Impact Factor ISSN: 1449-1907
Int J Med Sci 2018; 15(13):1522-1529. doi:10.7150/ijms.28442 This issue Cite
Research Paper
Comparisons of Pressure-controlled Ventilation with Volume Guarantee and Volume-controlled 1:1 Equal Ratio Ventilation on Oxygenation and Respiratory Mechanics during Robot-assisted Laparoscopic Radical Prostatectomy: a Randomized-controlled Trial
Department of Anesthesiology and Pain Medicine, and Anesthesia and Pain Research Institute, Yonsei University College of Medicine, Seoul, Republic of Korea.
Received 2018-7-10; Accepted 2018-9-6; Published 2018-10-20
Abstract
Background: During robot-assisted laparoscopic radical prostatectomy (RALP), steep Trendelenburg position and carbon dioxide pneumoperitoneum are inevitable for surgical exposure, both of which can impair cardiopulmonary function. This study was aimed to compare the effects of pressure-controlled ventilation with volume guarantee (PCV with VG) and 1:1 equal ratio ventilation (ERV) on oxygenation, respiratory mechanics and hemodynamics during RALP.
Methods: Eighty patients scheduled for RALP were randomly allocated to either the PCV with VG or ERV group. After anesthesia induction, volume-controlled ventilation (VCV) was applied with an inspiratory to expiratory (I/E) ratio of 1:2. Immediately after pneumoperitoneum and Trendelenburg positioning, VCV with I/E ratio of 1:1 (ERV group) or PCV with VG using Autoflow mode (PCV with VG group) was initiated. At the end of Trendelenburg position, VCV with I/E ratio of 1:2 was resumed. Analysis of arterial blood gases, respiratory mechanics, and hemodynamics were compared between groups at four times: 10 min after anesthesia induction (T1), 30 and 60 min after pneumoperitoneum and Trendelenburg positioning (T2 and T3), and 10 min after desufflation and resuming the supine position (T4).
Results: There were no significant differences in arterial blood gas analyses including arterial oxygen tension (PaO2) between groups throughout the study period. Mean airway pressure (Pmean) were significantly higher in the ERV group than in the PCV with VG group T2 (p<0.001) and T3 (p=0.002). Peak airway pressure and hemodynamic data were comparable in both groups.
Conclusion: PCV with VG was an acceptable alternative to ERV during RALP producing similar PaO2 values. The lower Pmean with PCV with VG suggests that it may be preferable in patients with reduced cardiovascular function.
Keywords: arterial oxygenation, autoflow, equal ratio ventilation, pressure-controlled ventilation with volume guarantee, respiratory mechanics, robot-assisted laparoscopic radical prostatectomy, volume-controlled ventilation
Introduction
Robot-assisted laparoscopic radical prostatectomy (RALP) has been widely used because it provides many benefits over open procedures [1,2]. However, steep Trendelenburg position and carbon dioxide (CO2) pneumoperitoneum are inevitably used during RALP to optimize surgical exposure, both of which have a major impact on the cardiovascular and pulmonary systems [3-5]. Combined with CO2 pneumoperitoneum, steep Trendelenburg position increases ventricular filling pressure and airway pressure during positive pressure ventilation, potentially resulting in hypoxia, pulmonary edema, and heart failure [6]. In addition, upward movement of the diaphragm leads to pulmonary atelectasis and reduced functional residual capacity and lung compliance [7,8]. Therefore, ventilatory strategies are required to protect the respiratory system and minimize adverse effects of the steep Trendelenburg position and CO2 pneumoperitoneum during RALP.
Inverse inspiratory to expiratory (I/E) ratio ventilation or prolonged I/E ratio ventilation (i.e., a 1:1 ratio) is a mechanical ventilation strategy proposed for improving oxygenation in acute respiratory distress syndrome. By increasing inspiratory time during the respiratory cycle, more alveoli are kept open, with the goal of reducing the occurrence of atelectasis and limiting peak inspiratory pressure (Ppeak). Recent studies, including a meta-analysis, have reported that prolonged I/E ratio ventilation during anesthesia improves respiratory mechanics and oxygenation [9-12]. However, concerns regarding possible intrinsic positive end-expiratory pressure (PEEP) and decreases in cardiac output during prolonged I/E ratio ventilation still limit its clinical application, especially in patients with chronic obstructive pulmonary disease (COPD) [9,10,13].
