Impact Factor ISSN: 1449-1907
Int J Med Sci 2010; 7(5):284-289. doi:10.7150/ijms.7.284 This issue Cite
Review
Pathogenic Mechanisms Shared between Psoriasis and Cardiovascular Disease
1. Academic Dermatology and Skin Cancer Institute, East Washington Street, Chicago, Illinois, USA
2. Department of Molecular Pathology, Institute of Gerontology, Nippon Medical School, Kawasaki, Japan
3. Department of Plastic and Reconstructive Surgery, Musashi-Kosugi Hospital, Nippon Medical School, Kawasaki, Japan
Received 2010-7-2; Accepted 2010-8-17; Published 2010-8-19
Abstract
Psoriasis is associated with an increased risk of cardiovascular disease, a hallmark of which is atherosclerosis. The objective of this study was to review the pertinent literature and highlight pathogenic mechanisms shared between psoriasis and atherosclerosis in an effort to advocate early therapeutic or preventive measures. We conducted a review of the current literature available from several biomedical search databases focusing on the developmental processes common between psoriasis and atherosclerosis. Our results revealed that the pathogenic mechanisms shared between the two diseases converged onto “inflammation” phenomenon. Within the lymph nodes, antigen-presenting cells activate naive T-cells to increase expression of LFA-1 following which activated T-cells migrate to blood vessel and adhere to endothelium. Extravasation occurs mediated by LFA-1 and ICAM-1 (or CD2 and LFA-3) and activated T-cells interact with dendritic cells (and macrophages and keratinocytes in psoriasis or smooth muscle cells in atherosclerosis). These cells further secrete chemokines and cytokines that contribute to the inflammatory environment, resulting in the formation of psoriatic plaque or atherosclerotic plaque. Additionally, some studies indicated clinical improvement in psoriasis condition with treatment of associated hyperlipidemia. In conclusion, therapeutic or preventive strategies that both reduce hyperlipidemia and suppress inflammation provide potentially useful approaches in the management of both diseases.
Keywords: psoriasis, cardiovascular disease, atherosclerosis, shared pathogenic mechanism
Introduction
Psoriasis is a hereditary, chronic immune-mediated inflammatory skin disorder of unknown etiology. The disease is estimated to affect 2-3% of the general population worldwide [1]. Indeed, psoriasis has a complex genetic predisposition, but its development and/or exacerbation appear to involve an interaction between multiple genetic and environmental risk factors. Hereditary or genetic factors play a part in the development of the disease. In some patients, family members may also be affected by psoriasis. However, the exact pattern of inheritance remains to be clarified. With the advent of recent developments in understanding the role of inflammation in the pathogenesis of psoriasis, it is now widely believed that psoriasis is not just a skin disease but a systemic inflammatory process [2, 3]. On the other hand, cardiovascular disease frequently develops in individuals with persistent hyperlipidemia. Other risk factors such as hypertension, vascular endothelial cell dysfunction, oxidative stress, hyperhomocysteinemia, diabetes, smoking, high alcohol consumption, obesity, metabolic syndrome and intra-abdominal adipose visceral tissue and their adipokines, can also be involved [4-6]. These factors contribute to the formation of atherosclerosis which is a hallmark of cardiovascular disease and in which inflammation plays a major role [7, 8]. In addition, the same factors are also implicated in psoriasis patients [9, 10].Cardiovascular disease is an important cause of morbidity and mortality in patients with psoriasis. The risk factors for cardiovascular disease as well as myocardial infarction occur with higher incidence in patients with psoriasis and appear to be highest for those with more severe disease [11]. Moreover, psoriasis was suggested as an independent risk factor for cardiovascular disease [12], however some recent studies have not supported this notion [13-15], therefore this issue remains to be controversial. Here, we review the pertinent literature to highlight pathogenic mechanisms shared between psoriasis and atherosclerosis in an effort to advocate early therapeutic or preventive measures.
