3.2
Impact Factor
ISSN: 1449-1907
Int J Med Sci 2008; 5(6):333-340. doi:10.7150/ijms.5.333 This issue Cite
Research Paper
Different effect of exercise on left ventricular diastolic time and interventricular dyssynchrony in heart failure patients with and without left bundle branch block
Department of Cardiology and Angiology, Marienhospital Herne, University of Bochum, Germany
Received 2008-10-1; Accepted 2008-11-3; Published 2008-11-4
Abstract
Background: In patients with idiopathic dilated cardiomyopathy (IDCM) a prolongation of left ventricular (LV) systole at the expense of diastolic time was demonstrated. Our study was aimed to evaluate the effect of exercise on heart rate corrected diastolic time in controls, IDCM with and without LBBB, and patients with LBBB and normal LV function.
Methods: 47 patients with IDCM, 30 without LBBB, and 17 with LBBB as well as 11 with isolated LBBB were studied during exercise using a combined hemodynamic-radionuclide angiographic approach. The phases of the cardiac cycle were derived with high temporal resolution from the ventricular time-activity curve. The loss of diastolic time per beat (LDT) was quantified using a regression equation obtained from a control group (n=24).
Results: A significant LDT was demonstrated at rest and during peak exercise in IDCM patients with LBBB (39.1±32 and 37.3±30 ms; p < 0.001). In IDCM patients with normal activation LDT was unaffected at baseline, but elevated during peak exercise. This response was paralleled by an increase in interventricular mechanical dyssynchrony.
Conclusion: During exercise an abnormal shortening of LV diastolic time is a common characteristic of heart failure patients which can be explained by the high prevalence of mechanical dyssynchrony.
Keywords: diastolic time, heart failure, left bundle branch block, exercise test, interventricular dyssynchrony
Introduction
The introduction of cardiac resynchronization therapy has recently renewed the clinical interest in the analysis of specific phases of the cardiac cycle and their temporal relationship (1). In heart failure patients two major abnormalities of the gross time course of cardiac contraction were demonstrated: a prolongation of total isovolumic time (t-IVT) which represents the time wasted within the cardiac cycle and a disproportionate shortening of left ventricular (LV) diastolic time which suggests an impaired temporal relation between LV systole and diastole (2,3,4). Whether left bundle branch block (LBBB) has a similar, consistent effect on the systolic-diastolic phase proportion at rest and during stress has not been thoroughly investigated. Therefore, our study sought to separate the effects of LBBB and ventricular disease on the relative duration of LV systole and diastole in heart failure patients during peak incremental exercise.
Methods
Patients
All NYHA class III patients undergoing invasive hemodynamic exercise testing for clinical reasons were consecutively screened as potential candidates. Inclusion criteria were: idiopathic dilated cardiomyopathy (IDCM) diagnosed on the basis of the exclusion of other causes of LV dysfunction, such as of evidence of myocarditis in endomyocardial biopsy, significant coronary artery stenoses revealed by angiography, valvular heart disease except of functional mitral regurgitation. Only patients with an echocardiographic ejection fraction (EF) < 40% were included. Patients with atrial fibrillation, QRS prolongation with non-LBBB pattern and disorders other than cardiac disease that limit exercise performance were excluded.
According to QRS duration the selected patients (n =47) were divided into two groups: 30 patients with normal QRS duration (< 120 ms; IDCM-na) and 17 patients with prolonged QRS duration (≥ 120 ms) and LBBB activation pattern (IDCM-LBBB).
Additional 11 patients with isolated LBBB were retrospectively selected from a larger cohort of patients evaluated in our hemodynamic laboratory. All of these patients had a normal left ventricular cavity size and baseline ejection fraction as evaluated by radionuclide angiography. Coronary artery disease was excluded by angiography. All of these patients had a limited exercise tolerance classified as NYHA II.
The control group consisted of 24 patients referred for ventricular function assessment with radionuclide angiography before cardiotoxic cancer treatment. None of these patients had a history of cardiac disease, diabetes or hypertension.
All patients included in the overall study gave their written informed consent prior to the examination.