Pressure-controlled ventilation with volume guarantee (PCV with VG) is a type of pressure regulated volume control (PRVC) ventilation modes which has both features of volume-controlled ventilation (VCV) and pressure-controlled ventilation (PCV). PCV with VG can deliver a constant tidal volume with a constant inspiratory pressure, using a decelerating flow pattern. In laparoscopic surgery, PCV might be advocated to maintain sufficient tidal volume and oxygenation against increases in airway pressure after Trendelenburg positioning and CO2 pneumoperitoneum. Previously, PCV alone failed to improve arterial oxygen tension but significantly reduced Ppeak and improved lung compliance compared to VCV during RALP [6]. Therefore, it is necessary to investigate the effects of PCV with VG, which combines the advantages of VCV and PCV on oxygenation and respiratory mechanics in laparoscopic surgery. Comparisons of PCV with VG with prolonged I/E ratio ventilation are needed, as prolonged I/E ratio ventilation has been suggested to improve oxygenation in laparoscopic surgery.
The aim of this study was to compare the effects of PCV with VG and volume-controlled 1:1 equal ratio ventilation (ERV) on gas exchange, respiratory mechanics and hemodynamics in patients undergoing RALP.
Methods
This prospective randomized double-blind study was conducted at Severance Hospital, Yonsei University Health System, Seoul, Korea, approved by our Institutional Review Board (ref: 4-2017-0400, Chairperson Professor Sun Young Rha) on 24 June 2017, and written informed consent was obtained from all subjects participating in the trial. The trial was registered prior to patient enrollment at www.ClinicalTrials.gov (NCT03202953, principal investigator Jin Ha Park, date of registration: on 29 June, 2017).
Patients
After obtaining written informed consent from all patients, 80 men between 20 and 80 years of age scheduled for elective RALP using the da VinciTM Surgical System (Intuitive Surgical, Inc., Mountain View, CA, USA) under general anesthesia were enrolled in the study. Patients were excluded if they had COPD, reactive airway disease, another pulmonary disease, a left ventriclular ejection fraction <50%, or obesity (body mass index > 30 kg/m2). We also excluded patients who were unable to read the informed consent form.
Patients were randomly assigned in a 1:1 ratio to either the PCV with VG or ERV group using a computerized randomization table by an investigator not involved in patient care. For each patient, anesthetic care was provided in the same manner by an independent, experienced anesthesiologist. The attending anesthesiologists were aware of the group assignment, but the patients, urologists, and outcome assessors were blinded to group assignment.
Anesthetic management
Upon arrival at the operating room, standard monitoring devices were applied. General anesthesia was induced with intravenous propofol 1.5 mg/kg, desflurane at an end-tidal concentration of 5%-6% with 100% oxygen, and an intravenous remifentanil infusion at 0.5-1 μg/kg/min. Intravenous rocuronium 0.6 mg/kg was used for neuromuscular blockade to facilitate intubation. After endotracheal intubation, anesthesia was maintained with desflurane at an end-tidal concentration of 5%-6% in an air-oxygen mixture (fraction of inspired oxygen = 0.5) and a remifentanil infusion at 0.1-0.3 μg/kg/min. The depth of anesthesia was adjusted to maintain the bispectral index score (BIS) (A-2000 BIS MonitorTM; Aspect Medical System Inc., Newton, MA) between 40 and 60. A continuous infusion of intravenous rocuronium 0.6 μg/kg/h was administered throughout surgery. The radial artery was cannulated after anesthesia induction for continuous blood pressure monitoring and arterial blood sampling. Mean arterial pressure (MAP) and heart rate (HR) were maintained within approximately 20% of baseline. At the end of surgery, all anesthetic agents were discontinued, oxygen 100% was administered and residual neuromuscular blockade was antagonized with sugammadex 4 mg/kg.
Intervention (ventilation management)
Immediately after induction, patients were ventilated with VCV mode using an I/E ratio of 1:2, a tidal volume of 8 mL/kg ideal body weight, no PEEP, and an inspiratory pause of 10%. Ideal body weight was calculated using the following formula for men: 50 + 0.91 (height [cm] - 152.4) [14]. After CO2 pneumoperitoneum was established with an intra-abdominal pressure of 15 mmHg in the supine position, each patient was placed in a 30º Trendelenburg position and the ventilation mode was adjusted according to the group allocation. In the PCV with VG group, the ventilation mode was changed from VCV to Autoflow mode by Primus® anesthesia machine (Dräger, Lübeck, Germany) using the same initial setting. In the ERV group, the I/E ratio was changed from 1:2 to 1:1, while maintaining but the other initial setting. At the end of surgery, immediately after CO2 desufflation and resumption of the supine position, the ventilation mode was changed to back to VCV with an I/E ratio of 1:2 for all patients. The respiratory rate was adjusted to maintain an end-tidal CO2 (ETCO2) between 35 and 45 mmHg throughout surgery in both groups. Patients were withdrawn from the study if more than 40 cm H2O of Ppeak was required to maintain the tidal volume or if oxygen desaturation (SpO2 < 95%) occurred.
