Impact Factor ISSN: 1449-1907
Int J Med Sci 2009; 6(5):287-295. doi:10.7150/ijms.6.287 This issue Cite
Review
Revision of late periprosthetic infections of total hip endoprostheses: pros and cons of different concepts
Department of Joint Replacement, General and Rheumatic Orthopaedics, Orthopaedic Clinic Markgröningen gGmbH, Kurt-Lindemann-Weg 10, 71706 Markgröningen, Germany
Received 2009-4-16; Accepted 2009-9-2; Published 2009-9-4
Abstract
Many concepts have been devised for the treatment of late periprosthetic infections of total hip prostheses. A two-stage revision with a temporary antibiotic-impregnated cement spacer and a cemented prosthesis appears to be the most preferred procedure although, in recent times, there seems to be a trend towards cementless implants and a shorter period of antibiotic treatment. Because of the differences in procedure, not only between studies but also within studies, it cannot be decided which period of parenteral antibiotic treatment and which spacer period is the most suitable. The fact that comparable rates of success can be achieved with different treatment regimens emphasises the importance of surgical removal of all foreign materials and the radical debridement of all infected and ischaemic tissues and the contribution of these crucial procedures to the successful treatment of late periprosthetic infections.
Keywords: periprosthetic infections, hip endoprostheses
Introduction
Periprosthetic infections occur with an incidence of less than 1% of patients but nevertheless are a serious complication of hip arthroplasties [1,2]. When early infections occur, within 4 weeks of implantation, the implant can be left in place with a high probability of cure whereas late infections require prosthesis revision to eradicate the infection [3,4]. In such cases, one can differentiate between one-stage and two-stage revisions. In the former a new prosthesis is implanted immediately after the removal of all foreign material in one operation. Two-stage revision involves an initial operation to remove all foreign materials and this is followed by an interim phase of 6 - 10 weeks, either left as a Girdlestone situation or with the implantation of a cement spacer. Individual aspects of both forms of revision have been treated very differently in the past so, in the following paragraphs, the different concepts are summarized and their respective advantages and disadvantages discussed.
One stage revision
The advantage of the one-stage revision is that only one operation is required and functional problems associated with a Girdlestone situation, such as leg shortening and instability, or, in the case of a cement spacer, spacer fracture, abraded particles from the spacer or bone resorption, can be avoided. Most surgeons have used bone cement laden with antibiotics during the re-implantation whereby the antibiotic contained in the cement or added to it is specific for the pathogen involved [5-7]. A prerequisite for this procedure is the isolation of the organism(s) from previously obtained aspirated fluid or biopsied material and the determination of their antibiotic susceptibility so that an organism-specific mixture of antibiotics can be added to the bone cement and a specific local antibiotic treatment initiated [5,6]. Here it is necessary for the fluid or tissue sample to be incubated for 14 days [6,8,9]. This long incubation period is necessary because the pathogens causing the periprosthetic infection usually occur in very small numbers in the form of a biofilm and are also often in a sessile state that is characterized by a slow rate of reproduction [8,10-13]. An analysis we carried out of 110 infected hip and knee endoprostheses showed that the culture detection rate after 7 days, the longest incubation period reported in most studies, was a mere 73.6%. To identify all infections it was necessary to cultivate for 13 days [14]. If the incubation period is of sufficient duration an accuracy of approximately 90% can be achieved with the aspiration method [15,16]. We believe that a lack of sufficient incubation led to the poor sensitivity of the pre-operative aspiration reported in other studies (for example, 46.1% reported by Hoffmann et al. [17]). The degree of success of one-stage revision of prostheses with antibiotics added to the cement led to 88% eradication reported by Steinbrink et al. [6], to 91% reported by Wroblewski et al. [7] and to 93,7% in a newer report by Rudelli et al. [18].
Mixing antibiotic into the cement affects the quality of the cement, which is why only antibiotic powder to a maximum of 10% of the total cement amount should be used [19]. Not all antibiotics can be used because they have to be available in powder form, be water-soluble and be thermostable. The most commonly used are gentamicin, clindamycin, vancomycin, tobramycin, aztreonam, ampicillin and ofloxacin [1,19-21]. There is little data available that addresses the release of antibiotics from spacers in vivo over a period of several weeks although the level of released antibiotic has been suggested by several authors to be sufficient for at least 4 months [21-23]. Furthermore, it has been found that the antibiotics affect each other's elution from the cement whereby the use of two antibiotics results in a synergistic effect and the release of the individual components is higher than that of the single antibiotics on their own [24-28]. It has also been demonstrated that the elution of antibiotic from hand-mixed cement is higher than that from cement mixed under vacuum because of the presence of air bubbles and their greater surface area. However the mechanical characteristics of hand-mixed cement are not as good [19].
Some newer studies of one-stage cementless revision of septic prostheses described the use of cancellous allografts that had been impregnated with antibiotics. Winkler et al. [29] reported 37 such cases of one-stage cementless revisions and demonstrated an eradication rate of 92% after a follow-up period of 4.4 years.
A one-stage revision can be indicated irrespective of the concept involved when a microorganism has been identified but spacer implantation is not possible because of a severely defective acetabulum and a Girdlestone situation is undesirable.
