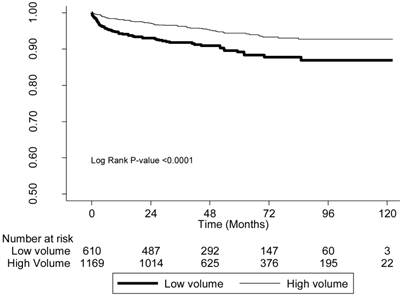
We also analyzed 10-year patient survival using a log-rank test to compare the likelihood of graft failure (including death) based on physician-volume and hospital-volume (Figures 1, 2). The low-volume groups had a significantly (P < 0.001) higher risk in both instances.
The survival curves of patients with kidneys transplanted by low-volume and high-volume physicians.
The survival curves of patients with kidneys transplanted in low-volume and high-volume hospitals.
To account for the possibility that patients within the same hospital-volume group may be more similar to each other than to patients in other hospital-volume groups because of specific physician and hospital treatment practices, we used a logistic regression model with the GEE method to explore the association of the 1-year severe sepsis risk with the same risk factors used for the other analyses. Patients treated by low-volume physicians still had 1.35 times the risk of developing severe sepsis than did patients treated by high-volume physicians. Physician gender was not a significant factor, but physician age was: patients treated by physicians ≤ 40 had a significantly lower risk (AOR: 0.633; P < 0.0001), but those treated by physicians > 50 had a significantly higher risk (AOR: 1.4; P < 0.0022). Female patients were significantly less likely to develop severe sepsis (AOR: 0.768; 95% CI: 0.619-0.953; P < 0.0001), but patients > 55 years old were significantly (P < 0.0001) more likely to develop it, as were patients with a higher CCI score (Table 5). Finally, patients treated in a private hospital were significantly (P < 0.0001) more likely to develop severe sepsis than were patients treated in a public hospital.
Risk factors for one-year severe sepsis of kidney transplantation patients.
| Risk Factor | Adjusted Odds Ratio (95% CI) | P-value |
|---|---|---|
| Physician attributes | ||
| Physician volume | 1.349 (1.254-1.452) | < 0.0001 |
| Low | ||
| High | ||
| Gender | ||
| Male | ||
| Female | 1.358 (0.449-4.109) | 0.5881 |
| Age (years) | ||
| ≤ 40 | 0.633 (0.616-0.651) | < 0.0001 |
| 40-49 | Reference | |
| ≥ 50 | 1.400 (1.129-1.737) | 0.0022 |
| Patient attributes | ||
| Gender | ||
| Male | ||
| Female | 0.768 (0.619-0.953) | 0.0165 |
| Age (years) | ||
| ≤ 20 | 1.241 (0.713-2.160) | 0.4442 |
| 21-55 | Reference | |
| > 55 | 1.411 (1.335-1.492) | < 0.0001 |
| CCI Score | ||
| 0 | ||
| 1 | 1.314 (1.216-1.421) | < 0.0001 |
| > 2 | 1.583 (1.392-1.800) | < 0.0001 |
| Hospital attributes | ||
| Hospital ownership | ||
| Public | ||
| Private | 2.537 (2.402-2.679) | < 0.0001 |
CCI, Charlson Comorbidity Index.
Discussion
In this study, we explored the relationship between physician and hospital volumes of kidney transplantations with postoperative severe sepsis and graft failure (including death). Several studies have reported serious postoperative complications because of sepsis, which is associated with a higher risk of mortality. We found that patient who had their transplantation surgery done either by high-volume physicians or in high-volume hospitals, had a lower risk of developing severe sepsis in the hospital, even within one year. Moreover, the risk of graft failure (including death) was significantly lower in the short term (within one year) and in the long term (~10 years) after a kidney transplantation in a high-volume hospital.
Since the first published article in English on the volume-outcome relationship [23], there have been numerous studies that provide similar results for research on, e.g., cardiovascular or orthopedic procedures and cancer surgery. High-volume hospitals not only have better survival rates, but they also have a lower infectious complication and reduced resource utilization [24, 25]. Scare studies in the kidney transplantation applied in the above two hypothesis (practice-makes-perfect or selective-referral), not only because the rare cases in kidney transplantation, but also the serious outcomes applied in a short time which produce the unstable predication.
The likelihood of graft failure (including death) within a year of the transplantation is significantly associated with each patient's personal characteristics, such as gender and age, and with relevant postoperative medical conditions, such as sepsis, which is consistent with other studies [26, 27]. We found that physician-volume was, indeed, related to the risk of severe sepsis, especially for the low-volume group, and recommend that health authorities consider certifying some institutions for economic reasons or to provide the transparent information about patient's choices of hospital for quality-procedures-and-outcomes reasons.