Clinical evaluations
The primary end point was the level of arterial oxygen tension (PaO2) measured at 30 min after the initiation of the Trendelenburg position. The secondary end points were arterial blood gas analysis (ABGA) results, respiratory mechanics data and hemodynamics data, which were collected at four times: 10 min after anesthesia induction, while in the supine position (T1); 30 min after initiation of the Trendelenburg position and CO2 pneumoperitoneum (T2); 60 min after initiation of the Trendelenburg position and CO2 pneumoperitoneum (T3); and 10 min after CO2 desufflation and resumption of the supine position (T4). Arterial pH, PaO2, arterial carbon dioxide tension (PaCO2) levels were obtained from the ABGA results. Respiratory mechanics included Ppeak, plateau airway pressure (Pplat), mean airway pressure (Pmean), static compliance (Cstat), ETCO2, and respiratory rate, which were measured by the Primus® anesthesia machine (Dräger, Lübeck, Germany). Hemodynamic data included MAP, HR, and SpO2. Intraoperative data, such as duration of surgery, volume of fluid and blood administered, urine output, blood loss, and use of vasoactive drugs, were recorded. Postoperative data, including duration of postoperative hospital stay and postoperative complications, were also assessed.
Statistical analysis
Sample size was calculated based on the results of a previous study comparing VCV with an I/E ratio of 1:1 versus 1:2 during RALP [11]. In that study, PaO2 at 30 min after initiation of the Trendelenburg position was 167 ± 32 mmHg in the 1:1 group. We considered a difference of 15% (25 mm Hg) in PaO2 between PCV with VG and ERV with an I/E ratio of 1:1 during VCV as clinically relevant. With a type 1 error (α) of 5% and power (1-β) of 90%, 36 patients were required in each group. Taking into consideration a potential 10% dropout rate, we decided to enroll 40 patients in each group.
Continuous variables are shown as mean ± standard deviation or median (interquartile range). Dichotomous variables are expressed as number of patients (percentage). Continuous variables were compared using independent Student's t tests or Mann-Whitney U tests, and dichotomous variables were compared using Chi-square or Fisher's exact tests as appropriate. A linear mixed model with patient indicator as a random effect, and group, time, and group-by-time as fixed effects was used to analyze repeatedly measured variables such as PaO2, PaCO2, Ppeak and Pplat. When interactions of group, time, and group-by-time of variables were statistically significant, post hoc analyses were performed with Bonferroni correction to adjust for multiple comparisons. SPSS 21 (SPSSFW, SPSS, IBM, Armonk, NY, USA) statistical software was used. P values less than 0.05 were considered statistically significant.
Results
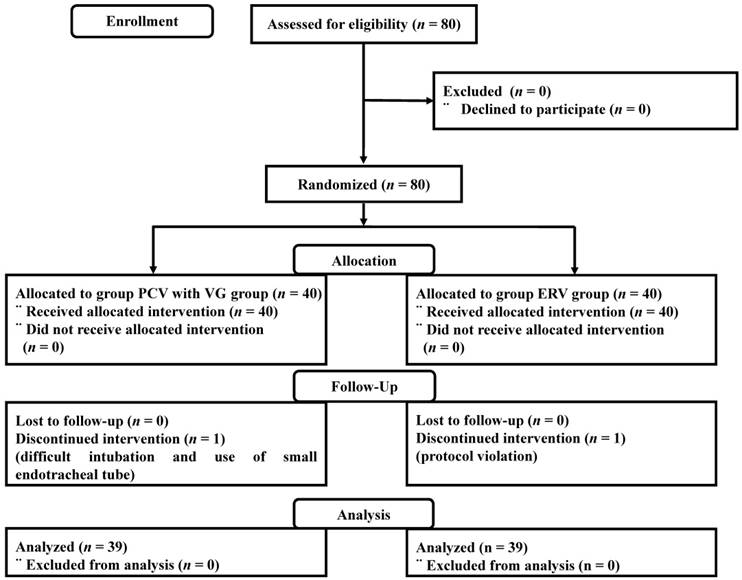
Between July 2017 and January 2018, a total of 80 patients were enrolled in the study. One patient in the PCV with VG group was excluded because endotracheal intubation was difficult and a small endotracheal tube was used. One patient in the ERV group was excluded because of a protocol violation. Consequently, 78 patients completed the study (Figure 1). Demographic and perioperative data were similar in the two groups (Table 1).
