Impact Factor ISSN: 1449-1907
Int J Med Sci 2010; 7(4):224-231. doi:10.7150/ijms.7.224 This issue Cite
Research Paper
Line bisection performance in patients with generalized anxiety disorder and treatment-resistant depression
1. Department of Psychiatry, Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou, China;
2. Department of Clinical Psychology and Psychiatry, Zhejiang University School of Medicine, Hangzhou, China;
3. Department of Psychiatry, Second Hospital, Zhejiang University School of Medicine, Hangzhou, China
Received 2010-4-6; Accepted 2010-6-29; Published 2010-7-2
Abstract
Background and Objectives The line bisection error to the left of the true center has been interpreted as a relative right hemisphere activation, which might relate to the subject's emotional state. Considering that patients with generalized anxiety disorder (GAD) or treatment-resistant depression (TRD) often have negative emotions, we hypothesized that these patients would bisect lines significantly leftward. Methods We tried the line bisection task in the right-handed healthy volunteers (n = 56), GAD (n = 47) and TRD outpatients (n = 52). Subjects also completed the Zuckerman - Kuhlman Personality Questionnaire, the Zuckerman Sensation Seeking Scales, and the Plutchik-van Praag Depression Inventory. Results GAD patients scored highest on the Neuroticism-Anxiety trait, TRD patients scored highest on depression, and both patients scored lower on the Sociability trait. Patients with GAD also bisected lines significantly leftward compared to the healthy subjects. The Frequency of the bisection error was negatively correlated with Disinhibition-Seeking in the healthy subjects, and with Total sensation-seeking and Experience-Seeking in GAD patients, while the Magnitude of the line bisection error was negatively correlated with depression in TRD patients. Conclusions The study suggests a stronger right hemispheric activation, a weaker left activation, or both in the GAD, instead of TRD patients.
Keywords: Generalized Anxiety Disorder, hemispheric activation, line bisection, treatment-resistant depression
Introduction
The functional asymmetry of the cerebral hemispheres has been reported in patients with many sorts of brain damage, who failed to orient, report, or respond to stimuli located in one hemispace 1. This phenomenon is called visuospatial neglect 2. Line bisection has been employed as a sensitive test for unilateral neglect 1,3. In this task, lateral deviation from the true center indicate the relative inattention for the contralateral side of space, and a consistent leftward error has been reported in healthy subjects in the Western world, indicating a relatively right cerebral dominance in them (reviewed in Ref 4). Nonetheless, patients with right hemispheric lesions usually place the subjective midpoint to the right of the true center 4.
Of interest to psychologists and psychiatrists are the possible functional cerebral asymmetries represented in sorts of psychiatric patients and in different emotional states of healthy individuals, which might be due to the different strategies for processing specific stimuli. In healthy subjects, negative emotions are more likely to be associated with the activation of the right hemisphere 5,6. For instance, some studies have shown that the induced anxiety in normal subjects selectively impaired their spatial, but not verbal performance 7,8, other studies in both infants and adults have found that negative affects, such as anxiousness and depression, are more relatively associated with the right hemisphere activation, particularly the frontal lobe 6,9-11. Moreover, the greater right hemisphere activation in depression patients has been demonstrated in studies that used the consonant-vowel task12, and the electroencephalogram measurements 13,14.
Individuals with generalized anxiety disorder (GAD) are found to be intolerant of uncertainty and perceive more potentially negative situations than the healthy subjects 15-17, and people with depression also employ a maladaptive problem-solving method, which contributes to the maintenance of negative emotions they perceived 18,19. One question therefore arises how patients with GAD, or with the treatment-resistant depression (TRD), a severe form of depression, would perform in the line bisection task. The possible answers might help us to understand better the hemispheric functions that contribute to these pathologies on the one hand, and probably help to further address the overlaps between anxiety and depression on the other.
Bearing that GAD and TRD patients often present negative emotions in mind, we have hypothesized that these subjects would bisect lines further leftward than the healthy volunteers would. In addition, given the high prevalence of anxiety/depression in the general population, it might be interesting to compare the levels of anxiety/depression between our patients and healthy subjects. Thus, we used the Zuckerman-Kuhlman Personality Questionnaire 20 to measure the subject's anxiety trait, and the Plutchik - van Praag Depressive Inventory 21 to measure their depressive mood. Moreover, since the sensation seeking trait is correlated with a hemispheric asymmetry 22,23, the Zuckerman Sensation Seeking Scales 24 were also used in the present protocol.