Exercise hemodynamics
The patients performed supine bicycle exercise beginning at a load of 25 watt with increases of 25 watt every 5 min until the development of fatigue or limiting dyspnoea. Rest and exercise hemodynamics were measured with a pulmonary artery balloon flotation right heart catheter. Resting measurements were obtained twice, once 15 min before and again immediately before the exercise. Peak and mean pulmonary arterial pressure (MPA) as well as mean pulmonary arterial wedge (PCWP) pressure were recorded. Right atrial pressure was measured at baseline and immediately after cessation of exercise. The brachial arterial systolic and diastolic pressures were measured by sphygmomanometer. Forward cardiac output was measured by the thermodilution technique both at rest and during exercise immediately after the pressure measurements. Between three and five thermodilution cardiac output measurements were carried out at each exercise stage for calculation of the mean value. Cardiac index (CI), stroke volume index (SVI) and systemic vascular resistance (SVR) were calculated according to standard formulae. LV end-diastolic volumes (EDVI) was calculated by dividing thermodilution stroke volume index by the respective ejection fraction.
Echocardiography
Two-dimensional echocardiography was performed in all patients using an ultrasonic device equipped with a 2.5-MHz transducer. The left ventricular diastolic and systolic dimensions were measured in the parasternal long-axis view according to the standard recommendations (5). The left ventricular ejection fraction was calculated according to the formula established by Quinones et al. (6)
Radionuclide angiography
Simultaneously with hemodynamic measurements, equilibrium radionuclide angiography was performed using a high sensitivity, low-energy parallel hole collimator interfaced to a computer system (Sopha-DS7-gammacamera) as previously described (7). Acquisition was carried out at LAO 20°-30° angulation and 15° caudal tilting to achieve a precise separation of the interventricular septum. Data were registered in a 64 x 64 matrix at 32 frames per cardiac cycle at rest and during exercise and time-activity curves were constructed. As usual, the R-wave of the electrocardiograph was used to identify the onset of each new cardiac cycle. A dynamic filtration acquisition mode was used, with rejection of cardiac cycles beyond ±10% of the mean R-R interval. The data were further processed and analyzed by a semi-automated computer routine (8). Right- and left-ventricular regions of interest were traced simultaneously on diastolic images and functional images obtained by Fourier amplitude and phase analysis to provide a clear definition of the septum and the atrio-ventricular junction. Left ventricular ejection fraction (LVEF) was calculated as (end-diastolic counts-end-systolic counts)/end-diastolic counts.
Duration of left ventricular systole and diastole (method of calculation)
The absolute duration of left ventricular systole was defined as the time interval between the onset of the R-wave and the minimal volume on the time-activity curve (end-systole). The relative duration of left ventricular systole was derived as the product of absolute systolic time and heart rate and expressed in seconds per minute. Diastolic time was calculated as the cardiac length (RR interval) minus the duration of systole. To define diastolic duration during exercise more precisely, diastolic time in IDCM patients was compared with its predicted value obtained from putting each heart rate of IDCM patients into a regression equation obtained from the control group. The loss of diastolic time per beat (LDT) was defined as the difference between and calculated and individually observed diastolic time values.
Interventricular mechanical dyssynchrony (method of calculation)
Phase image analysis was applied to the scintigraphic data using the above mentioned computer routine. The phase program assigns a phase angle to each pixel of the phase image, derived from the first Fourier harmonic of time. The phase angle corresponds to the relative sequence and pattern of ventricular contraction during the cardiac cycle. The mean phase angles were computed for right ventricular (RV) and LV blood pools as the arithmetic mean phase angle for all pixels in the ventricular region of interest. Interventricular mechanical dyssynchrony was evaluated as the difference between LV and RV mean phase angles (RV-LV-delay). Phase data were expressed in degrees between 0° and 360°.
Statistical analysis
All data are given in terms of the mean ± SD. Differences in group means were analyzed with the two tailed unpaired t-test. For comparison of the individually observed and predicted time values and for comparison of within-group changes a repeated measures analysis of variance was used. If analysis of variance showed an overall difference, pairwise comparison was performed with a paired t-test. Correlation was performed by linear regression analysis. Analyses were performed using the software package “SPSS for Windows 12.0.1“.
