Impact Factor
ISSN: 1449-1907
Int J Med Sci 2024; 21(6):1117-1128. doi:10.7150/ijms.94803 This issue Cite
Research Paper
Evaluation of Pm2.5 Influence on Human Lung Cancer Cells Using a Microfluidic Platform
1. International Ph.D. Program in Cell Therapy and Regenerative Medicine, Taipei Medical University, 250 Wuxing St., Taipei 11031, Taiwan.
2. Institute of Applied Mechanics, National Taiwan University, Taipei 10617 Taiwan.
3. Department of Life Science, National Dong-Hwa University, Hualien, 974301 Taiwan.
4. Department of Biomedical Sciences and Engineering, National Central University, Taoyuan 320, Taiwan.
5. Department of Statistics & Data Science, University of California Los Angeles, Los Angeles, CA 90095, USA.
6. Department of Critical Care Medicine, Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, New Taipei 231, Taiwan.
7. The Division of Chest Medicine, Department of Internal medicine, Taoyuan General Hospital, Tao Yuan, Ministry of Healthy and Welfare, Taoyuan 330, Taiwan.
8. Center for Astronautical Physics and Engineering, National Central University, Taoyuan, 320, Taiwan.
9. School of Biomedical Engineering, Taipei Medical University, 250 Wuxing St., International PhD Program for Biomedical Engineering, Taipei 11031, Taiwan.
Abstract
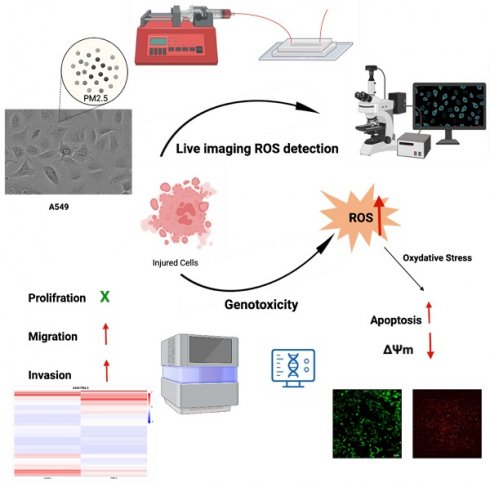
In this study, we developed a microfluidic device that is able to monitor cell biology under continuous PM2.5 treatment. The effects of PM2.5 on human alveolar basal epithelial cells, A549 cells, and uncovered several significant findings were investigated. The results showed that PM2.5 exposure did not lead to a notable decrease in cell viability, indicating that PM2.5 did not cause cellular injury or death. However, the study found that PM2.5 exposure increased the invasion and migration abilities of A549 cells, suggesting that PM2.5 might promote cell invasiveness. Results of RNA sequencing revealed 423 genes that displayed significant differential expression in response to PM2.5 exposure, with a particular focus on pathways associated with the generation of reactive oxygen species (ROS) and mitochondrial dysfunction. Real-time detection demonstrated an increase in ROS production in A549 cells after exposure to PM2.5. JC1 assay, which indicated a loss of mitochondrial membrane potential (ΔΨm) in A549 cells exposed to PM2.5. The disruption of mitochondrial membrane potential further supports the detrimental effects of PM2.5 on A549 cells. These findings highlight several adverse effects of PM2.5 on A549 cells, including enhanced invasion and migration capabilities, altered gene expression related to ROS pathways, increased ROS production and disruption of mitochondrial membrane potential. These findings contribute to our understanding of the potential mechanisms through which PM2.5 can impact cellular function and health.
Keywords: Lung cancer, PM2.5, invasion, migration, ROS, mitochondrial dysfunction

 Global reach, higher impact
Global reach, higher impact