Materials and Methods
Subjects
Considering that most Chinese people are trained to use their right hands as artful ones in their early lives 25, studies of such training in athletes showed consistently moderate rightward errors in the line bisection task 26,27, we therefore selected 155 moderate to strong right-handed subjects for our study. Fifty-six healthy volunteers were recruited among college students, medical staff members or paid volunteers. After a semistructured interview, it was determined that they were not suffering from any kinds of anxiety or depressive disorder. Forty-seven outpatients were diagnosed with GAD according to the criteria of the Diagnostic and Statistical Manual of Mental Disorders - Version IV - Text Revision 28. Fifty-two outpatients were diagnosed with TRD using following criteria (all the four criteria were met): (1) symptoms met criteria for major depressive disorder 28; (2) remission failed after using at least two antidepressants; (3) patients scored more than 25 on the Plutchik - van Praag Depression Inventory; (4) patients were without comorbidities of psychotic diseases or drug abuse. In addition, patients were ascertained not to have any organic brain lesions after going through computerized tomographic or magnetic resonance imaging scans. About 50% of patients had received anxiolytics or antidepressants before arriving at our clinic, but no participants had ingested alcohol, drugs or medication at least 72 hours prior to the test. Subjects' age and gender distributions are summarized in Table 1. There were no significant group differences when referring to age (one-way ANOVA, main effect, F (2,152) = .94, P > .05), or gender (main effect, F = .06, P > .05). This study protocol was approved by a local ethics committee and all subjects gave their written informed consent.
Age (in years) and gender distribution in healthy subjects (n = 56), Generalized Anxiety Disorder (GAD, n = 47) and Treatment-Resistant Depression (TRD, n = 52) patients
| Mean age | Age range | Gender | |
|---|---|---|---|
| Healthy | 26.2 ± 8.5 | 19-54 | 24 f, 32 m |
| GAD | 28.7 ± 9.5 | 17-48 | 21 f, 26 m |
| TRD | 27.5 ± 9.9 | 16-56 | 24 f, 28 m |
Handedness was determined using a Chinese translation of the Edinburgh Handedness Inventory 29. Such an inventory has been used in two previous Chinese studies 30,31. Each of the 12 items of the inventory were scored 1, 2 or 3 according to the left-hand, either left or right, or right preference. All subjects scored between 29 and 36, and were thus considered to be moderate or strong right-handers. Their vision was either normal or corrected to normal.
Inventories
Before the line bisection task, subjects were asked to complete three questionnaires on-site in a quiet room. A brief overview of each questionnaire is described below:
- The Zuckerman - Kuhlman Personality Questionnaire. One point is given to each chosen item corresponding to personality traits. The test provides five measurements: (a) Impulsive Sensation Seeking (19 items); (b) Neuroticism-Anxiety (19 items); (c) Aggression-Hostility (17 items); (d) Activity (17 items); and (e) Sociability (17 items). The internal reliabilities of these scales range from .72 to .86. In this questionnaire, 10 items of another scale of dissimulation (infrequency or lie) were randomly inserted into the test body. Any score above 3 on the infrequency scale suggests either inattention to the content of the items and acquiescence or a very strong social desirability set; therefore, the infrequency scale was used as a test validity indicator for individuals 20. The test has proven to be reliable in the Chinese culture 32;
- The Zuckerman Sensation Seeking Scales (form V, 40 items). This inventory was slightly modified by Carton et al. 33. One point is given for each chosen item corresponding to sensation seeking. The test provides four subscales of 10 items each, ( i.e., Disinhibition, Thrill and Adventure Seeking, Experience Seeking and Boredom Susceptibility). The Total score in each subject was also calculated as the sum of the four scale scores. The internal reliabilities of these scales range from .56 to .82 24. The test has proven to be reliable in the Chinese culture 34;
- The Plutchik-van Praag Depression Inventory. This inventory contains 34 items; each item has three scale points (0, 1, 2), corresponding to depressive tendencies. Subjects have “possible depression” if they score between 20 and 25, or “depression” if they score higher than 25. The internal reliability of this inventory is .93 21.