Two-stage revision
Two-stage septic revision surgery is the most common method for treating infected endoprostheses. A general advantage of the two-stage concept is that the surgical debridement is carried out twice whereby the second operation allows for the eradication of residual organisms following the initial debridement. The cement of the spacer is not intended as a means of fixing the prosthesis so the mechanical characteristics of the cement is not of primary importance at this stage. Thus, large amounts of antibiotics can be mixed into the cement before the spacer is formed. It has been possible to achieve a survival rate using two-stage revision concepts for infected hip arthroplasties of between 90% and 100% [1,30-32].
In most two-stage revisions an antibiotic-containing spacer is usually placed in position for a certain period of time before the final prosthesis is implanted [17,20,30,33,34]. The function of the spacer is on the one hand to release the antibiotic into the infected bed of the prosthesis and on the other to minimize soft-tissue contractures, retain soft tissue tension and so maintain reasonable functionality until a prosthesis can be re-implanted [30]. There are several different types of spacer: monoblock and two-part spacers, commercially available and customized spacers made in the operating theatre. The potential disadvantages of the monoblock spacers are spacer fracture and bone resorption while the two-part spacer can produce abraded cement particles [35-37]. In order to avoid spacer fractures we use a two-part spacer where the cup-shaped acetabulum spacer is formed out of antibiotic loaded cement (with a specific mixture of antibiotics recommended by the microbiologist). The spacer stem component consists of old prosthesis stem models, monoblock devices in most cases and no longer used for primary implantations, that are encased in antibiotic-supplemented cement and, just before implantation, coated in the patient's own blood in order to facilitate easier removal. The two spacer components are connected by a metal headpiece (Figure 1) [20]. However, a recent analysis of synovial membranes obtained during the operation to remove the spacer and to implant the new prosthesis revealed the presence of abraded cement debris, in particular, zirconium dioxide particles [unpublished data].
Radiograph of a hip spacer of a 63year old man with late periprosthetic infection of the left hip
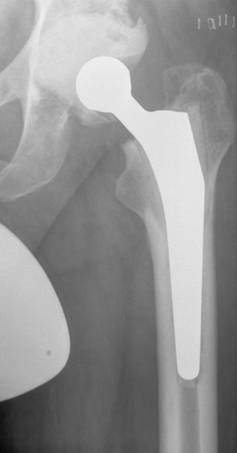
Another concept involves the use of antibiotic-laden beads although a disadvantage of this procedure is that ready-manufactured beads are usually employed and these only contain gentamicin or vancomycin [38,39]. Leg shortening and instability still occur and cause problems with mobilization. Re-implantation of a prosthesis is also often made more difficult because of scarring, tissue shrinkage and osteoporosis caused by inactivity [37,40,41]. In addition, abrasion of zirconium dioxide particles is to be expected during mobilization and this could lead to third-body-wear following re-implantation of the prosthesis. Disch et al. [35] decided therefore not to use local antibiotic carriers following removal of the prosthesis during two-stage revisions and found a reinfection rate of 6.3% in 32 hips and 41.3 months after re-implantation although there was a considerable reduction in the quality of life during the Girdlestone phase which lasted 13 months on average.
There are many questions pertaining to both one-stage and two-stage revisions that still have to be answered and existing procedures are based more on empirical findings than on data from prospective studies with a high level of evidence. It is for this reason that the following aspects of two-stage revision have been treated very differently by different groups: the type of antibiotic used in the spacer, the duration of the spacer period, the duration of systemic antibiotic treatment, aspiration before re-implantation and the type of re-implantation (cemented or cementless).
Type of antibiotic used in the spacer
Most published studies always include the same antibiotics in the cement. Some authors use vancomycin and tobramycin as local antibiotics on a regular basis because they have a broad spectrum of activity [38,42]. However, not all bacteria can be successfully treated with these agents (e.g., some gram-negative organisms), so this is an argument for investigating the antibiotic resistance pattern of the isolated bacteria and selecting a specific antibiotic for the treatment. Masri et al. [43] reported a success rate of 89.7% in their retrospective study involving bacteria-specific antibiotic mixed into the cement of a PROSTALAC® spacer (DePuy Orthopaedics, Inc, Warsaw, IN) and we saw no reinfection of 36 cases with a minimum follow-up of 2 years using this concept for handmade spacers [20].
Duration of antibiotic treatment
While most authors carry out a 6 week period of intravenous antibiotic therapy, there is a great variety of treatment regimens (Tables 1 and 2). In more recent studies, very much shorter periods of antibiotic treatment have been employed. Whittaker et al [44] reported a 92.7% eradication of infection for 41 re-implanted hip endoprostheses over a follow-up period of 4 years following a short, intravenous treatment with vancomycin alone in combination with cement spacers containing vancomycin and gentamicin. McKenna et al. [45] only found one reinfection after an average of 35 month's follow-up of 30 patients with infected hip arthroplasties who as part of the two-stage revision procedure, only received a 5 day systemic treatment with antibiotics. The design of the antibiotic administration after re-implantation of the prosthesis is even more variable and range from no antibiotic treatment at all to three months of post-surgery treatment (Tables 1 and 2).