Review of the Literature
We conducted a comprehensive search of the current literature on psoriasis and cardiovascular disease. The search method and data retrieval was mainly the same as reported previously [16]. Briefly, the biomedical search databases of PubMed (http://www.ncbi.nml.nih.gov/sites/entrez), EMBASE (http://embase.com), SCOPUS (http://www.scopus.com/home.url) and Google Scholar (http://scholar.google.com) were searched by entering the terms 'psoriasis', 'cardiovascular disease', or 'atherosclerosis' individually or in combinations. We also carefully checked the reference list of each publication to retrieve additional citations. Data were extracted from full texts and/or abstracts. We collected data focusing on the pathogenic mechanisms of psoriasis and atherosclerosis which is a principal cause of cardiovascular disease. Further information of interest was also retrieved and included in our discretion.
Results and Discussion
Several lines of evidence indicated that psoriasis is associated with enhanced atherosclerosis and risk of cardiovascular disease, and inflammation is a pivotal link between psoriasis and atherosclerosis [17, 18]. In fact, atherosclerosis has a number of common pathogenic features with psoriasis. For example, immunological activities and pro-inflammatory cytokines play a prominent role in both diseases. In addition, both conditions share T-helper 1 (Th1) cell mediated immune compromise [19-21] and same pattern of T cell activation and expression of adhesion molecules [22-24]. It has been shown that CD4+ T cells are necessary for inducing and maintaining psoriasis. It is also envisaged that CD8+ T-cells are involved in the control of the Th1 polarization that is observed in psoriasis lesions, and that fluctuations in the severity of psoriasis, and even the spontaneous remissions that are common in guttate psoriasis, can be explained by changes in the balance between CD4+ and CD8+ effector and regulatory cell subsets [20]. Although the mechanisms underlying the association between psoriasis and cardiovascular disease still remains poorly understood, it appears that inflammation which plays a principal role in both diseases provides a common pathogenic ground between the two conditions.
Intercellular adhesion molecules (ICAMs) and vascular cell adhesion molecules (VCAM-1), as well as some of the integrins, induce firm adhesion of inflammatory cells at the vascular surface, whereas platelet endothelial cellular adhesion molecules (PECAM-1) are involved in extravasation of cells from the blood compartment into the vessel and underlying tissue. Also, inflammatory cells roll along the blood vessel wall by the interaction between selectins (E and P-selectin) expressed by endothelial cells and selectin ligands expressed by inflammatory cells [25]. Several lines of evidence support a crucial role of adhesion molecules in the development of atherosclerosis and plaque instability [24]. Expression of VCAM-1, ICAM-1 and L-selectin has been consistently observed in atherosclerotic plaques. There is accumulating evidence from prospective studies for a predictive role of elevated circulating levels of sICAM-1 in initially healthy people, and of sVCAM-1 in patients at high risk or with overt cardiovascular disease. Likewise, it has been implied that several adhesion molecules including ICAM-1 and VCAM-1 are upregulated in psoriasis [26, 27], implicating their involvement in the pathogenesis of psoriasis.
Histologically, psoriasis and atherosclerosis show common features of infiltrating T-cells, monocytes/macrophages, neutrophils, dendritic cells (DCs) and mast cells [28, 29]. The cytokine network in psoriasis and atherosclerosis is mainly characterized by Th1 type cytokines such as IFNγ, IL-2 and TNFα [20, 22, 30]. In these lesions, the major cytokine producers are dendritic cells, CD4+ and CD8+ T-cells as well as keratinocytes. IFNγ and TNFα induce keratinocytes to produce IL-6, IL-7, IL-8, IL-12, IL-15, IL 18 and TNFα in addition to several other cytokines, chemokines and growth factors. IFNγ is an important mediator of inflammation in both psoriasis and atherosclerosis and can stimulate the expression of MHC