Procedures
All subjects were requested to bisect eight lines without measuring or folding the paper. The lines, drawn in black and oriented horizontally, ranged from 102 - 144 mm in length, were arranged randomly on a sheet of A4 size paper (in a portrait orientation) one below the other, and differed in their distances from the sheet margins so that their centers were not in alignment. The response sheet was always centered on the subject's mid-sagittal plane. No restrictions were placed on head or eye movements, and no time limits were imposed. Subjects were instructed to use their right hand to make a mark indicating the center of the line.
Data analyses and statistics
There are many classical methods to analyze line bisection performance, for instance the percentage expression of bias errors 35. Here we employed a method developed by Drake & Ulrich 36. Briefly, the distance of the line bisecting task mark was measured from the actual center to the nearest millimeter. The frequency of the directional errors (Frequency), irrespective of the magnitude, was calculated as (Right - Left)/ (Right + Left); negative values indicate errors to the left and positive ones indicate errors to the right. The magnitude of line bisection deviation (Magnitude) was calculated as an algebraic sum of the distance of marks from the true center divided by the number (e.g., 8) of trials. Negative values indicate errors to the left and positive ones indicate errors to the right.
Two-way ANOVA followed by a post-hoc, Duncan's multiple new range test was applied to the five trait scores of the Zuckerman-Kuhlman Personality Questionnaire or four scale scores of sensation-seeking in the three groups. The mean Frequency, Magnitude, or depression scores in the three groups were submitted to a one-way ANOVA plus Duncan's test. The relationship between the Frequency, Magnitude, five personality traits, four sensation seeking scales, and depression scores was assessed by the Spearman rank order correlation test. With the present sample size, power to detect an effect (e.g., a scale score) was larger than 80% at P < .05 in a sample of 47 subjects per group (the smallest group in the present study).
Results
When the two-way ANOVA was applied to the five personality trait scores in the three groups, main group (F (2, 152) = 5.16, P < .05, MSE = 77.89), scale (F (4, 608) = 58.33, P < .001, MSE = 632.62), and group-scale interaction (F (8, 608) = 10.14, P < .001, MSE = 109.29) effects were detected. The post-hoc Duncan's test also detected that the GAD subjects scored significantly higher on Neuroticism-Anxiety than the healthy subjects and TRD patients did; patients also scored significantly lower on Sociability than the healthy subjects did. The four sensation-seeking scale scores, however, were not significantly different between groups (main group effect, F (2, 152) = .45, P > .05, MSE = 9.746) (Table 2).
Scale scores in the healthy subjects (n = 56), Generalized Anxiety Disorder (GAD, n = 47), and Treatment-Resistant Depression (TRD, n = 52) patients.
| Healthy | GAD | TRD | ||
|---|---|---|---|---|
| The Zuckerman-Kuhlman Personality Questionnaire | ||||
| Impulsive Sensation Seeking | 8.13 ± 3.44 | 8.38 ± 3.15 | 8.50 ± 3.70 | |
| Neuroticism-Anxiety | 8.79 ± 4.20 | 14.53 ± 2.87* | 12.10 ± 3.60*+ | |
| Aggression-Hostility | 6.71 ± 2.97 | 7.79 ± 3.61 | 7.37 ± 3.40 | |
| Activity | 8.41 ± 3.12 | 8.02 ± 2.75 | 7.25 ± 3.32 | |
| Sociability | 7.63 ± 3.40 | 6.30 ± 3.08* | 5.85 ± 3.69* | |
| Sensation Seeking Scales | ||||
| Disinhibition | 3.05 ± 1.83 | 2.49 ± 1.83 | 2.96 ± 2.01 | |
| Thrill and Adventure Seeking | 5.48 ± 2.85 | 4.89 ± 2.29 | 4.64 ± 2.57 | |
| Experience Seeking | 3.45 ± 1.80 | 3.23 ± 1.68 | 3.48 ± 1.82 | |
| Boredom Susceptibility | 2.11 ± 1.46 | 2.55 ± 1.60 | 2.39 ± 1.47 | |
| TOTAL | 14.07 ± 5.79 | 13.09 ± 4.28 | 13.46 ± 5.29 | |
| The Plutchik-van Praag Depression Inventory | ||||
| 10.79 ± 6.95 | 19.55 ± 10.28* | 32.17 ± 12.56*+ | ||
Note: * p < .05, a given patient group vs. the healthy controls; + p < .05, a given group vs. GAD group.