Patient enrolment into the study (using CONSORT recommendations). PCV with VG, pressure-controlled ventilation with volume guarantee; ERV, equal ratio ventilation.
Demographic and perioperative data
| PCV with VG Group (n = 39) | ERV Group (n = 39) | P value | |
|---|---|---|---|
| Age (yr) | 67 [62 - 74] | 67 [59 - 71] | 0.606 |
| Weight (kg) | 69 [63 - 73] | 67 [62 - 73] | 0.697 |
| Height (cm) | 169 [166-172] | 169 [164-171] | 0.700 |
| Body surface area (m2) | 1.80 [1.72 - 1.88] | 1.76 [1.70 - 1.83] | 0.234 |
| Hypertension | 14 (35.9) | 17 (43.6) | 0.488 |
| Diabetes | 10 (25.6) | 10 (25.6) | >0.999 |
| Anesthetic time (min) | 175 [160 - 200] | 165 [150 - 185] | 0.120 |
| Operation time (min) | 130 [113 - 149] | 119 [104 - 139] | 0.105 |
| Duration of Trendelenburg position (min) | 62 [55 - 100] | 61 [47 - 105] | 0.455 |
| Fluid intake (ml) | 1650 [1450 - 2100] | 1600 [1350 - 1900] | 0.246 |
| Urine output (ml) | 230 [100 - 400] | 250 [150 - 400] | 0.699 |
| Bleeding (ml) | 300 [200 - 500] | 250 [100 - 400] | 0.085 |
| Use of vasoconstrictors | 30 (76.9) | 25 (64.1) | 0.214 |
Data are presented as median (interquartile range) and numbers (%). PCV with VG, pressure-controlled ventilation with volume guarantee; ERV, 1:1 equal ratio ventilation.
ABGA and ETCO2 data are shown in Table 2. Linear mixed model analysis did not show significant differences between groups for the primary endpoint (PaO2 at 30 min after initiation of CO2 pneumoperitoneum and the Trendelenburg position). There were likewise no significant differences in ETCO2 results or ABGA data throughout the study period between the PCV with VG and ERV groups.
Arterial blood gas analysis and ETCO2 data measured at each time point
| PCV with VG Group (n = 39) | ERV Group (n = 39) | P value | |
|---|---|---|---|
| pH | 0.857a | ||
| T1 | 7.43 [7.43 - 7.47] | 7.44 [7.42 - 7.46] | 0.956 |
| T2 | 7.35 [7.33 - 7.40] | 7.35 [7.32 - 7.38] | 0.294 |
| T3 | 7.35 [7.32 - 7.38] | 7.35 [7.32 - 7.37] | 0.631 |
| T4 | 7.35 [7.31 - 7.38] | 7.33 [7.31 - 7.37] | 0.353 |
| PaO2 (mmHg) | 0.122a | ||
| T1 | 185.6 [160.3 - 218.3] | 177.8 [152.3 - 214.8] | 0.635 |
| T2 | 176.8 [142.9 - 196.3] | 181.0 [159.0 - 208.7] | 0.366 |
| T3 | 191.9 [162.3 - 209.3] | 180.3 [156.6 - 203.7] | 0.723 |
| T4 | 188.9 [161.6 - 203.4] | 196.7 [170.7 - 210.4] | 0.157 |
| PaCO2 (mmHg) | 0.593a | ||
| T1 | 32.5 [30.0 - 34.5] | 32.8 [30.7 - 35.5] | 0.265 |
| T2 | 41.8 [36.9 - 47.8] | 44.6 [38.4 - 48.2] | 0.094 |
| T3 | 41.3 [37.3 - 45.2] | 44.1 [40.9 - 47.4] | 0.077 |
| T4 | 42.2 [38.9 - 49.2] | 48.5 [40.2 - 51.6] | 0.165 |
| ETCO2 (mmHg) | 0.846a | ||
| T1 | 34 [32 - 36] | 34 [33 - 36] | 0.896 |
| T2 | 39 [37 - 43] | 42 [37 - 45] | 0.074 |
| T3 | 41 [37 - 43] | 41 [38 - 44] | 0.632 |
| T4 | 41 [39 - 45] | 43 [39 - 47] | 0.336 |
Data are presented as median (interquartile range). PCV with VG, pressure-controlled ventilation with volume guarantee; ETCO2, end-tidal carbon dioxide; ERV, 1:1 equal ratio ventilation; PaO2, arterial oxygen tension; PaCO2, arterial carbon dioxide tension; T1, 10 min after anaesthesia induction under supine position; T2, 30 min after initiation of CO2 pneumoperitoneum and Trendelenburg position; T3, 60 min after initiation of CO2 pneumoperitoneum and Trendelenburg position; T4, 10 min after CO2 desufflation and resuming the supine position. *P-value of time and group interaction derived from the linear mixed model. aPgroup × time = P value of the group and time interaction obtained by linear mixed model analysis.