class II molecules and ICAM-1 [31, 32]. IFNγ is elevated in the serum and suction blister fluid from psoriatic patients [33, 34] and may modify the keratinocyte biology by increasing keratinocyte proliferation and causing defective cornification leading to typical psoriatic lesion [35]. Recent data also show that IFN-γ may be an essential component for growth stimulation of psoriatic keratinocyte stem cells, but it requires the presence of other growth factors as well [36]. TNFα activates and increases keratinocyte proliferation. TNFα also stimulates T-cell and macrophage cytokine and chemokine productions, and the expression of adhesion molecules on vascular endothelial cells [28-30]. IL-8 is a chemokine with main roles of neutrophil chemotaxis and stimulation of the activity of granulocytes in the inflammation process of psoriasis and atherosclerosis. In psoriasis, IL-8 from keratinocytes produces a chemotactic gradient for the migration of neutrophils into the epidermis [37] Furthermore, IL-8, IL-1 and TNF-α influence the adhesive properties of neutrophils due to an increase in the expression of surface adhesive molecules, thus improving the inter-cellular interactions with the endothelial cells, which in turn contributes to an increase in the passing of the neutrophils through the walls of the vessels. Therefore, IL-8 contributes to intensification of the reaction and to activation of the neutrophils in both conditions. IL-18 induces dendritic cells synergistically with IL-12, to increase the production of IFNγ. IL-7 and IL-15 have been reported to be important for the proliferation and homeostatic maintenance of the CD8+ T-cells [30]. IL-6 is produced by endothelial cells, DCs, and Th17 cells in lesional psoriatic skin and is encountered by trafficking T lymphocytes enabling them to escape from regulatory T cell suppression and Th17 participation in inflammation [38]. IL-6 mediates T cell activation and stimulates proliferation of keratinocytes [39], but also mediates the acute phase response. Indeed, C-reactive protein (CRP), a positive acute phase protein, is released in response to increased levels of cytokines, such as IL-6 and TNF-α, and patients with elevated levels of CRP seem to exhibit an increased risk for adverse cardiovascular outcome [40]. Furthermore, the levels of IL-6 and CRP have been reported to be raised in psoriatic patients and seem to correlate with psoriasis severity [41, 42].
Angiogenesis is a recognized feature common to psoriasis and atherosclerosis and vascular endothelial growth factor (VEGF) is a potent pro-angiogenic factor which has been reported to be upregulated in both conditions [43-45], thus may be a link between the two conditions. VEGF is also produced by human keratinocytes in response to stimulation with cytokines involved in psoriasis pathogenesis [43]. Also, pro-angiogenic cytokines such as TNFα, IL-8 and IL-17 which stimulate angiogenesis are involved in psoriasis and atherosclerosis development.
Presently, psoriasis is considered a Th1/Th17 involved inflammatory disease in which the keratinocytes are activated mainly by mediators produced by Th1 cells, but over time the mediators of Th17 cells appear to become increasingly important [46]. Likewise, Th17 cell response seems to have an important role in several cardiovascular diseases [47]. The persistent Th17 activation in psoriatic skin is characterized by infiltration of IL-23-producing DCs and Th17 cells as well as epidermal overexpression of Th17 chemokines. IL-17 cells mediate IL-12 and IL-23 which have an important role in the pathogenesis of psoriasis [48]. Also, the circulating IL-12 is thought to be the link between inflammation and Th1-type cytokine production in coronary atherosclerosis [32].
Finally, IL-17 which is produced by activated CD4+ T-cells acts synergistically to elicit further production of pro-inflammatory cytokines by the keratinocytes. In this fashion, the cytokine network in psoriasis can become a self-sustaining process. Thus, the production of pro-inflammatory cytokines together with the activation of inflammatory cells could contribute to the development of both psoriatic and atherosclerotic lesions.