The mean depression scores among the three groups also had statistically significant differences from each other (main effect, F (2, 152) = 60.64, P < .001, MSE = 6205.02), with that of the TRD patients higher than those of both the healthy subjects and the GAD patients (also see Table 2).
On average, TRD patients bisected slightly more frequently to the left of the true center, whereas healthy subjects bisected slightly more frequently to the right. The difference between the healthy subjects and TRD patients was not statistically significant. In contrast, GAD patients bisected significant more frequently to the left of the true center than the healthy subjects did. When the mean Frequency errors in the three groups were analyzed, one-way ANOVA detected a significant difference (F (2, 152) = 3.50, P <.05, MSE = 1.40). The post-hoc Duncan's test showed that the GAD group (-.32 ± .56 S. D.) was significantly different from the healthy control group (.01 ± .67, P < .05). The scatter plot of the Frequency is shown in Figure 1. The mean Magnitude errors were also significantly different among the three groups (F (2, 152) = 3.31, P < .05, MSE = 5.90), post-hoc Duncan's test detected that the mean Magnitude in the GAD group (-.54 mm ± 1.15 S.D.) was significantly different from that in the healthy controls' (.12 ± 1.42), but not from that in the TRD (-.33 ± 1.40); there was no statistical difference between the mean Magnitude errors of the healthy controls and those of the TRD either.
Scatter plot of the Frequency of errors in line bisection in the healthy subjects, Generalized Anxiety Disorder (GAD) and Treatment-Resistant Depression (TRD) patients. Positive value indicates the rightward to the true center, negative one the leftward. Big arrows in each group indicate the mean Frequency. Small arrows in each group indicate the standard deviation of Frequency.
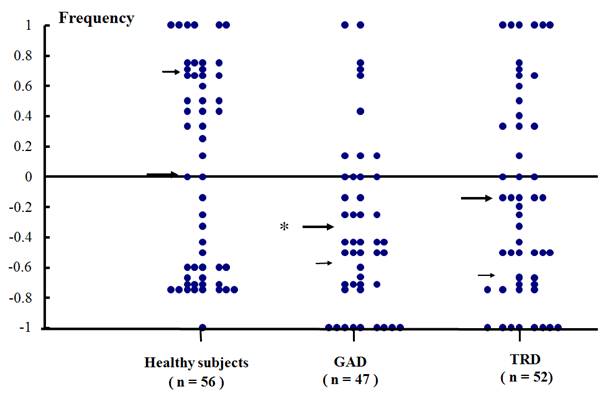
Frequency was negatively correlated with the Disinhibition-Seeking score (n = 56, r = -.28, P < .05) in healthy subjects, and with Total sensation-seeking (n = 47, r = -.30, P < .05) and Experience Seeking scores (n = 47, r = -.34, P < .05) in GAD patients. In addition, the depression score was negatively correlated with Magnitude (n = 52, r = -.30, P < .05) in TRD patients. No other correlations, such as between the handedness and Frequency/ Magnitude, or personality trait scores were found in our study.
Discussion
In the present study, patients scored higher on Neuroticism-Anxiety (with GAD patients scoring highest) and on depression (with TRD patients scoring highest) than the healthy subjects did. These results lead to the observation that there is a great overlap between anxiety and depression symptoms in clinics 37,38, and that these two disorders might share similar genetic dimensions and disease continuums 39,40. In addition, our patients scored lower on Sociability than the healthy subjects did, which was also in line with the previous report that major depression affected the Sociability trait 41, and this low extraversion (introversion) 42 tendency has also been suggested as personality endophenotypes in many anxiety disorders, e.g., social phobia and agoraphobia 43.
In compliance with our hypothesis, GAD patients erred significantly leftward in line bisection, which suggests a right hemispheric overactivation, left hypoactivation, or both for this disorder. As we have noted in our Introduction, negative emotions like anxiousness are related to the activation of the right hemisphere 8. Some neuroimaging and electrophysiological data have also shown that patients with anxiety disorders (e.g., panic disorder) displayed lower activation of the left parietal or superior temporal cortex, but relatively greater activation of the right frontal or hippocampal regions than the healthy subjects did 44-46. Contrary to our hypothesis, TRD patients did not show significant leftward line bisection errors in our study. Such a result is in line with previous studies, showing that the unipolar depressive patients displayed a non-significant leftward bias in manual line bisection, while schizophrenia patients bisected significantly leftward 47-49. However, results in regard to the hemispheric activation in the depressive disorder remain inconclusive up to date 50-55. Albeit, the slight rightward error found in our healthy subjects was different from those documented in Western countries 4, this result is similar to those in other studies conducted in Japan 56 and China 30,31. This discrepancy might result from a cultural background where most Chinese people are forced to use their right hands during their early lives 25.
