Impact Factor ISSN: 1449-1907
Int J Med Sci 2008; 5(6):303-308. doi:10.7150/ijms.5.303 This issue Cite
Research Paper
Enhanced Diagnostic Yield with Prolonged Small Bowel Transit Time during Capsule Endoscopy
1. Division of Gastroenterology and Hepatology, Johns Hopkins University School of Medicine, Johns Hopkins Hospital, Baltimore, Maryland, USA;
2. Division of Gastroenterology and Hepatology, State University of New York at Stony Brook, Stony Brook University Medical Center, Stony Brook, New York, USA
Received 2008-9-3; Accepted 2008-10-22; Published 2008-10-22
Abstract
Background: The effect of small bowel transit time (SBTT) on diagnostic yield during capsule endoscopy (CE) has not been previously evaluated. Our study aim was to assess the effect of SBTT on the likelihood of detecting intestinal pathology during CE. Methods: We reviewed collected data on CE studies performed at Johns Hopkins Hospital from January 2006 to June 2007. In patients investigated for anemia or obscure bleeding, the following lesions were considered relevant: ulcers, erosions, AVMs, red spots, varices, vascular ectasias, and presence of blood. In patients with diarrhea or abdominal pain, ulcers, erosions, and blood were considered relevant. Age, gender, study indication, hospital status, and quality of bowel preparation were identified as candidate risk factors affecting SBTT. Univariate logistic and linear regression analyses were performed to study the effect of SBTT on diagnostic yield. Results: Total of 212 CE studies were analyzed; most were in outpatients (n=175, 82.9%) and with excellent bowel preparation (n=177, 83.5%). Mean SBTT was 237.0min (3.9hrs). Age, gender, bowel prep, hospital status, and study indication did not significantly affect SBTT. However, increased SBTT was independently associated with increased diagnostic yield; OR=1.7 in SBTT=2-4hr (p=0.41), OR=1.8 in SBTT=4-6hrs (p=0.30), OR=9.6 in SBTT=6-8hrs (p=0.05). Conclusion: Prolonged SBTT during CE (>6 hr) is associated with an increased diagnostic yield. This may be due to a positive effect on image quality during a “slower” study. The use of promotility agents may adversely affect the ability of CE to detect significant intestinal pathology.
Keywords: capsule endoscopy, small bowel transit time, diagnostic yield, intestinal pathology
INTRODUCTION
Wireless capsule endoscopy (CE) was first developed for advanced imaging of the small intestine.1-3 Although its clinical efficacy is most proven in the diagnosis of obscure gastrointestinal bleeding,4-6 it has also been helpful as an adjunct to radiological studies for patients with suspected Crohn's disease, Celiac disease, small bowel tumors, anemia of unknown origin, chronic abdominal pain, and other indications.7-11
The quality of a CE study for any given patient is largely dependent upon capsule transit time through the stomach and small intestine. Early reports showed that approximately 20% of patients undergoing CE had incomplete studies based on failure to visualize the cecum within the allotted lifetime of the battery pack.12-13 Delayed gastric emptying and small bowel dysmotility can both significantly affect the rate at which the capsule moves from stomach to cecum. In order to increase capsule transit speed, some have advocated the use of promotility agents such as erythromycin14 and metocloperamide15 just prior to capsule ingestion. Others have found, however, that use of such agents may adversely affect image quality within the small bowel.16
The aim of our study was to assess the affect of small bowel transit time on the ability of CE to detect intestinal pathology in a large cohort of patients. The secondary aim was to identify candidate risk factors that may be used to predict capsule passage time through the small bowel.
METHODS
Consecutive patients undergoing CE without the use of promotility agents at Johns Hopkins Hospital between January 2006 and June 2007 were reviewed for study. Permission to review patient records was granted by the Johns Hopkins University Institutional Review Board.
All patients were asked to refrain from eating or drinking at least eight hours prior to swallowing the Given™ M2A video capsule endoscope (Given Imaging Ltd., Yoqneam, Israel). Laxative bowel preparation was not used. Patients were allowed to eat and drink four hours after the start of their study. Each CE study was interpreted by one of five board-certified/board-eligible gastroenterologists (J.M.B., S.A.G., P.M., J.O.C., and E.Y.) using the RAPID 4™ software system (Given Imaging Ltd., Duluth, GA). All five readers had experience reviewing greater than 50 cases each. Images were reviewed with two or four simultaneous frames at a speed of 8-15 frames/second. All captured thumbnail images and summary reports were re-examined and verified by a separate, board-certified gastroenterologist (G.E.M.) with at least 500 cases of experience. The verifying physician used his discretion to re-examine certain segments of the CE study, or to review the entire study in total, pending the results or findings of the initial review. There was greater than 95% concordance between the verifying reader (G.E.M.) and each of the five initial reviewers.