Results
Systolic and diastolic time intervals
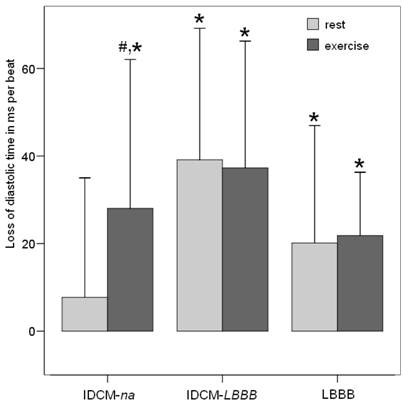
The clinical characteristics and time interval details of the patient groups and the control group are presented in Table 1. Medical treatment was similar in both patient groups with IDCM. In those with normal activation, 27 of 30 were receiving a beta-blocking agent, 30 an angiotensinconverting enzyme inhibitor or angiotensin receptor blocker, and 19 a diuretic. In patients with conduction disturbance, 13 of 17 patients were receiving a beta-blocking agent, 15 an angiotensinconverting enzyme inhibitor or angiotensin receptor blocker, and 10 a diuretic. 8 of 11 patients with isolated LBBB had beta-blocker therapy. All groups had similar baseline heart rates. During peak exercise heart rates were slightly lower in all three patient groups. To better separate the effect of heart rate from cardiac function time intervals diastolic time was plotted against the corresponding heart rate of each exercise stage and regression analysis was performed. An inverse, nonlinear relation between heart rate and LV diastolic time was found in either normal subjects and both patient groups with IDCM. The equations were: diastolic time = 101905xHR-1.23; r = 0.97, p < 0.001 for normal subjects, 187663xHR-1.38; r = 0.92 for IDCM-na patients; p < 0.001 and 174925xHR-1.38; r = 0.90 for IDCM-LBBB patients; p < 0.001. The regression equation obtained from the control group was used to quantify the loss of diastolic time per beat in all three patient groups. When predicted and observed values of diastolic time were compared a significant LDT was demonstrated at rest (20.1±29 ms; p = 0.003) and during peak exercise (21.8±16 ms; p < 0.001) in patients with isolated LBBB. In IDCM-LBBB patients the alteration was even more pronounced (39.1±32 ms at rest and 37.3±30 ms at peak exercise; p < 0.001). In IDCM-na patients LDT was not significant at baseline (5.4±31 ms; ns), however during exercise a significant LDT was observed (28.1 ± 36 ms; p = 0.03). Within this subgroup of patients a significant exercise-related increase in LDT was demonstrated (5.4 ± 31 ms vs. 28.1 ± 36 ms; p = 0.02) (Figure 1).
SVR at peak exercise did not correlate with LDT at peak exercise, nor did its exercise related change correlate with the corresponding change in SVR. Although a significant left ventricular chamber dilatation from rest to exercise was evident in IDCM-na and IDCM-LBBB patients (188 ± 57 vs. 211 ± 92 ml/m2; p = 0.04 and 193 ± 39 vs. 225± 88 ml/m2; p = 0.02), no correlations between the absolute values of EDVI at rest or during exercise or the exercise-related increase in EDVI and the corresponding LDT values were found in both IDCM subgroups.
Loss of diastolic time per beat at rest and during peak exercise in the different subgroups of patients. *p < 0.05 significant LDT when observed and predicted time values were compared. #p < 0.05 increase in LDT from rest to peak exercise within a study group.
Clinical characteristics, hemodynamic data and the time interval details of the entire patient group and the control group.