The negative correlation between Frequency and Disinhibition-Seeking scores in our healthy subjects, and the negative correlation between Frequency and the Total sensation-seeking and Experience-seeking scores in GAD patients, contradict the recent neurophysiologic results in sensation seekers. For instance, a greater left frontal EEG asymmetry at rest is related to a tendency to engage in sensation-seeking and risky behaviors in young adults 23. Likewise, the association of left hemisphere predominance and risk-taking in healthy university students has also proven by studying the line bisecting performance and Zuckerman's sensation seeking scales in Drake and Ulrich's study 36. There is now no plausible explanation for the paradox. As aforementioned, a rightward bias has been reported in healthy subjects in some Eastern countries like Japan and China, contradictory to those found in Western societies, such a tendency might contribute to our current findings. Moreover, whether the reversed correlation in our GAD patients was due to the severity of anxiety itself merits further investigation. In our TRD patients, the depression score was correlated negatively with Magnitude. This finding is in accordance with the results in the tension-type headache study 31, which might be because many tension-type headache sufferers also displayed signs of depression 57. On the other hand, we could not completely ruled out the medication effects on our findings, since previous studies have shown the effect of anxiolytics or antidepressants on cognition (e.g., attention, memory or learning) 58,59, behavioral aspects (e.g., executive function or motor reaction) 60, and hemispheric asymmetry 48. However, prior to the study, our patients were all medication-free for at least 72 hours, which helped to remove some effects of the anxiolytics or antidepressants. Nevertheless, further studies about the medication effects on brain asymmetry in anxiety and depression disorders would be of interest.
Some limitations in our study should be underlined. Firstly, we did not consider the menstrual cycles of our female subjects, since the line bisection performance might be influenced by the menstrual state of women 35,61; however, three groups of our subjects were gender-balanced. Secondly, we did not measure the disordered personality traits in our subjects, since the dependent personality disorder patients have shown a pronounced leftward line bisection error 30. Thirdly, for more extensive comparisons, we would need more data from left-handed subjects, and data obtained using the neuroimaging or other indirect neuropsychological techniques. Fourthly, we used lines between 102 - 144 mm which were shorter than what other investigators used, and the line length was demonstrated to have influenced line bisection performance (e.g., Ref 62). Fifthly, we did not analyze the medication effect on the line bisection task since the individual medication strategies varied among our patients. Finally, we did not employ other attention-control paradigms such as using a cue during the task.
In conclusion, the leftward line bisection errors in GAD might indicate a stronger right, a weaker left hemispheric activation, or both. The task is a non-invasive examination and easy to manipulate in typical clinics. Whether it could be used as a diagnostic auxiliary test for anxiety versus depression remains to be determined.
Acknowledgements
The study was supported by the grants from the Natural Science Foundation of China (Nos. 30770781 & 30971042) to Dr. W Wang. The authors are very grateful to Dr. Sejla Karalic, a Fulbright Fellow to correct English expression in our manuscript. W. He, H. Chai and Y. Zhang contributed equally to the paper.