Patient characteristics, operative time, and the identity of the surgeon are perhaps the three most important factors the affect surgery outcomes [28]. Although one study found a significant volume-outcome relationship at the physician level but not at the hospital level [29], there remain many unexplained factors to analyze before the relationship can be confirmed. One study [30] claims that a physician's skill or experience is important for determining clinical outcomes; however, other studies [31, 32] conclude that the volume of procedures is not the sole determiner of better outcomes and are concerned that physician overload may also affect the quality. If volume significantly affects outcomes, national departments of health may want to encourage the centralization of procedures in a few facilities. In addition, hospitals may have the benefit of producing better outcomes when treating only one or a few conditions [33]. Otherwise, healthcare officials may consider requiring surgeons to take additional training and education in the low-volume procedures that they perform, or enforcing the referral system for low-volume hospitals and implement some useful quality-improvement strategies for patients.
Based on all these findings, we may be able to infer some key economic implications for the feasibility and likelihood of volume-related policy options in some disease areas [34]. In addition, patients with chronic kidney disease (CKD) benefits from National Health Insurance (NHI) can apply for a catastrophic illness certificate, which grants exemption from all copayments to reduce most financial barriers for treatment under the universal healthcare system in Taiwan. For patients with CKD in Taiwan, the barriers of accessibility or finance were considerably lower than in other countries to encourage them to undergo treatment. However, even though we may find a positive relationship between outcomes and volume, it is difficult to reach consensus on a cut-point for “low volume”. Health authorities may want to consider improving the efficiency of their national referral systems to increase the quality of care and reduce mortality in their countries' hospitals [35].
This study has several limitations. First, the shortage of evidence supporting the hypothesis is that the volume-outcome association involves a causal relationship. For transplantation, an inverse volume-outcome relationship appears to exist [20, 36], but studies still need to consider several influences, such as patient and donor selection, case mix, timeliness of donor availability, operative technique, and so on. Moreover, one study [37] found comparable 90-day, 1-year, and 3-year survival outcomes between patients with end-stage renal disease (n = 14) and hepatocellular carcinoma (n = 14) who had undergone liver transplantation at a low-volume hospital. In addition, the NHI claim databases did not provide information related to kidney disease (for example, National Quality Forum measurement). Therefore, we are always to be cautious in the volume-outcome relationship.
Conclusions
Numerous studies have reported a positive association between high-volume physicians and better outcomes, but the debate for the threshold of a composite patient safety score for U.S. hospitals, which the Leapfrog Group has established [38], has still not ended; or perhaps the existing findings encourage patients to prefer facilities with better-than-expected outcomes and away from those with worse-than-expected outcomes. Moreover, despite studies [39] that have confirmed the volume-outcome relationship, more appropriate statistical tools are suggested to clarify some unsatisfactory situations. The findings also imply that training and staffing levels are important factors. Therefore, defining and exporting best practices through education outreach, and, if necessary, government regulation must be part of the national health policy agenda.
Acknowledgements
Our study was based in part on data from the Taiwan National Health Insurance Research Database provided by the Taiwan Bureau of National Health Insurance, Department of Health. This research was supported by National Science Council, Taiwan with grant number: NSC 102-2314-B-384-001 and by Chi-Mei Medical Center, Tainan, Taiwan with grant number: CMFHR10179. All authors declare that they have no potential conflicts of interest related to the research, writing, or publication of this article.
Competing interests
The authors declare that they have no competing interests.