Results of two-stage cemented revision of periprosthetic infection of the hip.
| Author | N | Follow-up | Spacer/ Beads | Local anti-biotics | Duration of intravenous antibiotics | Interval until re-implan-tation | Antbiotics after implanta-tion | Eradi-cation rate | Aseptic loosening |
|---|---|---|---|---|---|---|---|---|---|
| McDonald [46] | 82 | 5.5 years | Resection arthroplasty | No | 26.1 (4 - 59 days) | 1.5 years (6 days - 6.2 years) | No antibiotics in cement | 87 % | n.r. |
| Colyer [51] | 37 | 2.7 years | Resection arthroplasty | No | 6 weeks parenteral | 6 weeks (4 - 214 weeks) | 2 weeks parenteral, 3 months oral | 84 % | n.r. |
| Garvin [31] | 32 | ≥ 2 years, 4.1 years | Beads | Gentamicin | 6 weeks parenteral | 6 weeks | n.r. | 91 % | 0 % |
| Lieberman [32] | 32 | 40 (24-80) mo | Beads Spacer | Gentamicin Tobramycin Vancomycin | 6 weeks (20 - 49 days) | 8,8 weeks (3 weeks - 32 months) | n.r. | 91 % | n.r. |
| Younger [52] | 48 | 43 (24-63) mo | Spacer | Gentamicin | 3 weeks parenteral, 3 weeks oral | 13 weeks (5 - 42 weeks) | 3 weeks parenteral, 3 weeks oral | 94 % | 0 % |
| Leunig [37] | 12 | 2.2 years | Spacer | Gentamicin | n.r. | 4 (2-7) months | 100 % | n.r. | |
| Evans [33] | 23 | Spacer | Gentamicin | 6 weeks | 12 weeks | No | 95.7 % | n.r. | |
| Hsieh [36] | 24 | 4.2 years | Spacer | Specific: Vancomycin Piperacillin Aztreonam Teicoplanin | 2 weeks parenteral, 4 weeks oral | 11 - 17 weeks, when CRP normal | 1 week parenteral | 100 % | 0 % |
Results of two-stage cementless revision of periprosthetic infection of the hip.
| Author | N | Follow-up | Spacer/ Beads | Local anti-biotics | Duration of intravenous antibiotics | Interval until re-implan-tation | Antibiotics after implanta-tion | Eradi-cation rate | Aseptic loosening |
|---|---|---|---|---|---|---|---|---|---|
| Wilson [56] | 22/ 13** | ≥ 3 years, 48 months | Resection arthroplasty | no | 3 weeks parenteral | 6-12 weeks | 3 days parenteral | 91 % / 100 % cementless | 7.6 % stem loose |
| Nestor [58] | 34 | 47 (24-72) mo | Resection arthroplasty | no | ≥ 4 weeks parenteral | 8 (3-19) months | different | 82 % | 18% stem loose |
| Fehring [38] | 25 | 41 (24-98) mo | Beads | Tobramicin in 16 cases | 6 weeks parenteral | 4.8 months | 92 % | 0 % | |
| Haddad [39] | 50 | 5.8 (2-8.7) years | Beads + cement ball | Gentamycin | 5 days parenteral and than oral | 3 weeks | ≥ 3 months | 92 % | 8% stem subsidence |
| Koo [57] | 22 | 41 (24-78) mo | Spacer Beads | Vancomycin Gentamicin Cefotaxime | 6 weeks | 6-12 weeks | n.r. | 95 % | 5%cup loose 30% stem subsid. |
| Hofmann [17] | 27 | 76 (28-148) mo | Old stem and new polyethy-lene cup | Tobramicin | 6 weeks parenteral, in 17 cases additional oral for 6 weeks | n.r. | n.r. | 94 % | 0 % |
| Kraay [42] | 33 | ≥ 2 years | Spacer in 16 cases | Tobramicin in 16 cases | ≥ 6 weeks parenteral | 7.4 (3-37) months | n.r. | 92 % | 9 % cup 0% stem |
| Masri [43] | 29 | ≥ 2 years | Prostalac spacer | Tobramicin Vancomycin Cefuroxime Penicillin* | 6 weeks parenteral or in combina-tion with oral | 12 weeks | 5 days intra-venous | 90 % | 0 % |
| Yamamoto [60] | 17 | 38 mo | Spacer | Gentamicin Vancomycin | > 3 weeks | n.r. | 1 week parenteral, oral until CRP normal | 100 % | n.r. |
| Fink [20] | 36 | ≥ 2 years | Spacer | Specific: Gentamicin Clindamycin Vancomycin Ampicillin Ofloxacin | 2 weeks parenteral, 4 weeks oral | 6 weeks | 2 weeks parenteral, 4 weeks oral | 100 % | 6% stem subsidence 0% loose-ning |
* = combination of another local antibiotic with tobramycin, mo = months, ** = 13 of 22 re-implantations without cement; stem subsid = stem subsidence; nm = non-modular; pf = proximal fixation
The fact that there are differences in procedure not only between studies but also within studies means it cannot be decided which period of parenteral antibiotic treatment is the most suitable. That different durations of antibiotic therapy lead to similar clinical results emphasizes the fact that treatment with antibiotics is only a form of support therapy for the periprosthetic infection and that the crucial features of all concepts are the rigorous surgical removal of foreign material and the radical debridement of all infected and ischaemic tissues. These procedures are vital for the success of the revision process. However, in cases of haematogenous infection the systemic antibiotic therapy is essential for treating the focus and preventing of septic metastases.