Following each study, the interpreting physician was asked to record all endoscopic findings within a CE database. In those patients undergoing CE for the indications of obscure gastrointestinal bleeding or anemia of unknown origin, the following pathological lesions were considered relevant: ulcers, erosions, arteriovenous malformations (AVMs), mucosal red spots, varices, venous ectasias, blood and blood clots. Red spots were defined as small, flat, pinpoint, red marks on the gastrointestinal mucosa believed to be a possible site of bleeding. AVMs were defined as larger red spots, or a confluence of mucosal spots, thought to represent a possible bleeding site. In all cases, if any one of the above lesions was detected, the study was marked as “positive” for significant findings. For those patients undergoing CE to investigate complaints of diarrhea or abdominal pain, the following lesions were considered relevant: ulcers, erosions, blood and blood clots. Again, if any one these findings was detected, the study yielded “positive” findings.
Small bowel transit time was calculated for each study by subtracting the time of first duodenal image from the time of first cecal image on the Given™ software program. This value was recorded for each patient in minutes and then categorized into the following parameters: 0-2 hours (hrs), 2-4 hrs, 4-6 hrs, and 6-8 hrs. All patients who experienced capsule failure in reaching the cecum or exiting the stomach in the allotted eight hour study time were excluded from the analysis. The quality of the bowel preparation in each study was subjectively graded as poor, average, or excellent by the interpreting physician at the time of the initial read. Additional recorded variables included physician reading time, patient gender, patient age (<40, 40-60, and >60 years), and inpatient versus outpatient status.
Statistical analyses were performed using Stata 9.0 (Stata Corp, College Station, Texas). Both univariate logistic regression analysis and linear regression analysis were utilized. Identification of a positive (significant) finding on CE was considered the main study outcome. The associations between small bowel transit time (SBTT) and positive CE findings were analyzed, and odds ratios (OR) with associated p-values and 95% confidence intervals (CI) were appropriately calculated. Additional covariates of patient age, gender, bowel preparation, and study indications were analyzed to detect positive associations with SBTT. Odds ratios with associated confidence intervals were again calculated; a p-value of ≤0.05 was considered statistically significant throughout.
RESULTS
Total of 212 patient CE recordings were studied between January 2006 and June 2007. Table 1 highlights the patient characteristics and study indications for each procedure. There were 88 males (41.5%) and 124 females (58.5%) with a mean age of 51.8 years. Most CE studies (n=175, or 82.5%) were performed in outpatients, and most patients had an excellent bowel preparation (n=177, or 83.5%). When the total number of studies was divided according to clinical indication, the single most common indication was obscure GI bleeding (n=78, or 36.8%). The investigation of abdominal pain was the second most common indication (n=54, or 25.5%), followed by anemia of unknown origin (n=42, or 19.8%) and diarrhea (n=38, or 17.9%).
Demographics in 212 patients undergoing CE.
| Patient Characteristics | Mean ± SD, or Number (%) | |
|---|---|---|
| Age (yrs) | 51.8 ± 16.0 | |
| Males | 88 (41.5) | |
| Females | 124 (58.5) | |
| Inpatient | 37 (17.4) | |
| Outpatient | 175 (82.5) | |
| Bowel Prep | ||
| Excellent | 177 (83.5) | |
| Average | 29 (13.7) | |
| Poor | 6 (2.8) | |
| Indication for CE | ||
| Obscure Bleeding | 78 (36.8) | |
| Abdominal Pain | 54 (25.5) | |
| Anemia | 42 (19.8) | |
| Diarrhea | 38 (17.9) | |
Small bowel transit time (SBTT) in the 212 patients undergoing CE is shown in Table 2. Mean passage time through the intestine was 239.0 minutes (3.9 hrs), with a range between 19 and 480 minutes. Most patients (n=163, or 76.9%) recorded a SBTT of 120-240 minutes (n=91) and 240-360 minutes (n=72). Total of 20 patients (9.4%) had an exceptionally rapid SBTT of 0-120 minutes, while 29 patients (13.7%) registered a delayed SBTT of 360-480 minutes. The average time for physician review of an entire CE study was 39.0 ± 10.2 minutes.