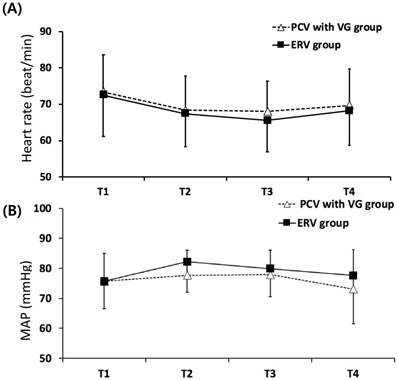
Respiratory data are shown in Table 3. The interaction of group and time for Pmean was significant between groups in the linear mixed model analysis (p = 0.038). After post hoc analysis with Bonferroni correction, Pmean was noted to be significantly lower in the PCV with VG group at 30 and 60 min after initiation of CO2 pneumoperitoneum and the Trendelenburg position (p<0.001 and p=0.002, respectively). Ppeak, Pplat and Cstat were not different between groups at any time. Hemodynamic data were similar between the two groups (Figure 2).
Respiratory mechanics measured at each time point
| PCV with VG Group (n = 39) | ERV Group (n = 39) | P value | |
|---|---|---|---|
| Ppeak (cm H2O) | 0.909a | ||
| T1 | 14 [13 - 15] | 14 [13 - 15] | 0.811 |
| T2 | 28 [26 - 32] | 29 [27 - 33] | 0.113 |
| T3 | 27 [25 - 29] | 28 [26 - 30] | 0.121 |
| T4 | 17 [16 - 19] | 18 [17-19] | 0.275 |
| Pplat (cm H2O) | 0.917a | ||
| T1 | 13 [12 - 14] | 13 [12 - 15] | 0.859 |
| T2 | 28 [26 - 30] | 29 [26 - 32] | 0.262 |
| T3 | 27 [25 - 29] | 27 [24 - 30] | 0.369 |
| T4 | 16 [14 - 17] | 16 [14 - 17] | 0.895 |
| Pmean (cm H2O) | 0.038a | ||
| T1 | 4.0 [4.0 - 4.3] | 4.0 [4.0 - 4.0] | 0.625 |
| T2 | 9.0 [8.0 - 9.0] | 10.0 [9.0 - 11.0] | <0.001 |
| T3 | 9.0 [7.8 - 9.3] | 10.0 [9.0 - 10.0] | 0.002 |
| T4 | 5.0 [4.0 - 5.0] | 5.0 [5.0 - 6.0] | 0.073 |
| Cstat (mL cm H2O-1) | 0.203a | ||
| T1 | 41.2 [37.0 - 45.1] | 43.4 [36.7 - 47.4] | 0.313 |
| T2 | 19.2 [17.4 - 21.6] | 18.1 [16.4 - 20.9] | 0.232 |
| T3 | 21.0 [18.5 - 22.4] | 18.9 [16.8 - 21.0] | 0.127 |
| T4 | 37.0 [32.1 - 39.9] | 35.7 [31.0 - 38.9] | 0.807 |
| RR (breaths min-1) | 0.015a | ||
| T1 | 14 [13 - 15] | 14 [12 - 14] | 0.098 |
| T2 | 17 [15 - 20] | 18 [16 - 20] | 0.839 |
| T3 | 16 [14 - 18] | 17 [14 - 19] | 0.291 |
| T4 | 16 [14 - 20] | 20 [18 - 20] | 0.038 |
Data are presented as median (interquartile range). PCV with VG, pressure-controlled ventilation with volume guarantee; ERV, 1:1 equal ratio ventilation; Ppeak, peak inspiratory pressure; Pplat, plateau airway pressure; Pmean, mean airway pressure; Cstat, static compliance; RR, respiratory rate. T1, 10 min after anesthesia induction under supine position; T2, 30 min after initiation of carbon dioxide (CO2) pneumoperitoneum and Trendelenburg position; T3, 60 min after initiation of CO2 pneumoperitoneum and Trendelenburg position; T4, 10 min after CO2 desufflation and resuming the supine position. aPgroup × time = P value of the group and time interaction obtained by linear mixed model analysis.