Duration of the spacer period and antibiotic therapy
The period of time between the two operations of a two-stage revision is also very variable, ranging from a few days to several years (Tables 1 and 2). Many authors determine the time of re-implantation of a prosthesis according to clinical parameters and clinical chemistry data and carry out an aspiration of the area before surgery is carried out [32,36,43,46]. Other authors have a more or less rigid procedural plan [31,33,39]. These differences in procedure, not only between studies but also within studies, means that it cannot be decided which time period between the two steps and spacer period is the most suitable. This also appears to underscore the importance of the surgical debridement for therapeutic success of the two-stage revision.
Aspiration before re-implantation
Many authors recommend aspiration before the re-implantation operation in order to check whether or not the joint is free of infection [43,47]. The disadvantage of this concept is that the second aspiration requires a pause in the antibiotic therapy for at least 2 weeks, if not 4 weeks [48]. This is then followed by a 2-week incubation period so the second operation can be delayed by up to 4 or 6 weeks. Moreover, the local levels of antibiotic released by the spacer would likely influence the detection of viable bacteria [3]. For these reasons we do not perform an aspiration before re-implantation and rather make a decision based on clinical findings and CRP values as described by Hsieh et al. [41,49].
Cemented re-implantation
The fixation method chosen for the final prosthesis in the two-stage technique usually involves the use of cement because this allows the surgeon to add antibiotics to the cement to help prevent recurrent infection [1,31-33,50]. Rates of eradication between 84% and 100% have been described for this procedure (Table 1).
Cementless re-implantation
The disadvantage of the cemented revision technique is related to the fact that the osseous bed of the prosthesis has not only been enlarged by the loosening of the primary prosthesis but also become thinner and sclerotic. This reduces the ability of the cement to adhere to the bone. Dohmae et al. [53] reported the resistance of the bone-cement interface to shear force-related failure is reduced by 79% when comparing a cemented revision implant to a cemented primary implant. Wirtz and Niethard [54] reported a higher revision rate associated with aseptic loosening of cemented revision prostheses compared to cementless components (i.e., 15.1% versus 4.3% for the acetabular cup and 12.7% versus 5.5% for the stem). Therefore, the advantage of cementless revision may also exist for implant fixation in two-stage septic revisions although exact data concerning mid- and long-term survival rates of cemented and cementless implants in septic revision are rare in the literature [40]. Sanchez-Sotelo et al. [55] reported a 10-year infection-free survival rate of 87,5% and a mechanical survival rate of only 75,2% for re-implanted femoral components mostly fixed with cement.
Nevertheless, because the use of cementless components at the second stage does not allow the surgeon to add local antibiotics to the cement to help prevent recurrent infection, there is some concern that recurrent infection rates will be higher with cementless fixation [50,56]. A few retrospective studies have reported promising results with two-stage revision operations using cementless implants with rates of eradication between 82% and 100% (Table 2) [38,39,43,56-59].
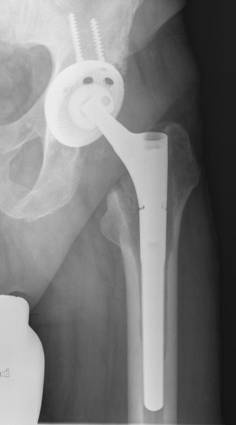
Some reports describe the stability of cementless fixation after septic revision surgery using mostly non-modular implants: Fehring et al. [38] achieved stable bone-ingrown fixation in 96% of their cases using non-modular and modular cementless prostheses with proximal fixation, while Nestor et al. [58] reported an implant stability of 79% using non-modular, proximal porous-coated stems. Wilson and Dorr [56] on the other hand, only achieved a 38% bone-ingrown fixation after 3 years in, admittedly, a small group of 13 patients using a cementless non-modular stem with proximal fixation. Moreover, the rate of early loosening of cementless revisions stems varies from 0% to 18% (Table 2). We found low rates of subsidence (6%) and loosening (0%) and a high rate of bone-ingrown fixation (94%) of a cementless modular revision stem system (Revitan curved, Zimmer GmbH, Winterthur, Switzerland), which we believe is due to the distal fixation procedures in viable bone on the one hand and to the modularity of the stems on the other hand [20] (Figure 2). Thus, as already described in an anatomic study, the in situ assembly of the components enabled the effective distal fixation of the distal prosthetic component in an adequate osseous bed before the proximal component is added and corrected for leg length and antetorsion [61].
Radiograph two years after re-implantation of a cementless modular revision stem and a press-fit-cup
Allografts
In septic revision major bone loss presents a difficult problem for reconstructive surgery. One possibility is to restore the bone defects using allografts. Many studies on allografts in septic two-stage revision do not provide enough evidence for a valid conclusion to be drawn because they include the treatment of patients with both structural and morselized allografts (e.g. in the form of an impaction graft) that are biologically very different with respect to porosity, vascularisation and incorporation. However, they have shown re-infection rates between 9% and 14% [47,62,63].