Small bowel transit time (SBTT) in 212 patients undergoing CE.
| Small Bowel Transit Time (SBTT) | Mean ± SD, or Number (%) |
|---|---|
| Average (min) | 237.0 ± 91.8 |
| 0-120 min | 20 (9.4) |
| 120-240 min | 91 (42.9) |
| 240-360 min | 72 (34.0) |
| 360-480 min | 29 (13.7) |
Table 3 shows the different pathological lesions identified in each patient undergoing CE. Overall, there were 181 lesions detected in 212 total patients. Mucosal red spots were the most common lesion recognized (56/181, or 30.9%), followed by mucosal erosions (45/181, or 24.9%), AVMs (33/181, or 18.2%), ulcers (27/181, or 14.9%), venous ectasias (8/181, or 4.4%), and intestinal varices (2/181, or 1.1%). There were 10 patients (5.5%) who had evidence of recent or ongoing intestinal bleeding without an obvious lesion seen during the study. These findings were considered relevant in those undergoing CE for any of the four indications.
Pathological lesions found in 212 CE studies according to study indication.
| Type of Lesion | Total (%) (n=212) | Obscure Bleeding (%) (n=78) | Abdominal Pain (%) (n=54) | Anemia (%) (n=42) | Diarrhea (%) (n=38) |
|---|---|---|---|---|---|
| Red Spots | 56 (30.9) | 20 (26.0) | 16 (34.8) | 14 (38.9) | 6 (27.3) |
| Erosions | 45 (24.9) | 18 (23.4) | 15 (32.6) | 6 (16.7) | 6 (27.3) |
| AVMs | 33 (18.2) | 15 (19.5) | 7 (15.2) | 10 (27.8) | 1 (4.5) |
| Ulcers | 27 (14.9) | 11 (14.3) | 5 (10.9) | 2 (5.6) | 9 (40.9) |
| Varices | 2 (1.1) | 2 (2.6) | 0 | 0 | 0 |
| Venous Ectasias | 8 (4.4) | 5 (6.5) | 3 (6.5) | 0 | 0 |
| Blood Clots or Bleeding | 10 (5.5) | 6 (7.8) | 0 | 4 (11.1) | 0 |
| Total No. Lesions | 181 | 77 | 46 | 36 | 22 |
When the total number of CE findings was divided according to indication for study, mucosal red spots were the most common finding in those patients investigated for obscure GI bleeding, abdominal pain, and anemia of unknown origin (Table 3). In patients with complaints of diarrhea, however, the most common lesion identified was an ulcer (40.9%). Mucosal erosions were the second most common lesion discovered in studies performed for obscure GI bleeding, abdominal pain, and diarrhea; while AVMs were second (27.8%) among patients with anemia of unknown origin.
Table 4 summarizes the odds ratios (OR) with respective p-values for the association between small bowel transit time (SBTT) and reader detection of a positive finding during CE. Compared to patients with SBTT of 0-120 minutes (0-2 hrs), there was a weak association between positive findings and SBTT of 120-240 minutes (2-4 hrs) (OR=1.7, 95%CI=0.5-6.2, p=0.41) and 240-360 minutes (4-6 hrs) (OR=1.8, 95%CI=0.5-6.6, p=0.30). Conversely, there was a stronger association between transit time and positive findings in those patients with SBTT of 360-480 minutes (6-8 hrs) (OR=9.6, 95%CI=1.9-10.5, p=0.05). This finding was statistically significant (Figure 1).
Association between SBTT and reader detection of a positive finding.
| Small Bowel Transit Time (SBTT) | Odds Ratio (95% C.I.) | p-value |
|---|---|---|
| 120-240 min | 1.7 (0.5-6.2) | 0.41 |
| 240-360 min | 1.8 (0.5-6.6) | 0.30 |
| 360-480 min | 9.6 (1.9-10.5) | 0.05 |
Odds of detecting a positive finding during CE according to small bowel transit time (x-axis).