References
1. Borowski DW, Bradburn DM, Mills SJ, Bharathan B, Wilson RG, Ratcliffe AA. et al. Volume-outcome analysis of colorectal cancer-related outcomes. The British journal of surgery. 2010;97:1416-30 doi:10.1002/bjs.7111
2. Ioka A, Tsukuma H, Ajiki W, Oshima A. Hospital procedure volume and survival of cancer patients in Osaka, Japan: a population-based study with latest cases. Japanese journal of clinical oncology. 2007;37:544-53 doi:10.1093/jjco/hym052
3. Lee CC, Ho HC, Chou P. Multivariate analyses to assess the effect of surgeon volume on survival rate in oral cancer: a nationwide population-based study in Taiwan. Oral oncology. 2010;46:271-5 doi:10.1016/j.oraloncology.2010.01.006
4. Shahin J, Harrison DA, Rowan KM. Relation between volume and outcome for patients with severe sepsis in United Kingdom: retrospective cohort study. Bmj. 2012;344:e3394. doi:10.1136/bmj.e3394
5. Sato K, Tadokoro F, Ishida K, Matsuzawa K, Nakayama Y, Yokota K. et al. Causes of death after renal transplantation: a long-term follow-up study. Transplantation proceedings. 1994;26:2017-8
6. Marcen R. Immunosuppressive drugs in kidney transplantation: impact on patient survival, and incidence of cardiovascular disease, malignancy and infection. Drugs. 2009;69:2227-43 doi:10.2165/11319260-000000000-00000
7. Reese PP, Yeh H, Thomasson AM, Shults J, Markmann JF. Transplant center volume and outcomes after liver retransplantation. American journal of transplantation: official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2009;9:309-17 doi:10.1111/j.1600-6143.2008.02488.x
8. Mouloudi E, Massa E, Georgiadou E, Iosifidis E, Kydona C, Sgourou K. et al. Course and outcome of renal transplant recipients admitted to the intensive care unit: a 20-year study. Transplantation proceedings. 2012;44:2718-20 doi:10.1016/j.transproceed.2012.09.097
9. Dharnidharka VR, Stablein DM, Harmon WE. Post-transplant infections now exceed acute rejection as cause for hospitalization: a report of the NAPRTCS. American journal of transplantation: official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2004;4:384-9
10. Prakash J, Ghosh B, Singh S, Soni A, Rathore SS. Causes of death in renal transplant recipients with functioning allograft. Indian journal of nephrology. 2012;22:264-8 doi:10.4103/0971-4065.101245
11. Bone RC, Balk RA, Cerra FB, Dellinger RP, Fein AM, Knaus WA. et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee. American College of Chest Physicians/Society of Critical Care Medicine. Chest. 1992;101:1644-55
12. American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference. definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Critical care medicine. 1992;20:864-74
13. Bateman BT, Schmidt U, Berman MF, Bittner EA. Temporal trends in the epidemiology of severe postoperative sepsis after elective surgery: a large, nationwide sample. Anesthesiology. 2010;112:917-25 doi:10.1097/ALN.0b013e3181cea3d0
14. Sands KE, Bates DW, Lanken PN, Graman PS, Hibberd PL, Kahn KL. et al. Epidemiology of sepsis syndrome in 8 academic medical centers. JAMA: the journal of the American Medical Association. 1997;278:234-40
15. Kirilov D, Cohen J, Shapiro M, Grozovski E, Singer P. The course and outcome of renal transplant recipients admitted to a general intensive care unit. Transplantation proceedings. 2003;35:606
16. Finfer S, Bellomo R, Lipman J, French C, Dobb G, Myburgh J. Adult-population incidence of severe sepsis in Australian and New Zealand intensive care units. Intensive care medicine. 2004;30:589-96 doi:10.1007/s00134-004-2157-0
17. Martin CM, Priestap F, Fisher H, Fowler RA, Heyland DK, Keenan SP. et al. A prospective, observational registry of patients with severe sepsis: the Canadian Sepsis Treatment and Response Registry. Critical care medicine. 2009;37:81-8 doi:10.1097/CCM.0b013e31819285f0
18. Fishman JA, Rubin RH. Infection in organ-transplant recipients. The New England journal of medicine. 1998;338:1741-51 doi:10.1056/NEJM199806113382407
19. Sloan FA, Shayne MW, Doyle MD. Is there a rationale for regionalizing organ transplantation services? Journal of health politics, policy and law. 1989;14:115-67
20. Tsao SY, Lee WC, Loong CC, Chen TJ, Chiu JH, Tai LC. High-surgical-volume hospitals associated with better quality and lower cost of kidney transplantation in Taiwan. Journal of the Chinese Medical Association: JCMA. 2011;74:22-7 doi:10.1016/j.jcma.2011.01.003
21. Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Critical care medicine. 2001;29:1303-10
22. Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. Journal of clinical epidemiology. 1992;45:613-9 doi:0895-4356(92)90133-8 [pii]