The advantages of the use of large allografts include the restoration of depleted bone stock, the correction of leg-length discrepancy and the ability to use conventional revision prostheses (and not megaprostheses). The preservation of the soft-tissue envelope including the greater trochanter and its reattachment to the allograft allows restoration of abductor function [64, 65]. The disadvantage of its use is at first the risk of infection because allografts are non-vascularised osseous segments and may represent a potential sequestrum [66,67]. However, in two-stage revisions Hsieh et al. [36] reported no recurrence of infection in 24 patients after a mean follow-up of 4.2 years and Ilyas et al. [65] in 10 patients after a mean follow-up of 5 years. Allexeeff et al. [64] also reported no recurrence of infection and only one graft failure after a mean follow-up of 47.8 months in 11 cases with two-stage revisions. They advocate structural allografts only in two-stage revisions with an interval before re-implantation of three months for Gram-positive and of six months for Gram-negative organisms or polymicrobial infections. English et al. [68] reported a success rate of 93% in the elimination of infection at a mean follow-up of 53 months in 53 patients. Buttaro et al. [69] used vancomycin-impregnated morselized allografts for impaction grafting in two-stage revision and saw a reinfection-rate of 3.3 % in 29 cases after a mean follow-up of 32.4 months.
Whereas Winkler et al. [70] used morselized allografts with local antibiotic impregnation, Rudelli et al. [18] did not impregnate with antibiotic during one-stage septic revisions and achieved success rates of 92% and 93.7% after 4.4 and 8.6 years respectively.
The relatively few in vitro and in vivo studies of the release of antibiotics from allografts indicate that it is possible to achieve high local concentrations of antibiotics with this technique, some reporting concentrations up to many times the minimal inhibitory concentration of the antibiotic concerned [70,71]. However, further study is required in order to determine the duration of antibiotic release in vivo from such allografts before a final assessment of the technique can be made.
Our own concept
We carry out two-stage revisions with cementless hip prostheses (Figures 1,2). Our technique differs from previously published techniques with cementless two-stage revision surgery in four ways (Table 2). Firstly, the antibiotic used in the antibiotic-loaded cement of the spacer and used for the systemic treatment is chosen on the basis of the sensitivity of the bacterium causing the infection. Since the use of several antibiotics seems to result in synergistic effects with regard to local release patterns, we always use at least two antibiotics in the cement and prefer COPAL® cement to Palacos® R-G cement (Heraeus Medical, Wehrheim, Germany) whenever possible because the former exhibits better release of gentamicin [27]. Secondly, we employ a short period of 2 weeks of intravenous antibiotic treatment. Thirdly, re-implantation is performed after a 6 week spacer interval and fourthly, we use modular revision stems with distal fixation in the femoral diaphysis. In a prospective study using this standardized protocol for two-stage cementless revision of periprosthetic infection of hip prostheses we were able to demonstrate 100 % eradication of infection [20]. We achieved implant stability with no early aseptic loosening, bone-ingrown fixation in 94% of the stems and absence of stem subsidence in 94%, as well as Harris hip scores of 90 points resulting in the conclusion that this concept is sufficient for treatment of periprosthetic late infections of hip prostheses [20].
The 2-week period of parenteral antibiotics we use appears short. It is, however, consistent with the recommendations of Zimmerli et al. [72,73] and Trampuz and Zimmerli [74] and has been used in other studies, e.g., Hsieh et al. [49] with 95% eradication [41,75]. Also, the total duration of antibiotic treatment of 3 months in our study was consistent with the recommendations of Zimmerli [73] and Trampuz and Zimmerli [74]. The 6-week spacer period in our study is also short but has been used by other authors (Table 1,2).