| Controls n = 24 | IDCM-na n = 30 | IDCM-LBBB n = 17 | LBBB n = 11 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age | 49 | ± | 12 | 52 | ± | 15 | 54 | ± | 12 | 41 | ± | 16* | |
| Male/ female ratio | 20 | : | 4 | 24 | : | 6 | 14 | : | 3 | 9 | : | 2 | |
| Hypertension/ Diabetes mellitus (n) | 0 | : | 0 | 7 | : | 2 | 3 | : | 2 | 3 | : | 0 | |
| Baseline LV end-diastolic diameter (cm) | - | ± | - | 7.0 | ± | 0.8 | 7.1 | ± | 1.0 | 5.3 | ± | 1.1† | |
| Duration of exercise (min) | 19 | ± | 6 | 12 | ± | 4* | 11 | ± | 5* | 16 | ± | 6*,† | |
| Ejection fraction (%) | |||||||||||||
| rest | 60 | ± | 7 | 23 | ± | 7* | 20 | ± | 7* | 61 | ± | 10† | |
| peak exercise | 71 | ± | 10 | 24 | ± | 8* | 18 | ± | 10* | 66 | ± | 11† | |
| Heart rate (beats/min) | |||||||||||||
| rest | 73 | ± | 14 | 73 | ± | 11 | 80 | ± | 9 | 67 | ± | 6 | |
| peak exercise | 127 | ± | 23 | 114 | ± | 17* | 122 | ± | 17 | 117 | ± | 17* | |
| Systolic blood pressure (mmHg) | |||||||||||||
| rest | 120 | ± | 15 | 123 | ± | 16 | 122 | ± | 20 | 129 | ± | 24 | |
| peak exercise | 168 | ± | 20 | 155 | ± | 24 | 159 | ± | 26 | 173 | ± | 26 | |
| Diastolic blood pressure (mmHg) | |||||||||||||
| rest | 83 | ± | 13 | 81 | ± | 9 | 84 | ± | 12 | 88 | ± | 14 | |
| peak exercise | 87 | ± | 8 | 85 | ± | 7 | 89 | ± | 10 | 93 | ± | 14 | |
| Duration of systole (ms) | |||||||||||||
| rest | 299 | ± | 45 | 308 | ± | 48 | 325 | ± | 45 | 337 | ± | 32*,† | |
| peak exercise | 217 | ± | 42 | 258 | ± | 55* | 254 | ± | 42* | 245 | ± | 18* | |
| Relative duration of systole (s/min) | |||||||||||||
| rest | 21.5 | ± | 3.2 | 22.4 | ± | 3.6# | 25.9 | ± | 3.4* | 22.6 | ± | 2.1 | |
| peak exercise | 26.7 | ± | 3.2 | 29.4 | ± | 4.1 | 30.9 | ± | 3.4* | 28.6 | ± | 2.9 | |
| Relative duration of diastole (s/min) | |||||||||||||
| rest | 38.5 | ± | 3.2 | 37.6 | ± | 3.6# | 34.1 | ± | 3.4* | 37.4 | ± | 2.1 | |
| peak exercise | 33.3 | ± | 3.2 | 30.6 | ± | 4.1 | 29.1 | ± | 3.4* | 31.4 | ± | 2.9 | |
| RV-LV delay (°) | |||||||||||||
| rest | -0.4 | ± | 6.8 | 1.5 | ± | 6.4# | 9.3 | ± | 7.3* | 8.6 | ± | 6.7*,† | |
| peak exercise | -1.3 | ± | 6.1 | 5.1 | ± | 7.7* | 10.4 | ± | 7.5* | 7.9 | ± | 9.5* | |
| Loss of diastolic time per beat (ms) | |||||||||||||
| rest | 5.4 | ± | 31# | 39.1 | ± | 32* | 20.1 | ± | 29*,† | ||||
| peak exercise | 28.1 | ± | 36* | 37.3 | ± | 30* | 21.8 | ± | 16* | ||||
| Cardiac index (l/min/m2) | |||||||||||||
| rest | - | - | 2.9 | ± | 0.9 | 2.9 | ± | 0.6 | 3.3 | ± | 0.5 | ||
| peak exercise | - | - | 5.0 | ± | 1.1 | 4.8 | ± | 2.0 | 7.1 | ± | 1.4† | ||
| Stroke volume index (ml/m2) | |||||||||||||
| rest | - | - | 41.0 | ± | 15 | 38.0 | ± | 8 | 51.6 | ± | 9† | ||
| peak exercise | - | - | 44.1 | ± | 11 | 40.5 | ± | 12 | 63.2 | ± | 12† | ||
| End-diastolic volume index (ml/m2) | |||||||||||||
| rest | - | - | 188 | ± | 57 | 193 | ± | 39 | 85 | ± | 27† | ||
| peak exercise | - | - | 211 | ± | 92 | 225 | ± | 88 | 97 | ± | 33† | ||
| Systemic vascular resistance (dyn*s*cm-5) | |||||||||||||
| rest | 1417 | ± | 311 | 1455 | ± | 281 | 1367 | ± | 310 | ||||
| peak exercise | - | - | 903 | ± | 212 | 951 | ± | 195 | 717 | ± | 223† | ||
| Mean pulmonary artery pressure (mmHg) | |||||||||||||
| rest | - | - | 21.9 | ± | 9 | 18.8 | ± | 7 | 12.8 | ± | 2† | ||
| peak exercise | - | - | 42.5 | ± | 11 | 38.1 | ± | 11 | 33.5 | ± | 9† | ||
IDCM-na = patients with idiopathic dilated cardiomyopathy and normal activation pattern; IDCM-LBBB = IDCM patients with left bundle branch block; LBBB = patients with isolated left bundle branch block
* p < 0.05 compared with controls
# p < 0.05 IDCM-na vs. IDCM-LBBB
† p < 0.05 LBBB vs. IDCM-na
Interventricular mechanical dyssynchrony