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Heilman K, Watson R, & Valenstein E. Neglect and related disorders. In: (ed.) Heilman K, Valensten E. Clinical Neuropsychology. New York: Oxford University Press. 1985:243-93
2. Stone S, Halligan P, Marshall J, & Greenwood R. Unilateral neglect: a common but heterogeneous syndrome. Neurology. 1998;50:1902-5
3. Schenkenberg T, Bradford DC, & Ajax ET. Line bisection and unilateral visual neglect in patients with neurologic impairment. Neurology. 1980;30:509-517
4. Jewell G, & McCourt M. Peseudoneglect: a review and meta-analysis of performance factors in line bisection tasks. Neuropsychologia. 2000;38:93-110
5. Smith SD, & Bulman-Fleming MB. A hemispheric asymmetry for the unconscious perception of emotion. Brain Cogn. 2004;55:452-457
6. Sato W, & Aoki S. Right hemispheric dominance in processing of unconscious negative emotion. Brain Cogn. 2006;62:261-266
7. Lavric A, Rippon G, & Gray JR. Threat-evoked anxiety disrupts spatial working memory performance: an attentional account. Cogn Ther Res. 2003;27:498-504
8. Shackman AJ, Sarinopoulos I, Maxwell JS, Pizzagalli DA, Lavric A, & Davidson RJ. Anxiety selectively disrupts visuospatial working memory. Emotion. 2006;6:40-61
9. Lee GP, Meador KJ, Loring DW, Allison JD, Brown WS, Paul LK. et al. Neural substrates of emotion as revealed by functional magnetic resonance imaging. Cogn Behav Neurol. 2004;17:9-17
10. Hellige JB. Hemispheric asymmetry. Ann Rev Psychol. 1990;41:55-80
11. Otto MW, Yeo RA, & Dougher MJ. Right hemisphere involvement in depression: Toward a neuropsychological theory of negative affective experiences. Biol Psychiatry. 1987;22:1201-1215
12. Bruder GB, Quitkin FM, Stewart JW, Marin C, Voglmaier MM, & Harrisom WM. Cerebral laterality and depression: Differences in perceptual asymmetry among diagnostic subtypes. J Abnorm Psychol. 1989;98:177-186
13. Tucker DM, Stenslie CE, Roth RS, & Shearer SL. Right frontal lobe activation and right hemisphere performance: Decrement during a depressed mood. Arch Gen Psychiatry. 1981;38:169-174
14. Knott V, Mahoney C, Kennedy S, & Evans K. EEG power, frequency, asymmetry and coherence in male depression. Psychiatry Res: Neuroim Sect. 2001;106:123-140
15. Provencher MD, Freeston MH, Dugas MJ, & Ladouceur R. Catastrophizing assessment of worry and threat schemata among worriers. Behav Cogn Psychother. 2000;28:211-224
16. Dugas MJ, Letarte H, Rhéaume J, Freeston MH, & Ladouceur R. Worry and problem solving: evidence of a specific relationship. Cogn Ther Res. 1995;19:109-120
17. Ladouceur R, Gosselin P, & Dugas MJ. Experimental manipulation of intolerance of uncertainty: a study of a theoretical model of worry. Behav Res Ther. 2000;38:933-941
18. Butzer B, & Kuiper NA. Relationships between the frequency of social comparisons and self-concept clarity, intolerance of uncertainty, anxiety, and depression. Person Indiv Diff. 2006;41:167-176
19. Dupuy JB, & Ladouceur R. Cognitive process of generalized anxiety disorder in comorbid generalized anxiety disorder and major depressive disorder. J Anx Disord. 2008;22:505-514
20. Zuckerman M, Kuhlman DM, Joireman J, Teta P, & Kraft M. A comparison of the three, the big five, and the alternative five. J Person Soc Psychol. 1993;65:747-68
21. Plutchik R, & van Praag HM. Interconvertability of five self-reported measures of depression. Psychiatry Res. 1987;22:243-56
22. Sutton SK, & Davidson RJ. Prefrontal brain asymmetry: a biological substrate of the behavioral approach and inhibition systems. Psychol Sci. 1997;8:204-210
23. Santesso DL, Segalowitz SJ, Ashbaugh AR, Antony MM, McCabe RE, & Schmidt LA. Frontal EEG asymmetry and sensation seeking in young adults. Biol Psychol. 2008;78:164-172
24. Zuckerman M. Sensation seeking: beyond the optimal level of arousal. Hillsdale, NJ: Lawrence Elbaum. 1979
25. Hoosain R. Left handedness had handedness switch amongst the Chinese. Cortex. 1990;26:451-4
26. Carlstedt RA. Line-bisection performance in highly skilled athletes: Does preponderance of rightward error reflect unique cortical organization and functioning? Brain Cogn. 2004;54:52-7