23. Luft HS, Hunt SS, Maerki SC. The volume-outcome relationship: practice-makes-perfect or selective-referral patterns? Health services research. 1987;22:157-82
24. Vogel TR, Dombrovskiy VY, Graham AM, Lowry SF. The impact of hospital volume on the development of infectious complications after elective abdominal aortic surgery in the Medicare population. Vascular and endovascular surgery. 2011;45:317-24 doi:10.1177/1538574411403167
25. Muilwijk J, van den Hof S, Wille JC. Associations between surgical site infection risk and hospital operation volume and surgeon operation volume among hospitals in the Dutch nosocomial infection surveillance network. Infection control and hospital epidemiology: the official journal of the Society of Hospital Epidemiologists of America. 2007;28:557-63 doi:10.1086/513613
26. Weiss-Salz I, Mandel M, Galai N, Nave I, Boner G, Mor E. et al. Factors associated with primary and secondary graft failure following cadaveric kidney transplant. Clinical transplantation. 2004;18:571-5 doi:10.1111/j.1399-0012.2004.00228.x
27. Gill P, Lowes L. The kidney transplant failure experience: a longitudinal case study. Progress in transplantation. 2009;19:114-21
28. Manilich E, Vogel JD, Kiran RP, Church JM, Seyidova-Khoshknabi D, Remzi FH. Key factors associated with postoperative complications in patients undergoing colorectal surgery. Diseases of the colon and rectum. 2013;56:64-71 doi:10.1097/DCR.0b013e31827175f6
29. Lien YC, Huang MT, Lin HC. Association between surgeon and hospital volume and in-hospital fatalities after lung cancer resections: the experience of an Asian country. The Annals of thoracic surgery. 2007;83:1837-43 doi:10.1016/j.athoracsur.2006.12.008
30. Lin HC, Lee HC. Caseload volume-outcome relation for pulmonary embolism treatment: association between physician and hospital caseload volume and 30-day mortality. Journal of thrombosis and haemostasis: JTH. 2008;6:1707-12 doi:10.1111/j.1538-7836.2008.03098.x
31. Birkmeyer JD, Stukel TA, Siewers AE, Goodney PP, Wennberg DE, Lucas FL. Surgeon volume and operative mortality in the United States. The New England journal of medicine. 2003;349:2117-27 doi:10.1056/NEJMsa035205
32. Gasper WJ, Glidden DV, Jin C, Way LW, Patti MG. Has recognition of the relationship between mortality rates and hospital volume for major cancer surgery in California made a difference?: A follow-up analysis of another decade. Annals of surgery. 2009;250:472-83 doi:10.1097/SLA.0b013e3181b47c79
33. Huesch MD. Learning by doing, scale effects, or neither? Cardiac surgeons after residency. Health services research. 2009;44:1960-82 doi:10.1111/j.1475-6773.2009.01018.x
34. Chirikos TN, French DD, Luther SL. Potential economic effects of volume-outcome relationships in the treatment of three common cancers. Cancer control: journal of the Moffitt Cancer Center. 2004;11:258-64
35. Nobilio L, Fortuna D, Vizioli M, Berti E, Guastaroba P, Taroni F. et al. Impact of regionalisation of cardiac surgery in Emilia-Romagna, Italy. Journal of epidemiology and community health. 2004;58:97-102
36. Axelrod DA, Guidinger MK, McCullough KP, Leichtman AB, Punch JD, Merion RM. Association of center volume with outcome after liver and kidney transplantation. American journal of transplantation: official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2004;4:920-7 doi:10.1111/j.1600-6143.2004.00462.x
37. Lin NC, Hsia CY, Loong CC, Liu CS, Tsai HL, Lui WY. et al. Liver transplantation at a small-volume procedure center--preliminary results from Taipei Veterans General Hospital. Journal of the Chinese Medical Association: JCMA. 2008;71:186-90 doi:10.1016/S1726-4901(08)70102-3
38. Shahian DM, Normand SL. The volume-outcome relationship: from Luft to Leapfrog. The Annals of thoracic surgery. 2003;75:1048-58
39. Livingston EH, Cao J. Procedure volume as a predictor of surgical outcomes. JAMA: the journal of the American Medical Association. 2010;304:95-7 doi:10.1001/jama.2010.905
Author contact
![]() Corresponding author: Shang-Jyh Chiou, DrPH. Department of Health Care Management, National Taipei University of Nursing and Health Sciences, 89 Nei-Chiang Street, Taipei 10845, Taiwan. Tel.: +886-2-2388-5111 ext. 6122; Email: shangjyhedu.tw.
Corresponding author: Shang-Jyh Chiou, DrPH. Department of Health Care Management, National Taipei University of Nursing and Health Sciences, 89 Nei-Chiang Street, Taipei 10845, Taiwan. Tel.: +886-2-2388-5111 ext. 6122; Email: shangjyhedu.tw.

 Global reach, higher impact
Global reach, higher impact