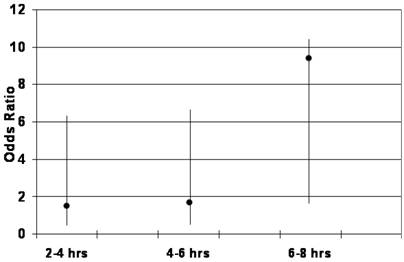
Table 5 outlines the results of analysis demonstrating a lack of association between SBTT and patient age, gender, bowel preparation, hospital status, and indication for study. Compared to patients <40 years old (mean SBTT=242.4 min), average passage time through the small intestine was 237.3 and 229.8 minutes among patients 40-60 and >60 years old, respectively (p=0.70, 0.50). In male patients, mean SBTT was only 3.9 minutes slower than female patients (235.1 min vs. 231.2 min, p=0.71). There was a trend towards more rapid transit time in patients with a cleaner bowel preparation; that is, compared to patients with a poor bowel prep (SBTT=253.7 min), mean SBTT was 240.6 and 237.5 minutes in those with an average and excellent prep, respectively (p=0.75, 0.67). Outpatients recorded a slightly faster transit time than inpatients undergoing CE (236.4 min vs. 241.5 min, p=0.61), but this difference was not significant. Finally, compared to those undergoing CE for anemia of unknown origin (mean SBTT=245.1 min), there were no statistically significant differences in SBTT for those in the obscure GI bleeding group (235.1 min, p=0.57), diarrhea group (225.6 min, p=0.35), or abdominal pain group (243.7 min, p=0.94).
Association between small bowel transit time (SBTT) and age, gender, bowel preparation, hospital status, and indication for study.
| Patient Factors | SBTT (min) | p-value | |
|---|---|---|---|
| Age | |||
| <40 yrs | 242.4 | -- | |
| 40-60 yrs | 237.3 | 0.70 | |
| >60 yrs | 229.8 | 0.50 | |
| Females | 231.2 | -- | |
| Males | 235.1 | 0.71 | |
| Bowel Preparation | |||
| Poor | 253.7 | -- | |
| Average | 240.6 | 0.75 | |
| Excellent | 237.5 | 0.67 | |
| Inpatients | 241.5 | -- | |
| Outpatients | 236.4 | 0.61 | |
| Indication for Study | |||
| Anemia | 245.1 | -- | |
| Obscure Bleeding | 235.1 | 0.57 | |
| Diarrhea | 225.6 | 0.35 | |
| Abdominal Pain | 243.7 | 0.94 | |
DISCUSSION
Since the inception of capsule endoscopy (CE), it has been most frequently and most effectively utilized in the evaluation of obscure gastrointestinal bleeding.18-19 More recently the indications have broadened with its usefulness demonstrated in the diagnosis of inflammatory bowel disease and the evaluation of chronic abdominal pain.7-11 The utility of a particular CE study, however, is largely dependent upon capsule transit time through the small intestine. Nearly one-fifth of patients experience capsule study failure;12-13 that is, lack of cecal visualization during the allotted study time. The reasons for an incomplete study are many, but some causes include delayed gastric emptying, chronic intestinal dysmotility, small bowel strictures, and areas of intestinal diverticulosis promoting regional transit abnormalities.20 As a result, the use of promotility agents such as domperidone, erythromycin, metocloperamide, and even chewing gum have been advocated in order to decrease small bowel transit time, and thus diminish the likelihood of an incomplete study.14-15,17 At some centers, use of such agents prior to CE has become standard of practice; if not to prevent capsule failure, then to decrease overall study time and physician reader time.
One potential problem with decreasing intestinal transit time during CE is impaired visualization of the entire small bowel mucosa. During most CE procedures, the image quality in the proximal intestine is superior to that of the terminal ileum, mainly because of residual fecal material.16 Decreasing the gastric and small bowel emptying times with the use of promotility agents may, in effect, create poorer visualization as the capsule moves rapidly through the ileum or distal portions of intestine. In fact, Fireman et al studied 29 patients receiving 200 mg of erythromycin 1 hour prior to capsule ingestion.16 They showed that image quality was significantly diminished compared to 40 patients that were not pretreated with erythromycin, and to 26 patients that received polyethylene glycol (PEG) solution before the procedure. The reason for these findings may be related to poorer image acquisition, and overall weaker bowel preparation, as the capsule endoscope moves rapidly through regions of retained intestinal fluid or residual fecal material. It is on this background in which we aimed to assess the affect of small bowel transit time (SBTT) on the ability of CE to detect significant intestinal pathology in a large cohort of patients. Furthermore, we aimed to identify certain risk factors that may help to predict either rapid or delayed capsule passage time through the small bowel.