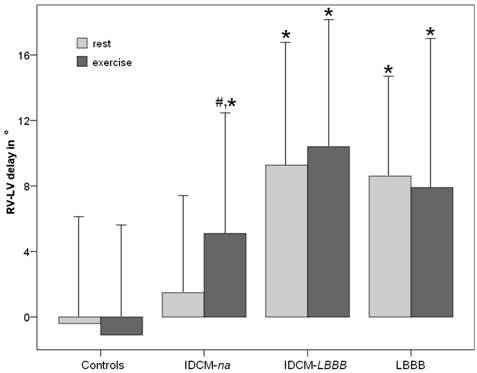
At baseline a significant RV-LV delay was demonstrable in LBBB patients, but not in IDCM patients with normal QRS duration. During peak exercise, however, all three subgroup of patients showed a significant RV-LV delay compared to control subjects (Table 1, Figure 2). Within IDCM-na patients a significant increase of RV-LV delay from rest to peak exercise was observed (1.5 ± 6.4 vs. 5.1 ± 7.7°; p < 0.001).
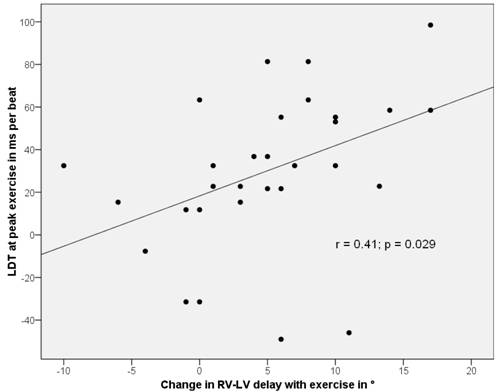
In patients with LBBB no significant correlation between the rest or exercise values of RV-LV delay and the corresponding LDT was found. In addition, there was no significant correlation between the exercise-related change of RV-LV-delay and LDT within these subgroups. In IDCM-na, however, there was a weak correlation between the exercise related change of both parameters (r = 0.34; p = 0.04). Furthermore, a moderate correlation between the exercise-related change in RV-LV delay and LDT at peak exercise was demonstrable (Figure 3).
Interventricular mechanical dyssynchrony (RV-LV delay) at rest and during peak exercise in the different subgroups of patients. *p < 0.05 significant RV-LV delay compared to controls. #p < 0.05 significant increase in RV-LV delay from rest to peak exercise within a study group.
Correlation between the loss of diastolic time at peak exercise in IDCM-na patients and the exercise-related increase in interventricular mechanical dyssynchrony.
Discussion
Our study demonstrated a significant prolongation of LV systole and a consecutively shortened LV diastole in patients with LBBB irrespective of the presence of left ventricular systolic dysfunction. The absolute loss of diastolic time was consistent in these groups whether measurements were taken at rest or during exercise. In contrast, heart failure patients without LBBB exhibited a more dynamic response pattern. At baseline LV diastolic time was within physiological limits. During exercise, however, a significant shortening of LV diastolic time was observed.
These findings strongly suggest that conduction mediated and conduction independent factors did not additively contribute to the derangement of the systolic-diastolic phase proportion during exercise in heart failure patients.
Principally, alterations of the two principal components of the entire systolic phase may have contributed to its prolongation. LV ejection time was generally found to be unchanged or rather reduced in severe systolic heart failure (2,9). However, a few exceptions were noted. During exercise a progressive left ventricular dilatation and a reduced systemic vasodilator capacity may produce sub-optimal afterload conditions resulting in a prolongation of left ventricular ejection and total LV systole (10,11). This effect of loading conditions on the duration of LV systole is a well documented finding in cardiac disease states with considerable afterload-mismatch as aortic stenosis and hypertrophic obstructive cardiomyopathy (12,13). The end-diastolic volume increase during exercise, may bring the failing ventricle onto the descending limb of its function curve. In this situation a prolongation of left ventricular ejection was demonstrated which may be viewed as a compensatory mechanism that serves to maintain left ventricular pump performance (14,15). However, systemic vascular resistance decrease during exercise and the degree of left ventricular enlargement was similar in IDCM patients with and without LBBB. Hence, these mechanisms may equally be operative in both subgroups and may not have contributed to the observed differences in temporal characteristics induced by exercise.