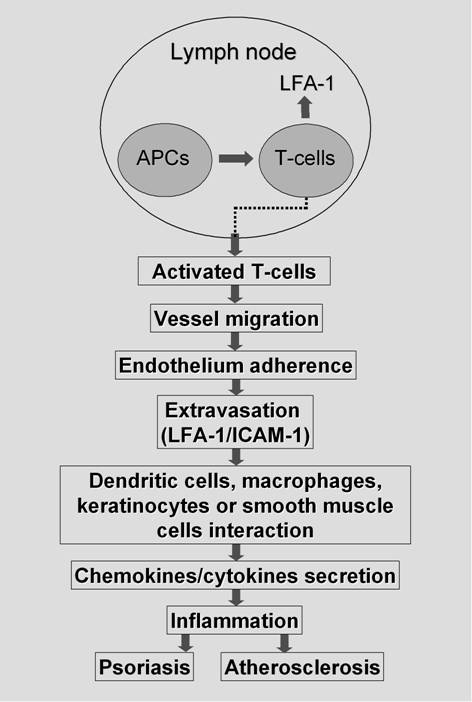
In brief, the pathogenic mechanisms shared between psoriasis and cardiovascular disease i.e. atherosclerosis may be explained stepwise as depicted in Figure 1 [49]: 1. Within the lymph node, antigen-presenting cells (APCs) activate naive T-cells to increase expression of leukocyte-function-associated antigen-1 (LFA-1); 2. Activated T-cells migrate to blood vessel; 3. Activated T-cells adhere to endothelium (plus macrophages in atherosclerosis); 4. Extravasation occurs mediated by LFA-1 and intercellular adhesion molecule-1 (ICAM-1); 5. Activated T-cell interacts with dendritic cells (plus macrophages and keratinocytes in psoriasis but smooth muscle cells in atherosclerosis); 6. Re-activated T-cells and macrophages secrete chemokines and cytokines that contribute to the inflammatory environment, resulting in the formation of psoriatic plaque or atherosclerotic plaque. In addition to the critical role of interaction between LFA-1 and its ligand, ICAM-1, the interaction of CD2 and its ligand, LFA-3 is also important in facilitation of antigen-recognition in the molecular pathways of lymphocyte adhesion [50].
Schematic representation of stepwise developmental process shared between psoriatic and atherosclerotic lesions. In the lymph node, antigen-presenting cells (APCs) activate naive T-cells to increase expression of leukocyte-function-associated antigen-1 (LFA-1). Activated T-cells migrate to blood vessel and adhere to endothelium (and macrophages in case of atherosclerosis). After extravasation mediated by LFA-1 and intercellular adhesion molecule-1 (ICAM-1) or CD2 and LFA-3, they interact with dendritic cells and macrophages and keratinocytes in psoriasis but smooth muscle cells in atherosclerosis. These re-activated T-cells and macrophages secrete chemokines and cytokines that contribute to the inflammatory environment, resulting in the formation of psoriatic plaque or atherosclerotic plaque.
It is noteworthy to elaborate on some studies that have indicated clinical improvement in psoriasis condition with treatment of associated hyperlipidemia. A pilot study evaluated the effectiveness of simvastatin which is a cholesterol lowering statin on serum lipoprotein levels and dermatitis in patients with severe psoriasis [51]. The authors found elevated high-density lipoprotein cholesterol levels and diminished PASI during the therapy. It was concluded that statins can correct lipid metabolism and reduce cutaneous lesion in psoriasis. Also, Wolkenstein P, et al. [52] reported a survey-based, case-control study of 10,000 subjects aged 15 years or more of which 356 cases were identified to have psoriasis. Of these, 71 (19.9%) received treatment for hypercholestrolemia (37 had statins and 32 other drugs). Their study confirmed the association of overweight, smoking habits and beta-blocker intake with psoriasis and reported a decreased risk of psoriasis associated with statin intake. Other drugs with potential benefits may include thiazolidindiones (TZD) family that has positive effects on both cardiovascular risk factors and psoriasis. Shafiq et al. [53] studied the effect of rosiglitazone, a commercially available TZD in psoriasis. In 70 patients with moderate to severe disease, the PASI scores improved significantly in treated vs. placebo patients with greater benefit being noted in those receiving higher doses of pioglitazone. No serious adverse effects were noted. Psoriasis cleared or almost cleared in 40% of treated patients compared to 12.5% of patients receiving placebo. It was suggested that two-thirds of patients with plaque psoriasis will improve with pioglitazone therapy.
Traditional systemic therapies for psoriasis using methotrexate and cyclosporine may reduce the risk of cardiovascular disease by decreasing inflammation however these treatments are limited by the potential for adverse effects such as hypertension, dyslipidemia, hyperhomocysteinemia, and renal and hepatic toxicity. Thus preventive measures may be required during therapy. Targeted biological therapies with efalizumab, a humanized monoclonal IgG1 antibody against CD11a, the a-subunit of leukocyte function-associated antigen 1 (LFA-1) [54, 55] and infliximab, a TNF-α blocking agents [56, 57] have provided a major advance in the treatment of the disease. Using these agents an integrated approach targeting at inflammation underlying both psoriasis and atherosclerosis may be useful in reducing cardiovascular risk in patients with psoriasis.