27. Dumbrava A, & Tatu MK. Visuo-spatial pseudohemineglect in professional sportsmen. Cogn Process. 2006;54:52-7
28. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (4th ed.-text revised). Washington, DC: American Psychiatric Press. 2000
29. Oldfield RC. The assessment and analysis of handedness: The Edinburgh Inventory. Neuropsychologia. 1971;9:97-133
30. Wang W, Wang Y, Gu J, Drake RA, Livesley WJ, & Jang KL. Line bisection performance in patients with personality disorders. Cogn Neuropsychiatry. 2003;8:273-85
31. Hu X, Liu Y, Liu X, Shen M, Drake RA, & Wang W. Line bisection performance in right-handed primary headache sufferers. Neurol India. 2007;55:333-337
32. Wu Y, Wang W, Du W, Li J, Jiang X, & Wang Y. Development of a Chinese version of the Zuckerman-Kuhlman Personality Questionnaire: Reliabilities and gender/age effects. Soc Behav Person. 2000;28:241-9
33. Carton S, Jouvent R, & Widlocher D. Cross-cultural validity of the sensation seeking scale: Development of a French abbreviated form. Eur Psychiatry. 1992;7:225-34
34. Wang W, Wu Y, Peng Z, Lu S, Yu L, Wang G. et al. Test of sensation seeking in a Chinese sample. Person Indiv Diff. 2000;28:169-79
35. Hausmann M, Ergun G, Yanzgan Y, & Gunturkun O. Sex differences in line bisection as a function of hand. Neuropsychologia. 2002;40:235-40
36. Drake RA, & Ulrich G. Line bisecting as a predictor of personal optimism and desirability of risky behaviors. Acta Psychol. 1992;79:219-26
37. Rouillon F. Anxiety with depression: a treatment need. European Neuropsychopharmacology. 1999;3:S87-S92
38. DeVane CL, Chiao E, Franklin M, & Kruep EJ. Anxiety Disorders in the 21st Century: Status, Challenges, Opportunities, and Comorbidity with Depression. Am J Managed Care. 2005;11:S344-S353
39. Baldwin DS, Evans DL, Hirschfeld RMA, & Kasper S. Can we distinguish anxiety from depression? Psychopharmacol Bull. 2002;36:158-165
40. Kendler KS. Major depression and generalized anxiety disorder: same genes, (partly) different environments-revisited. Focus. 2004;2:416-425
41. Wang W, Cao M, Zhu S, Gu J, Liu J, & Wang Y. Zuckerman-Kuhlman's personality Questinnaire in patients with major depression. Soc Behav Person. 2002;30:757-764
42. Bienvenu OJ, Samuels JF, Costa PT, Reti IM, Eaton WW, & Nestadt G. Anxiety and depressive disorders and the five-factor model of personality: A higher-and lower-order personality trait investigation in a community sample. Depr Anx. 2004;20:92-97
43. Bienvenu OJ, Hettema JM, Neale MC, Prescott CA, & Kendler KS. Low Extraversion and high neuroticism as indices of genetic and environmental risk for social phobia, agoraphobia, and animal phobia. Am J Psychiatry. 2007;164:1714-1721
44. Nordahl TE, Semple WE, Cross M, Mellman TA, Stein MB, Goyer P. et al. Cerebral glucose metabolic adifferences in patients with panic disorder. Neuropsychopharmacology. 1990;3:261-272
45. Wiedemann G, Pauli P, Dengler W, Lutzenberger W, Birbaumer N, & Buchkremer G. Frontal brain asymmetry as a biological substrate of emotions in patients with panic disorders. Arch Gen Psychiatry. 1999;56:78-84
46. Meyer JH, Swinson R, Kennedy SH, Houle S, & Brown GM. Increased left posterior parietal-temporal cortex activation after d-fenfluramine in women with panic disorder. Psychiatry Res: Neuroimag. 2000;98:133-143
47. Ramos-Brieva JA, Olivan J, Palomares A, & Vela A. Is there right hemisphere dysfunction in major depression? Neuroscience. 1984;23:103-110
48. Cavézian C, Danckert J, Lerond J, Daléry J, d'Amato T, & Saoud M. Visual-perceptual in healthy subjects, depressed patients, and schizophrenia patients. Brain and Cogn. 2007;64:257-264
49. McCourt ME, Shpaner M, Javitt DC, & Foxe JJ. Hemispheric asymmetry and callosal integration of visuospatial attention in schizophrenia: A tachistoscopic line bisection study. Schizophr Res. 2008;102:189-196
50. Schaffer CE, Davidson RJ, & Saron C. Frontal and parietal electroencephalogram asymmetry in depressed and non-depressed subjects. Biolog Psychiatry. 1983;18:753-762