On the other hand, there is consistent evidence that isovolumic contraction time as the other major component of LV systolic duration is increased in patients with dilated cardiomyopathy. A disturbed electromechanical activation leading to intraventricular dyssynchrony is typically present in patients with LBBB but also common in patients without LBBB (16). As a result of the heterogeneous onset of LV contraction and its heterogeneous termination LV isovolumic time intervals were found to be increased at the expense of LV diastolic time in heart failure patients (17,18).
Since our findings and other previous studies suggested that tachycardia acts as a mechanism that aggravates abnormalities of the systolic-diastolic phase proportion in IDCM patients with narrow QRS complex, it is tempting to relate these observation to an exercise induced exacerbation of intra-LV dyssynchrony (3,7). This assumption is supported by recent findings. Lafitte et al. demonstrated that the mean degree of intra LV dyssynchrony did not change with exercise in heart failure patients with predominantly wide QRS duration (19). Similarily, Valzania et al. reported a lack of increase in LV dyssynchrony during dobutamine stress in patients with wide QRS duration (20). In contrast, Kurita et al. reported on an increased mechanical dyssynchrony during pacing-induced tachycardia in patients with normal QRS duration (21). A remarkable increase in intra-LV dyssynchrony was also demonstrated during pharmacological stress in systolic heart failure patients with normal QRS duration. The prevalence of dyssynchrony was low in this patient group at rest, but approached that observed in patients with wide QRS during stress (22). Furthermore, exercise LV dyssynchrony may play a more important role in the pathophysiology of left ventricular remodelling than baseline LV dyssynchrony (23). Probably for that reason additional hemodynamic benefits from cardiac resynchronization were obtained when RV-LV delay was separately optimized during exercise (24).
Our findings are in good concordance with these results and underline the highly dynamic nature of mechanical ventricular dyssynchrony in heart failure patients with normal QRS duration. By characterizing mechanical interventricular dyssynchrony, which was commonly found to be associated with intra-LV dyssynchrony in heart failure patients (16), we were able to demonstrate that the abnormal shortening of the duration of left ventricular diastole is closely related to the phenomenon of mechanical dyssynchrony. These findings may further explain why patients with normal QRS duration may benefit from cardiac resynchronization whether or not they have mechanical dyssynchrony under resting conditions. As demonstrated in patients with left bundle branch block biventricular pacing typically improves the time course of cardiac contraction at rest and during exercise by reducing LV dyssynchrony and thereby liberating a further segment of the RR interval for diastole (24). Considering that direct evaluation of LV dyssynchrony is still complex and time-consuming, the extent of diastolic time loss may represent a simple surrogate measure of the degree of LV dyssynchrony. Furthermore, the shortening of diastolic time is an often overlooked mechanism of cardiac dysfunction. The duration of diastole is a principal determinant of myocardial perfusion and of the blood volume received during diastole. Since both, a reduced myocardial perfusion reserve and diastolic heart failure, are an integral part of systolic heart failure any abnormal shortening of diastolic time, particularly during exercise, should exaggerate these preexisting abnormalities (25,26).
Limitations
The present study included a relatively small number of subjects. Our results were limited to patients with advanced heart failure and NYHA III functional class and may therefore not be readily generalizable to patients with a milder or a more severer degree of heart failure. Furthermore, all patients with dilated cardiomyopathy were examined under cardioactive medication. Beta-blocker therapy should have significantly contributed to a reduction in peak exercise heart rate in these patients. However, beta-blockers were shown to have no effect on the relationship between heart rate and diastolic time and to reduce rather than aggravate LV dyssynchrony in heart failure (27,28). Although patients with isolated LBBB were of younger age than the other three study groups it would be highly unlikely that the observed abnormalities in this group were due to an age-dependent effect. Basic cardiac time intervals as diastolic filling time were shown to be similar in young and elderly populations (29).