The results of our study suggest that a prolonged small bowel transit time during CE may be associated with a higher diagnostic yield. That is, in those patients that recorded an intestinal passage time of longer than six hours (average SBTT=3.9 hrs), the likelihood of detecting a positive finding was nearly 10 times greater than in those with a passage time under two hours (OR=9.6, 95%CI=1.9-10.5, p=0.05). Furthermore, patients with intestinal transit times between 2-4 hours and 4-6 hours were not associated with an increase in diagnostic yield (OR=1.7, p=0.41; OR=1.8, p=0.30 respectively). In addition, after studying over 200 patients undergoing CE without bowel preparations or the use of promotility agents, we found there to be a lack of association between small bowel transit time and patient age, gender, inpatient versus outpatient status, and the indication for the CE study. Lastly, we also demonstrated that the quality of a patient's bowel preparation does not appear to have a significant effect on intestinal transit time in our large cohort of patients.
How might a prolonged small bowel transit time be related to enhanced diagnostic yield during CE? Certainly one possibility is that the rate of intestinal transit may correspond with impaired motility in the context of small bowel pathology.21 In other words, intestinal peristalsis is reduced or diminished in the setting of a large mucosal ulceration or polypoid tumor. This, in turn, accounts for the increased likelihood of making a positive diagnosis in the presence of delayed intestinal emptying. On the other hand, the increase in diagnostic yield may be due to superior image acquisition or improvement in image quality as the capsule endoscope moves more slowly through areas of intestinal mucosa with discrete pathological lesions. In either case, a prolonged small bowel transit time appears to be associated with an increased chance of finding some form of pathology, thus arguing against the use of certain promotility agents to decrease CE procedure time, or to help prevent capsule failure.
The main limitation of our study is its retrospective design. A large prospective study would be necessary to verify our results, or provide further evidence that strengthens the association between delayed intestinal transit and enhanced diagnostic yield. It is currently unknown as to whether or not even a repeat CE study in the same patient would produce a similar intestinal transit time as the initial study. In the future, it is likely that improvements in CE software systems or capsule endoscope design will mitigate some of these issues. For example, modification in image acquisition that varies according to the quality of a patient's bowel preparation or the rate of intestinal transit would eliminate this problem, and thus more readily allow for the use of adjunctive promotility drugs. Likewise, a wider viewing angle on the camera itself, or improvements in software design that enable a greater number of images to be recorded and viewed, may also provide enhanced mucosal visualization and result in improved diagnostic capability, regardless of intestinal transit. Lastly, it is certainly within question as to whether or not the presence of mucosal red spots during CE represents truly relevant pathology. In our study, red spots were the most common lesion identified in patients undergoing CE for obscure GI bleeding and anemia of unknown origin; but from a clinician's standpoint, it is often difficult to attribute such lesions to significant intestinal blood loss, and thus meaningful pathology.
Despite these limitations, it appears plausible to assume that rapid small bowel transit time may limit our detection capabilities in some patients undergoing CE. As such, the widespread, preemptive use of promotility agents might contribute to this negative effect, and the use of such agents should be considered on a case-by-case basis. Furthermore, one may speculate that in patients with exceptionally rapid intestinal transit (e.g. <2 hrs), use of anticholinergics or other motility-delaying agents would provide a more complete study with maximum mucosal detection; at least until improvements in the current versions of the capsule endoscope and software system allow us to overcome this issue.
In conclusion, our retrospective study demonstrated that a prolonged small bowel transit time (>6 hrs) during CE may be associated with an increased diagnostic yield. This finding does not appear to be related to other factors such as the quality of the bowel preparation, patient age or gender, inpatient versus outpatient status, or indication for the study. There may be a positive effect on image quality during a “slower” study, thereby suggesting the use of promotility agents adversely affects the ability of CE to detect significant intestinal pathology.
Abbreviations
AVM: arteriovenous malformation; CE: capsule endoscopy; SBTT: small bowel transit time.
Specific author contributions
Dr. Buscaglia initiated the study design, aided in the data analysis, and prepared the manuscript. Dr. Kapoor performed the majority of the data analysis. Dr. Bucobo aided in the data analysis and edited the manuscript draft. Drs. Clarke, Giday, Magno and Yong collectively interpreted greater than 75% of the CE studies, and each edited the manuscript draft. Dr. Mullin reviewed all CE studies, contributed to the study design and data analysis, and edited the manuscript draft.
Conflict of Interest
None of the authors of this manuscript have any relevant financial disclosures or conflicts of interest to state. There are no personal, financial, or other relevant relationships with Given Imaging.