Conclusion
In conclusion, considering the common mechanisms underlying the development of psoriasis and atherosclerosis, it is reasonable to postulate that early therapeutic strategies targeting such shared mechanisms would have considerable effects on both conditions. To this end, pharmaceutical drugs that both reduce hyperlipidemia and suppress inflammation such as statins could provide important candidates for further clinical studies. It is intriguing to determine whether treatment of hyperlipidemia associated with psoriasis would result in clinical improvement in psoriasis or alternatively treatment of psoriasis could improve cardiovascular disease. Indeed, several studies reported that treatment of psoriasis contributes to the reduction of some risk factors of cardiovasculsr disease such as oxidative stress and inflammation, which may diminish the probability of cardiovascular events. However, an atherogenic profile, at least an atherogenic lipidic profile and a residual inflammation seems to persist after treatment of psoriasis as reported in few studies. Taken together, it is important not only to be aware of the associations between psoriasis and other cardiovascular risk factors besides hyperlipidemia, but also to be able to identify all potentially treatable conditions which seem to favor the response to therapy in psoriasis patients, contributing to a better clearing of the lesions.
Conflict of Interest
The authors declare no conflict of interest.
References
1. Langley RG, Krueger GG, Griffiths CE. Psoriasis: epidemiology, clinical features, and quality of life. Ann Rheum Dis. 2005;64(suppl 2):ii18-23
2. Lowes MA, Bowcock AM, Krueger JG. Pathogenesis and therapy of psoriasis. Nature. 2007;445:866-73
3. Krueger JG, Bowcock A. Psoriasis pathophysiology: current concepts of pathogenesis. Ann Rheum Dis. 2005;64(Suppl. 2):ii30-ii36
4. Dahlof B. Cardiovascular disease risk factors: epidemiology and risk assessment. Am J Cardiol. 2010;105(1 Suppl):3A-9A
5. Ferket BS, Colkesen EB, Visser JJ. et al. Systemic review of guidelines on cardiovascular risk assessment: which recommendations should clinicians follow for cardiovascular health check? Arch Intern Med. 2010;170:27-40
6. Zhou Q, Mrowietz U, Rostami-Yazdi M. Oxidative stress in the pathogenesis of psoriasis. Free Radic Biol Med. 2009;47:891-905
7. Ross R. Atherosclerosis - an inflammatory disease. N Engl J Med. 1999;340:115-26
8. Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685-95
9. Raychaudhuri SP, Gross J. Psoriasis risk factors: role of lifestyle practices. Cutis. 2000;66:348-52
10. Weinberg JM. Lifestyle issues and psoriasis. Cutis. 2006;78:160
11. Kaye JA, Li L, Jick SS. Incidence of risk factors for myocardial infarction and other valscular diseases in patients with psoriasis. Brit J Dermatol. 2008;159:895-902
12. Gelfand JM, Neimann AL, Shin DB. et al. Risk of myocardial infarction in patients with psoriasis. JAMA. 2006;296:1735-41
13. Wakkee M, Herings RMC, Nijsten T. Psoriasis may not be an independent risk factor for acute ischemic heart disease hospitalizations. Results of a large population-based Dutch cohort. J Invest Dermatol. 2010;130:962-7
14. Mehta NN, Azfar RS, Shin DB. et al. Patients with severe psoriasis are at increased risk of cardiovascular mortality: cohort study using the General Practice Research Database. Eur Heart J. 2010;31:1000-6
15. Stern RS. Psoriasis is not a useful independent risk factor for cardiovascular disease. J Invest Dermatol. 2010;130:917-9