Conclusion
The current study, which evaluated the effect of exercise on heart rate corrected diastolic time in IDCM patients yielded the following findings: LBBB was a major determinant of the duration of LV systole, irrespective of the presence of left ventricular disease. The effect of the electrical conduction defect was independent from heart rate causing a relatively constant loss of diastolic time per beat. Unlike patients with LBBB those with IDCM and normal activation demonstrated a more dynamic response. A prolongation of LV systole and hence shortening of LV diastole was solely evident during exercise probably as a result of an increase in LV mechanical dyssynchrony.
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Veyrat C, Larrazet F, Pellerin D. Renewed interest in preejectional isovolumic phase: new applications of tissue Doppler indexes: implications to ventricular asynchrony. Am J Cardiol. 2005;96:1022-30
2. Duncan AM, Francis DP, Henein MY. Limitation of cardiac output by total isovolumic time during pharmacologic stress in patients with dilated cardiomyopathy: activation-mediated effects of left bundle branch block and coronary artery disease. J Am Coll Cardiol. 2003;41:121-8
3. Friedberg MK, Silverman NH. Cardiac ventricular diastolic and systolic duration in children with heart failure secondary to idiopathic dilated cardiomyopathy. Am J Cardiol. 2006;97:101-5
4. Plehn G, Vormbrock J, Zühlke C, Christ M, Perings C, Perings S, Trappe HJ, Meissner A. Disproportionate shortening of left ventricular diastolic duration in patients with dilated cardiomyopathy. Med Klin. 2007;102:707-13
5. Schiller NB, Shah PM, Crawford M, DeMaria A, Devereux R, Feigenbaum H, Gutgesell H, Reichek N, Shah D, Schnittger I, Silverman NH, Tajik AJ. American Society of Echocardiography Committee on Standards, Subcommittee on Quantitation of Two-Dimensional Echocardiogram: recommendations for quantitation of the left ventricle by two-dimensional echocardiography. J Am Soc Echocardiogr. 1989;2:358-367
6. Quinones MA, Waggoner AD, Reduto LA, Nelson JG, Young JB, Winters WL Jr, Ribeiro LG, Miller RR. A new, simplified and accurate method for determining ejection fraction with two-dimensional echocardiography. Circulation. 1981;64:744-753
7. Plehn G, Vormbrock J, Perings C, Machnick S, Zuehlke C, Trappe HJ, Meissner A. Loss of diastolic time as a mechanism of exercise-induced diastolic dysfunction in dilated cardiomyopathy. Am Heart J. 2008;155:1013-9
8. Standke R, Hör G, Maul D. Fully automated equilibrium radionuclide ventriculography. - Proposal of a method for routine use: exercise and follow-up. Eur J Nucl Med. 1983;8:77-83
9. Zhou Q, Henein M, Coats A, Gibson D. Different effects of abnormal activation and myocardial disease on left ventricular ejection and filling times. Heart. 2000;84:272-6
10. Hayashida W, Kumada T, Nohara R, Tanio H, Kambayashi M, Ishikawa N, Nakamura Y, Himura Y, Kawai C. Left ventricular regional wall stress in dilated cardiomyopathy. Circulation. 1990;82:2075-83
11. Latham RD, Thornton JW, Mulrow JP. Cardiovascular reserve in idiopathic dilated cardiomyopathy as determined by exercise response during cardiac catheterization. Am J Cardiol. 1987;59:1375-9
12. Moskowitz RL, Weschler BM. Left ventricular ejection time in aortic and mitral valve disease. Am Heart J. 1961;62:367-78
13. Arshad W, Duncan AM, Francis DP, O'Sullivan CA, Gibson DG, Henein MY. Systole-diastole mismatch in hypertrophic cardiomyopathy is caused by stress induced left ventricular outflow tract obstruction. Am Heart J. 2004;148:903-9
14. Braunwald E, Sarnoff SJ, Stainsby WN. Determinants of Duration and Mean Rate of Ventricular Ejection. Circ Res. 1958;6:319-325
15. Siri FM, Malhotra A, Factor SM. Sonnenblick EH, Fein FS. Prolonged ejection duration helps to maintain pump performance of the renal-hypertensive-diabetic rat heart: correlations between isolated papillary muscle function and ventricular performance in situ. Cardiovas Res. 1997;34:230-40