Conflict of interest
The author has no conflict of interest
References
1. Garvin KL, Hanssen AD. Current concepts review: Infection after total hip arthroplasty. J Bone Joint Surg Am. 1995;77:1576-1588
2. Fitzgerald Jr RH. Infected total hip arthroplasty: Diagnosis and treatment. J Am Acad Orthop Surg. 1995;3:249-262
3. Cui Q, Mihalko WM, Shields JS, Ries M, Saleh HJ. Antibiotic-impregnated cement spacers for the treatment of infection associated with total hip or knee arthroplasty. J Bone Joints Surg Am. 2007;89:871-882
4. Hanssen AD, Osmon DR. Evaluation of a staging system for infected hip arthroplasty. Clin Orthop Rel Res. 2002;403:16-22
5. Steinbrink K. The case of revision arthroplasty using antibiotic-loaded acrylic cement. Clin Orthop Rel Res. 1990;261:19-22
6. Steinbrink K, Frommelt L. Treatment of periprosthetic infection of the hip using one-stage exchange surgery. Orthopäde. 1995;24:335-343
7. Wroblewski BM. One-stage revision of infected cemented total hip arthroplasty. Clin Orthop Relat Res. 1986;211:103-107
8. Gollwitzer H, Diehl P, Gerdesmeyer L, Mittelmeier W. Diagnostic strategies in cases of suspected periprosthetic infection of the knee. A review of the literature and current recommendations. Orthopäde. 2006;35:904-916
9. Ince A, Rupp J, Frommelt L, Katzer A, Gille J, Löhr JF. Is „aseptic“ loosening of the prosthetic cup after total hip replacement due to non-culturable bacterial pathogens in patients with low-grade infection? Clin Inf Dis. 2004;39:1599-1603
10. Costerton JW. Biofilm theory can guide the treatment of device-related orthopaedic infections. Clin Orthop Rel Res. 2005;437:7-11
11. Gallo J, Kolar M, Novotny R, Rihakova P, Ticha V. Pathogenesis of prosthesis-related infection. Biomed Papers. 2003;147:27-35
12. Neut D, van Horn JR, van Kooten TG, van der Mei HC, Busscher HJ. Detection of biomaterial-associated infections in orthopaedic joint implants. Clin Orthop Rel Res. 2003;413:261-268
13. Peters G, Hermann M, von Eiff C. The changing pattern of coagulase-negative staphylococci as infectious pathogens. Curr Opin Infect Dis. 1995;8(Suppl):S12-S19
14. Schäfer P, Fink B, Sandow D, Margull A, Berger I, Frommelt L. Prolonged bacterial culture to identify late periprosthetic joint infection: A promising strategy. Clin Inf Dis. 2008;47:1403-1409
15. Ali F, Wilkinson JM, Cooper JR, Kerry RM, Hamer AJ, Norman P, Stockley I. Accuracy of joint aspiration for the preoperative diagnosis of infection in total hip arthroplasty. J Arthroplasty. 2006;21:221-226
16. Williams JL, Norman P, Stockley I. The value of hip aspiration versus tissue biopsy in diagnosing infection before exchange hip arthroplasty surgery. J Arthroplasty. 2004;10:582-586
17. Hofmann AA, Goldberg TD, Tanner AM, Cook TM. Ten-year experience using an articulating antibiotic cement hip spacer for the treatment of chronically infected total hip. J Arthroplasty. 2005;20:874-879
18. Rudelli S, Uiip D, Honda E, Lima ALLM. One-stage revision of infected total hip arthroplasty with bone graft. J Arthroplasty. 2008;23:1165-1177
19. Hanssen AD, Spangehl MJ. Practical applications of antibiotic-loaded bone cement for treatment of infected joint replacements. Clin Orthop Relat Res. 2004;427:79-85
20. Fink B, Grossmann A, Fuerst M, Schäfer P, Frommelt L. Two-stage cementless revision of infected hip endoprostheses. Clin Orthop Relat Res. 2009 [Epub ahead of print]
21. Hsieh PH, Chang YH, Chen SH, Ueng SW, Shih CH. High concentration and bioactivity of vancomycin and aztreonam eluted from Simplex cement spacers in two-stage revision of infected hip implants: a study of 46 patients at an average follow-up of 107 days. J Orthop Res. 2006;24:1615-1621
22. Masri BA, Duncan CP, Beauchamp CP. Long-term elution of antibiotics form bone-cement. An in vivo study using the prosthesis of antibiotic-loaded acrylic cement (PROSTALAC) system. J Arthroplasty. 1998;13:331-338
23. Bertazonni Minelli E, Benini A, Magnan B, Bartolozzi P. Release of gentamicin and vancomycin from temporary human hip spacers in two-stage revision of infected arthroplasty. J Antimicrob Chemother. 2004;53:329-334
24. Simpson PMS, Dall GF, Breusch SJ, Heisel C. In vitro elution and mechanical properties of antibiotic-loaded SmartSet HV and Palocor R acrylic bone cements. Orthopäde. 2005;34:1255-1262
25. Baleani M, Persson C, Zolezzi C, Andollina A, Borelli AM, Tigani D. Biological and biomechanical effects of Vancomycin and Meropenem in acrylic bone cement. J Arthroplasty. 2008;23:1232-1238
26. Anagnostakos K, Kelm J, Regitz T, Schmitt E, Jung W. In vitro elution of antibiotic release from and bacteria growth inhibition by antibiotic-loaded acrylic bone cement spacers. J Biomed Mater Res. 2005;72:373-378