16. Torchia D, Miteva M, Hu Shasa. et al. Papuloerythroderma 2009. Two new cases and systematic review of the worldwide literature 25 years after its identification by Ofuji et al. Dermatology. 2009;220:311-20
17. Alexandroff A.B, Pauriah M Camp RD. et al. More than skin deep: atherosclerosis as systemic manifestation of psoriasis. Br J Dermatol. 2009;161:1-7
18. Abou-Raya A, Abou-Raya S. Inflammation: a pivotal link between autoimmune diseases and atherosclerosis. Autoimmun Rev. 2006;5:331-7
19. Biedermann T, Röcken M, Carballido JM. TH1 and TH2 lymphocyte development and regulation of TH cell mediated immune responses of the skin. J Invest Dermatol. 2004;9:5-14
20. Gudjonsson JE, Johnston A, Sigmundsdottir H. et al. Immunopathogenic mechanisms in psoriasis. Clin Experience Immunol. 2004;135:1-8
21. Hansson GK. Immune mechanisms in atherosclerosis. Arterioscler Thromb Vasc Biol. 2001;21:1876-90
22. Hansson GK, Libby P. The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol. 2006;6:508-19
23. Schön MP, Boehncke WH. Psoriasis. N Engl J Med. 2005;352:1899-12
24. Blankenberg S, Barbaux S, Tiret L. Adhesion molecules and atherosclerosis. Atherosclerosis. 2003;170:191-203
25. Sabat R, Sterry W, Philipp S, Wolk K. Three decades of psoriasis research: where has it led us? Clinics in Dermatology. 2007;25:504-9
26. de Boer OJ, Wakelkamp IM, Pals ST. et al. Increased expression of adhesion receptors in both lesional and non-lesional psoriatic skin. Arch Dermatol Res. 1994;286:304-11
27. Cabrijan L, Batinac T, Lenkovic M. et al. The distinction between lesional and non-lesional skin in psoriasis vulgaris through expression of adhesion molecules ICAM-1 and VCAM-1. Med Hypotheses. 2009;72:327-9
28. Nickoloff BJ, Nestle FO. Recent insights into the immunopathogenesis of psoriasis provide new therapeutic opportunities. J Clin Invest. 2004;113:1664-75
29. Nickoloff BJ, Qin JZ, Nestle FO. Immunopathogenesis of psoriasis. Clin Rev Allergy Immunol. 2007;33:45-56
30. Nickoloff BJ, Xin H, Nestle FO. et al. The cytokine and chemokine network in psoriasis. Clin Dermatol. 2007;25:568-73
31. Krueger JG. The immunologic basis for the treatment of psoriasis with new biologic agents. J Am Acad Dermatol. 2002;46:1-23
32. Ranjbaran H, Sokol SI, Gallo A. et al. An inflammatory pathway of IFN-gamma production in coronary atherosclerosis. J Immunol. 2007;178:592-604
33. Gomi T, Shiohara T, Munakata T. et al. Interleukin 1 alpha, tumor necrosis factor alpha, and interferon gamma in psoriasis. Arch Dermatol. 1991;127:827-30
34. Bjerke JR, Livden JK, Degre´ M, Matre R. Interferon in suction blister fluid frompsoriatic lesions. Br J Dermatol. 1983;108:295-9
35. Barker JN. Psoriasis as a T cell-mediated autoimmune disease. Hosp Med. 1998;59:530-3
36. Bata-Csorgo ZS, Hammerberg C, Voorhees JJ. et al. Kinetics and regulation of human keratinocyte stem cell growth in shortterm primary ex vivo culture. Cooperative growth factors from psoriatic lesional T lymphocytes stimulate proliferation among psoriatic uninvolved, but not normal, stem keratinocytes. J Clin Invest. 1995;95:317-27
37. Krueger J. G. The immunologic basis for the treatment of psoriasis with new biologicagents. J Am Acad Dermatol. 2002;46:1-23
38. Goodman WA, Levine AD, Massari JV. et al. IL-6 signaling in psoriasis prevents immune suppression by regulatory T cells. J Immunol. 2009;183:3170-6
39. Grossman RM, Krueger J, Yourish D. et al. Interleukin 6 is expressed in high levels in psoriatic skin and stimulates proliferation of cultured human keratinocytes. Proc Natl Acad Sci USA. 1989;86:6367-71