16. Ghio S, Constantin C, Klersy C, Serio A, Fontana A, Campana C, Tavazzi L. Interventricular and intraventricular asynchrony are common in heart failure patients, regardless of QRS duration. Eur Heart J. 2004;25:571-8
17. Hay I, Melenovsky V, Fetics BJ, Judge DP, Kramer A, Spinelli J, Reister C, Kass DA, Berger RD. Short-term effects of right-left heart sequential cardiac resynchronization in patients with heart failure, chronic atrial fibrillation, and atrioventricular nodal block. Circulation. 2004;110:3404-10
18. Salukhe TV, Francis DP, Morgan M. et al. Mechanism of cardiac output gain from cardiac resynchronization therapy in patients with coronary artery disease or idiopathic dilated cardiomyopathy. Am J Cardiol. 2006;97:1358-64
19. Lafitte S, Bordachar P, Lafitte M, Garrigue S, Reuter S, Reant P, Serri K, Lebouffos V, Berrhouet M, Jais P, Haissaguerre M, Clementy J, Roudaut R, DeMaria AN. Dynamic ventricular asynchrony: an exercise-echocardiography study. J Am Coll Cardiol. 2006;47:2253-9
20. Valzania C, Gadler F, Eriksson MJ, Olsson A, Boriani G, Braunschweig F. Electromechanical effects of cardiac resynchronization therapy during rest and stress in patients with heart failure. Eur J Heart Fail. 2007;9:644-50
21. Kurita T, Onishi K, Dohi K, Tanabe M, Fujimoto N, Tanigawa T, Setsuda M, Isaka N, Nobori T, Ito M. Impact of heart rate on mechanical asynchrony and left ventricular contractility in patients with heart failure and normal QRS duration. Eur J Heart Fail. 2007;9:637-43
22. Chattopadhyay S, Alamgir MF, Nikitin NP, Fraser AG, Clark AL, Cleland JG. The effect of pharmacological stress on intraventricular dyssynchrony in left ventricular systolic dysfunction. Eur J Heart Fail. 2008;10:412-420
23. Kang SJ, Lim HS, Choi BJ, Choi SY, Yoon MH, Hwang GS, Shin JH, Tahk SJ. The Impact of Exercise-induced Changes in Intraventricular Dyssynchrony on Functional Improvement in Patients with Nonischemic Cardiomyopathy. J Am Soc Echocardiogr. 2008 [Epub ahead of print]
24. Bordachar P, Lafitte S, Reuter S, Garrigue S, Laborderie J, Reant P, Jais P, Haisaguerre M, Roudaut R, Clementy J. Echocardiographic assessment during exercise of heart failure patients with cardiac resynchronization therapy. Am J Card. 2006;97:1622-25
25. Inoue T, Sakai Y, Morooka S, Hayashi T, Takayanagi K, Yamanaka T, Kakoi H, Takabatake Y. Coronary flow reserve in patients with dilated cardiomyopathy. Am Heart J. 1993;125:93-8
26. Little WC. Diastolic dysfunction beyond distensibility: adverse effects of ventricular dilatation. Circulation. 2005;112:2888-90
27. Boudoulas H, Rittgers SE, Lewis RP, Leier CV, Weissler AM. Changes in diastolic time with various pharmacologic agents: implication for myocardial perfusion. Circulation. 1979;60:164-9
28. Takemoto Y, Hozumi T, Sugioka K, Takagi Y, Matsumura Y, Yoshiyama M, Abraham TP, Yoshikawa J. Beta-blocker therapy induces ventricular resynchronization in dilated cardiomyopathy with narrow QRS complex. J Am Coll Cardiol. 2007;49:778-83
29. Stratton JR, Levy WC, Caldwell JH, Jacobson A, May J, Matsuoka D, Madden K. Effects of aging on cardiovascular responses to parasympathetic withdrawal. J Am Coll Cardiol. 2003;41:2077-83
Author contact
![]() Correspondence to: Gunnar Plehn, MD, Department of Cardiology and Angiology, Marienhospital Herne, University of Bochum, Hölkeskampring 40, 44625 Herne, Germany. Tel.: (0049)-2323-4995617; Fax: (0049)-2323-499301; Email: gunnar.plehnde
Correspondence to: Gunnar Plehn, MD, Department of Cardiology and Angiology, Marienhospital Herne, University of Bochum, Hölkeskampring 40, 44625 Herne, Germany. Tel.: (0049)-2323-4995617; Fax: (0049)-2323-499301; Email: gunnar.plehnde

 Global reach, higher impact
Global reach, higher impact