References
1. Iddan G, Meron G, Glukhovsky A. et al. Wireless capsule endoscopy. Nature. 2000:405-7
2. Meron GD. The development of the swallowable video capsule M2A. Gastrointest Endosc. 2000;52:817-9
3. ASGE Technology Assessment Committee. Wireless capsule endoscopy. Gastrointest Endosc. 2002;56:621-4
4. Saurin JC, Delvaux M, Vahedi K. et al. Clinical impact of capsule endoscopy compared to push enteroscopy: 1-year follow-up study. Endoscopy. 2005;37:318-23
5. Hartman D, Schmidt H, Bolz G. et al. A prospective two-center study comparing wireless capsule endoscopy with intraoperative enteroscopy in patients with obscure GI bleeding. Gastrointest Endosc. 2005;61:826-32
6. Ell C, Remke S, May A. et al. The first prospective controlled trial comparing wireless capsule endoscopy with push enteroscopy in chronic gastrointestinal bleeding. Endoscopy. 2002;34:685-9
7. Eliakim R, Fischer D, Suissa A. et al. Wireless capsule video endoscopy is a superior diagnostic tool in comparison to barium follow-through and computerized tomography in patients with suspected Crohn's disease. Eur J Gastroenterol Hepatol. 2003;15:363-7
8. Costamagna G, Shah SK, Riccioni ME. et al. A prospective trial comparing small bowel radiographs and video capsule endoscopy for suspected small bowel disease. Gastroenterology. 2002;123:999-05
9. Herreiras JM, Caunedo A, Rodriguez-Tellez M. et al. Capsule endoscopy in patients with suspected Crohn's disease in negative endoscopy? Endoscopy. 2003;35:1-5
10. Bardan E, Nadler M, Chowers Y. et al. Capsule endoscopy for the evaluation of patients with chronic abdominal pain. Endoscopy. 2003;35:688-9
11. Vasquez-Iglesias J, Gonzalez-Conde B, Estevez-Prieto E. et al. A prospective study of COX-2 inhibitors versus nonspecific NSAIDs induced small bowel lesions using video capsule endoscopy. Endoscopy. 2003;35:A183
12. Fireman Z, Mahajna E, Broide E. et al. Diagnosing small bowel Crohn's disease with wireless capsule endoscopy. Gut. 2003;52:390-2
13. Mylonaki M, Fritscher-Ravens A, Swain P. Wireless capsule endoscopy: a comparison with push enteroscopy in patients with gastroscopy and colonoscopy negative gastrointestinal bleeding. Gut. 2003;502:1122-6
14. Leung WK, Chan FK, Fung SS. et al. Effect of oral erythromycin on gastric and small bowel transit of capsule endoscopy. World J Gastroenterol. 2005;11:4865-8
15. Selby W. Complete small-bowel transit in patients undergoing capsule endoscopy: determining factors and improvement with metacloperamide. Gastrointest Endosc. 2005;61:80-5
16. Fireman Z, Paz D, Kopelman Y. Capsule endoscopy: improving transit time and image view. World J Gastroenterol. 2005;11:5863-6
17. Apostolopoulos P, Kalantzis C, Gralnek IM. et al. Clinical effectiveness of chewing-gum in accelerating capsule endoscopy transit time—a prospective randomized, controlled pilot study. Aliment Pharmacol Ther. 2008;28:405-11
18. Soussan BE, Antonietti M, Herve S. et al. Diagnostic yield and therapeutic implications of capsule endoscopy in obscure gastrointestinal bleeding. Gastroenterol Clin Biol. 2004;28:1068-73
19. Gupta R, Lakhtakia S, Tandan M. et al. Capsule endoscopy in obscure gastrointestinal bleeding—an Indian experience. Indian J Gastroenterol. 2006;25:188-90
20. Sears DM, Avots-Avotins A, Culp K. et al. Frequency and clinical outcome of capsule retention during capsule endoscopy for GI bleeding obscure origin. Gastrointest Endosc. 2004;60:822-7
21. Endo H, Matsuhashi N, Inamori M. et al. Abdominal surgery affects small bowel transit time and completeness of capsule endoscopy. Dig Dis Sci. in press
Author contact
Correspondence to: Jonathan M. Buscaglia, M.D., Stony Brook University Medical Center, State University of New York at Stony Brook, Health Sciences Center, Tower 17, Room 060, Stony Brook, New York 11794 USA, (p) 631-444-2119, (f) 631-444-8886, jmbuscagliacc.sunysb.edu.

 Global reach, higher impact
Global reach, higher impact