40. Koenig W, Khuseyinova N, Baumert J. et al. Increased Concentrations of C-Reactive Protein and IL-6 but not IL-18 Are independently associated with incident coronary events in middle-aged men and women. Arterioscler Thromb Vasc Biol. 2006;26:2745-51
41. Gisondi P, Girolomoni G. Psoriasis and atherothrombotic diseases: Disease-specific and non-disease-specific risk factors. Semin Thromb Hemost. 2009;35:313-24
42. Coimbra S, Oliveria H, Reis F. et al. C-reactive protein and leucocyte activation in psoriasis vulgaris according to severity and therapy. J Eur Acad Dermatol Venereol. 2010;24:789-96
43. Canavese M, Altruda F, Ruzicka T. et al. Vascular endothelial growth factor (VEGF) in the pathogenesis of psoriasis--a possible target for novel therapies? J Dermatol Sci. 2010;58:171-6
44. Inoue M, Itoh H, Ueda M. et al. Vascular endothelial growth factor (VEGF) expression in human coronary atherosclerotic lesions: possible pathophysiological significance of VEGF in progression of atherosclerosis. Circulation. 1998;98:2108-16
45. Herrmann J, Lerman LO, Mukhopadhyay D. et al. Angiogenesis in atherogenesis. Arterioscler Thromb Vasc Biol. 2006;26:1948-57
46. Ghoreschi K, Weigert C, Röcken M. Immunopathogenesis and role of T cells in psoriasis. Clinics in Dermatology. 2007;25:574-80
47. Chen S, Crother TR, Arditi M. Emerging role of IL-17 in atherosclerosis. J Innate Immun. 2010;2:325-33
48. Federman DG, Shelling M, Prodanovich S. et al. Psoriasis: an opportunity to identify cardiovascular risk. Br J Dermatol. 2009;160:1-7
49. Spah F. Inflammation in atherosclerosis and psoriasis: common pathogenic mechanisms and the potential for an integrated treatment approach. Brit J Dermatol. 2008;159(suppl 2):10-17
50. Makgoba MW, Sanders ME, Shaw S. The CD2-LFA-3 and LFA-1-ICAM pathways: relevance to T-cell recognition. Immunol Today. 1989;10:417-22
51. Shirinsky IV, Shirinsky VS. Efficacy of simvastatin in plaque psoriasis: A pilot study. J Am Acad Dermatol. 2007;57:529-31
52. Wolkenstein P, Revuz J, Roujeau JC. et al. Psoriasis in France and associated risk factors: results of a case-control study based on a large community survey. Dermatology. 2009;218:103-9
53. Shafiq N, Malhotra S, Pahndhi P. et al. Pilot trial: pioglitazone versus placebo in patients with plaque psoriasis (the P6). Int J Dermatol. 2005;44:328-33
54. Werther WA, Gonzalez TN, O'Connor SJ. et al. Humanization of an anti-lymphocyte function-associated antigen (LFA)-1 monoclonal antibody and reengineering of the humanized antibody for binding to rhesus LFA-1. J Immunol. 1996;157:4986-95
55. Lowes MA, Chamian F, Abello MV. et al. Increase in TNF-alpha and inducible nitric oxide synthase-expressing dendritic cells in psoriasis and reduction with efalizumab (anti-CD11a). Proc Natl Acad Sci USA. 2005;102:19057-62
56. Reich K, Nestle FO, Papp K. et al. Infliximab induction and maintenance therapy for moderate-to-severe psoriasis: a phase III, multicentre, double-blind trial. Lancet. 2005;366:1367-74
57. Menter A, Feldman SR, Weinstein GD. et al. A randomized comparison of continuous vs. intermittent infliximab maintenance regimens over 1 year in the treatment of moderate-to-severe plaque psoriasis. J Am Acad Dermatol. 2007;56:e1-15
Author contact
![]() Corresponding author: Ramin Ghazizadeh, MD, Academic Dermatology and Skin Cancer Institute, 50 East Washington Street, Chicago, IL 60602, USA. E-mail: rghazi1com
Corresponding author: Ramin Ghazizadeh, MD, Academic Dermatology and Skin Cancer Institute, 50 East Washington Street, Chicago, IL 60602, USA. E-mail: rghazi1com

 Global reach, higher impact
Global reach, higher impact