27. Ensing GT, van Horn JR, van der Mei HC, Busscher HJ, Neut D. Copal bone cement is more effective in preventing biofilm formation than Palacos R-G. Clin Orthop Relat Res. 2008;466:1492-1498
28. Penner MJ, Masri BA, Duncan CP. Elution characteristics of Vancomycin and Tobramycin combined in acrylic bone-cement. J Arhtroplasty. 1996;11:939-944
29. Winkler H, Stoiber A, Kaudela K, Winter F, Menschik F. One stage uncemented revision of infected total hip replacement using cancellous allograft bone impregnated with antibiotics. J Bone Joint Surg. 2008;90:1580-1584
30. Burnett RSJ, Kelly MA, Hanssen AD, Barrack RL. Technique and timing of two-stage exchange for infection in TKA. Clin Orthop Relat Res. 2007;464:164-178
31. Garvin KL, Evans BG, Salvati EA, Brause BD. Palacos gentamicin for the treatment of deep periprosthetic hip infections. Clin Orthop Relat Res. 1994;298:97-105
32. Lieberman JR, Callaway GH, Salvati EA, Pellici PM, Brause BD. Treatment of the infected total hip arthroplasty with a two staged reimplantation protocol. Clin Orthop Relat Res. 1994;301:205-212
33. Evans RP. Successful treatment of total hip and knee infection with articulating antibiotic components: a modified treatment method. Clin Orthop Relat Res. 2004;427:37-46
34. Goldman RT, Scuderi GR, Insall JN. 2-stage reimplantation for infected total knee replacement. Clin Orthop Relat Res. 1996;331:118-124
35. Disch AC, Matziolis G, Perka C. Two-stage operative strategy without local antibiotic treatment for infected hip arthroplasty: clinical and radiological outcome. Arch Orthop Trauma Surg. 2007;127:691-697
36. Hsieh PH, Shih CH, Chang YH, Lee MS, Yang WE, Shih HN. Treatment of deep infection of the hip associated with massive bone loss. Two-stage revision with an antibiotic-loaded interim cement prosthesis followed by reconstruction with allograft. J Bone Joint Surg Br. 2005;87:770-775
37. Leunig M, Chosa E, Speck M, Ganz R. A cement spacer for two-stage revision of infected implants of the hip joint. Int Orthop. 1998;22:209-214
38. Fehring TK, Calton TF, Griffin WL. Cementless fixation in 2-stage reimplantation for periprosthetic sepsis. J Arthroplasty. 1999;14:175-181
39. Haddad FS, Muirhead-Allwood SK, Manktelow AR, Bacarese-Hamilton I. Two-stage uncemented revision hip arthroplasty for infection. J Bone Joint Surg Br. 2000;82:689-694
40. Mitchell PA, Masri BA, Garbuz DS, Greidanus NV, Duncan CP. Cementless revision for infection following total hip arthroplasty. Instr Course Lect. 2003;52:323-330
41. Hsieh PH, Chen LH, Chen CH, Lee MS, Yand WE, Shih CH. Two-stage revision hip arthroplasty for infection with a custom-made, antibiotic-loaded, cement prosthesis as as an interim spacer. J Trauma. 2004;56:1247-1252
42. Kraay MJ, Goldberg VM, Fitzgerald SJ, Salata MJ. Cementless two-staged total hip arthroplasty for deep periprosthetic infection. Clin Orthop Relat Res. 2005;441:243-249
43. Masri BA, Panagiotopoulos KP, Greidanus NV, Garbuz DS, Duncan CP. Cementless two-stage exchange arthroplasty for infection after total hip arthroplasty. J Arthroplasty. 2007;22:72-78
44. Whittaker JP, Warren RE, Jones RS, Gregson PA. Is prolonged systemic antibiotic treatment essential in two-stage revision hip replacement for chronic Gram-positive infection? J Bone Joint Surg. 2009;91:44-51
45. McKenna PB, O'Shea K, Masterson EL. Two-stage revision of infected hip arthroplasty using a shortened post-operative course of antibiotics. Arch Orthop Trauma Surg. 2009;129:489-494
46. McDonald DJ, Fitzgerald RA, Ilstrup DM. Two-stage revision of the total hip arthroplasty because of infection. J Bone Joint Surg. 1989;71:828-832
47. Berry DJ, Chandler HP, Reilly DT. The use of bone allografts in two-stage reconstruction after failure of hip replacements due to infection. J Bone Joint Surg. 1991;73:1460-1468
48. Mont MA, Waldman BJ, Hungerford DS. Evaluation of preoperative cultures before second-stage reimplantation of a total knee prosthesis complicated by infection: a comparison-group study. J Bone Joint Surg Am. 2000;82:1552-1557
49. Hsieh PH, Shih CH, Chang YH, Lee MD, Shih HN, Yang WE. Two-stage revision hip arthroplasty for infection: comparison between the interim use of antibiotic-loaded cement beads and a spacer prosthesis. J Bone Joint Surg Am. 2004;86:1989-1997
50. Duncan CP, Masri BA. The role of antibiotic-loaded cement in the treatment of an infection after a hip replacement. Instr Course Lect. 1995;44:305-313
51. Colyer RA, Capello WN. Surgical treatment of the infected hip implant. Two-stage reimplantation with a one-month interval. Clin Orthop Relat Res. 1994;298:75-79
52. Younger ASE, Duncan CP, Masri BA, McGraw RW. The outcome of two-stage arthroplasty using a custom-made interval spacer to treat the infected hip. J Arthroplasty. 1997;12:615-623