51. Henriques J, & Davidson R. Regional brain electrical asymmetries discriminate between previously depressed and healthy control subjects. J Abnorm Psychol. 1990;99:23-31
52. Fromm D, & Schopflocher D. Neuropsychological test performance in depressed patients before and after drug therapy. Biol Psychiatry. 1984;19:55-71
53. Agarwa N, Bellani M, Perlini C, Rambaldelli G, Atzori M, Cerini R. et al. Increased fronto-temporal perfusion in bipolar disorder. J Affect Disord. 2008;110:106-114
54. Brody AL, Saxena S, Stoessel P, Gillies LA, Fairbanks LA, Alborzian S. et al. Regional brain metabolic changes in patients with major depression treated with either paroxetine or interpersonal therapy. Arch Gen Psychiatry. 2001;58:631-640
55. Martin SD, Martin E, Ra SS, Richardson MA, & Royal R. Brain blood flow changes in depressed patients treated with interpersonal psychotherapy or venlafaxine hydrochloride. Arch Gen Psychiatry. 2001;58:641-648
56. Fujii G, Fukatsu R, Yamadori A, & Kimura I. Effects of age on the line bisection test. J Clin Exp Neuropsychol. 1995;17:941-4
57. Bertolotti G, Vidotto G, Sanavio E, & Frediani F. Psychological and emotional aspects and pain. Neurol Sci. 2003;24:S71-5
58. Pache EM, Fernandez-Perez S, Sewell RD. Buspirone differentially modifies short-term memory function in a combined delayed matching/non-matching to position task. Eur J Pharmacol. 2003;477:205-211
59. Unrug A, van Luijtelaar EL, Coles MG. et al. Eventrelated potentials in a passive and active auditory condition: effects of diazepam and buspirone on slow wave positivity. Biol Psychol. 1997;46:101-111
60. Troisi JR, Critchfield TS, Griffiths RR. Buspirone and lorazepam abuse liability in humans: behavioral effects, subjective effects and choice. Behav Pharmacol. 1993;4:217-230
61. McCourt ME, Mark VW, Radonovich KJ, Willison S, & Freeman P. The effects of gender, practice and menstrual phase on the perceived location of the midsagittal plane. Neuropsychologia. 1997;35:717-724
62. Ricci A, & Chatterjee A. Context and crossover in unilateral neglect. Neuropsychologia. 2001;39:1138-1143
Authors' biography
 Dr. Wei HE got her Bachelor's degree from the West China Center of Medical Sciences, Sichuan University. She is currently a doctoral fellow in the Department of Clinical Psychology and Psychiatry, Zhejiang University School of Medicine. Her current research interests include the neurocognitive aspects of treatment-resistant depression, using brain potentials and other neuropsychological techniques. Some of her studies had been awarded by the Zhejiang Psychiatric Association.
Dr. Wei HE got her Bachelor's degree from the West China Center of Medical Sciences, Sichuan University. She is currently a doctoral fellow in the Department of Clinical Psychology and Psychiatry, Zhejiang University School of Medicine. Her current research interests include the neurocognitive aspects of treatment-resistant depression, using brain potentials and other neuropsychological techniques. Some of her studies had been awarded by the Zhejiang Psychiatric Association.
 Prof. Dr. Wei WANG, B.Med. (Anhui, China), D.Sc. (Liège, Belgium), is the PI in the Department of Clinical Psychology and Psychiatry, Zhejiang University School of Medicine. His current team work includes the clinical psychology and clinical psychiatry, being funded by the Natural Science Foundation of China, the H.J. Eysenck Memorial Foundation, and others.
Prof. Dr. Wei WANG, B.Med. (Anhui, China), D.Sc. (Liège, Belgium), is the PI in the Department of Clinical Psychology and Psychiatry, Zhejiang University School of Medicine. His current team work includes the clinical psychology and clinical psychiatry, being funded by the Natural Science Foundation of China, the H.J. Eysenck Memorial Foundation, and others.
Corresponding author: Dr. Wei WANG, B.Med., D.Sc.; Department of Clinical Psychology and Psychiatry; Zhejiang University School of Medicine; Yuhangtang Road 388; Hangzhou, Zhejiang 310058, China; Tel: +86-571-88208188. Email: wangmufancom or DrWangcom

 Global reach, higher impact
Global reach, higher impact