53. Dohmae Y, Bechthold JE, Sherman RE, Puno RM, Gustilo RB. Reduction in cement-bone interface shear strength between primary and revision arthroplasty. Clin Orthop Relat Res. 1988;236:214-220
54. Wirtz DC, Niethard FU. Etiology, diagnosis and therapy of aseptic hip prosthesis loosening—a status assessment. Z Orthop Ihre Grenzgeb. 1997;135:270-280
55. Sanchez-Sotelo J, Berry DJ, Hanssen AD, Cabanela ME. Midterm to long-term follow-up of staged reimplantation for infected hip arthroplasty. Clin Orthop Relat Res. 2009;467:219-224
56. Wilson MG, Dorr LD. Reimplantation of infected total hip arthroplasties in the absence of antibiotic cement. J Arthroplasty. 1989;4:263-269
57. Koo KH, Yang JW, Cho SH, Song HR, Park HB, Ha YC, Chang JD, Kim SY, Kim YH. Impregnation of vancomycin, gentamicin, and cefotaxime in the cement spacer for two-stage cementless reconstruction in infected total hip arthroplasty. J Arthroplasty. 2001;16:882-892
58. Nestor BJ, Hanssen AD, Ferrer-Bonzalez R, Fitzgerald RH. The use of porous prostheses in delayed reconstruction of total hip replacements that have failed because of infection. J Bone Joint Surg Am. 1994;76:349-359
59. Lai KA, Shen WJ, Yang CY, Lin RM, Lin CJ, Jou IM. Two-stage cementless revision THR after infection: 5 recurrences in 40 cases followed 2.5-7 years. Acta Orthop Scand. 1996;67:325-328
60. Yamamoto K, Miyagawa N, Masaoka T, Katori Y, Shishido T, Imakiire A. Cement spacer loaded with antibiotics for infected implants of the hip joint. J Arthroplasty. 2009;24:83-89
61. Fink B, Hahn M, Fuerst M, Thybaut L, Delling G. Principle of fixation of the cementless modular revision stem Revitan. Unfallchirurg. 2005;108:1029-1037
62. Ammon P, Stockley I. Allograft bone in two-stage revision of the hip for infection. Is it safe? J Bone Joint Surg. 2004;86:962-965
63. Wang JW, Chen CE. Reimplantation of infected hip arthroplasties using bone allografts. Clin Orthop Relat Res. 1997;335:202-210
64. Alexeeff M, Mahomed N, Morsi E, Garbuz D, Gross A. Structural allograft in two-stage revisions for failed septic hip arthroplasty. J Bone Joint Surg. 1996;78:213-216
65. Ilyas I, Morgan DAF. Massive structural allograft in revision of septic hip arthroplasty. Int Orthop. 2001;24:319-322
66. Salvati EA, Chekofsky KM, Braus BD, Wilson PD Jr. Reimplantation in infection: a 12-year experience. Clin Orthop Relat Res. 1982;170:62-75
67. Tornford WW, Thongphasuk J, Mankin HJ, Ferrero MJ. Frozen musculoskeletal allografts: a study of the clinical incidence and causes of infection associated with their use. J Bone Joint Surg. 1990;72:1137-1143
68. English H, Timperley AJ, Dunlop D, Gie G. Impaction grafting of the femur in two-stage revision for infected total hip replacement. J Bone Joint Surg. 2002;84:700-705
69. Buttaro MA, Pusso R, Piccaluga F. Vancomycin-supplemented impacted bone allografts in infected hip arthroplasty. Two-stage revision results. J Bone Joint Surg. 2005;87:314-319
70. Winkler H, Janata O, Berger C, Wein W, Georogopoulos A. In vitro release of vancomycin and tobramycin form impregnated human and bovine bone grafts. J Antimicrob Chemother. 2000;46:423-428
71. Witso E, Persen L, Loseth K, Benum P, Bergh K. Cancellous bone as an antibiotic carrier. Acta Orthop Scand. 2000;71:80-84
72. Zimmerli W. The role of antimicrobial agents in the management of infected arthroplasties. Orthopäde. 1995;24:308-313
73. Zimmerli W. Infection and musculoskeletal conditions: prosthetic-joint-associated infections. Best Pract Res Clin Rheumatol. 2006;20:1045-1063
74. Trampuz A, Zimmerli W. New strategies for the treatment of infectious associated with prosthetic joints. Curr Opin Investig Drugs. 2005;6:185-190
75. Zimmerli W, Widmer AF, Blatter M, Frei R, Ochsner PE. Role of rifampicin for treatment of orthopedic implant-related staphylococcal infections: a randomized controlled trial. Foreign-Body Infection (FBI) Study Group. JAMA. 1998;279:1537-1541
Author biography
Bernd Fink, Prof. Dr. med., is head of the Department of Joint Replacement, General and Rheumatic Orthopaedics of the Orthopaedic Clinic Markgroeningen in Germany. He is specialized in joint replacement with minimal-invasive techniques and foot surgery. His special interest lies in the revision surgery of endoprostheses and he has a big experience in treating infected endoprostheses. Several high level papers have been published in high ranked journals concerning these topics.
Correspondence to: Prof. Dr. med. Bernd Fink, M.D., Department of Joint Replacement, General and Rheumatic Orthopaedics, Orthopaedic Clinic Markgröningen gGmbH, Kurt-Lindemann-Weg 10, 71706 Markgröningen gGmbH, Kurt-Lindemann-Weg 10, 71706 Markgröningen, Germany, Phone: ++49-7145-912201, Fax: ++49-7145-912922, E-mail: b.finkde

 Global reach, higher impact
Global reach, higher impact