Perioperative hemodynamic variables. Values are mean ± standard deviation. PCV with VG, pressure-controlled ventilation with volume guarantee; ERV, equal ratio ventilation; MAP, mean arterial pressure. T1, 10 min after anesthesia induction under supine position; T2, 30 min after initiation of carbon dioxide (CO2) pneumoperitoneum and Trendelenburg position; T3, 60 min after initiation of CO2 pneumoperitoneum and Trendelenburg position; T4, 10 min after CO2 desufflation and resuming the supine position.
Postoperative outcomes were comparable between the two groups (Table 4). Eight patients in the PCV with VG group and 11 patients in the ERV group experienced postoperative fever; these rates were not significantly different (p=0.429).
Postoperative outcomes
| PCV with VG Group (n = 39) | ERV Group (n = 39) | P value | |
|---|---|---|---|
| PACU time (min) | 48 [36 - 54] | 45 [38 - 60] | 0.813 |
| Postoperative fever | 8 (20.5) | 11 (28.2) | 0.429 |
| Postoperative hospital stay (d) | 3 [2 - 4] | 2 [2 - 4] | 0.275 |
| Readmission within 30 days | 3 (7.7) | 3 (7.7) | >0.999 |
Data are presented as median (interquartile range) and numbers (%). PCV with VG, pressure-controlled ventilation with volume guarantee; ERV, 1:1 equal ratio ventilation; PACU, postoperative anesthesia care unit.
Discussion
The objective of this study was to compare the effects of PCV with VG and ERV on gas exchange, respiratory mechanics and hemodynamics during RALP. Our results indicate that, although Pmean was reduced with PCV with VG 30 and 60 min after initiation of CO2 pneumoperitoneum and Trendelenburg position, no differences in oxygenation were observed between the PCV with VG and ERV group. Gas exchange, respiratory mechanics except Pmean, and hemodynamics were also comparable regardless of the ventilator mode used.
PCV with VG is a type of dual-controlled ventilation mode that combines the advantages of PCV and VCV. This new ventilation mode includes Autoflow ventilation (Dräger), PCV with volume guaranteed (PCV-VG; General Electric), and PRVC (Maquet), and has the potential to reduce inspiratory pressure and atelectasis [15]. Theoretically, dual-controlled ventilation is suitable for maintaining an appropriate tidal volume during laparoscopic surgery, where sudden changes in intra-abdominal pressure may occur because of CO2 pneumoperitoneum and position changes. Otherwise, frequent adjustments in the Ppeak would be required with PCV to provide adequate ventilation according to the changes in lung compliance [16]. Notwithstanding these theoretical advantages, however, many studies evaluating PCV with VG have been conducted as cross-over studies [16-18]. Thus, scant information is available to assess the superiority of PCV with VG including Autoflow ventilation over other ventilation modes during laparoscopic surgery.
The goals of anesthetic management in laparoscopic surgery are to maintain oxygenation and prevent barotrauma. Although many studies have suggested that ERV enhances oxygenation in patients with acute respiratory distress syndrome [19,20], the effects of ERV on oxygenation during surgery remain controversial. In a meta-analysis of seven prospective trials involving one-lung ventilation or CO2 pneumoperitoneum, ERV significantly improved oxygenation at 60 min after intervention, but not at 20 or 30 min after intervention [12]. The main mechanism responsible for oxygen improvement by ERV is alveolar recruitment through an increased Pmean [21]. A higher Pmean allows collapsed alveoli to reopen in a manner similar to applying extrinsic PEEP; as a result, arterial oxygenation is improved and Ppeak is reduced [22,23]. Despite its theoretical benefits, ERV has the major drawback of possibly impeding venous return and reducing cardiac output. These hemodynamic effects limit widespread clinical application of ERV during surgery. Therefore, we conducted the present study to evaluate the hypothesis that PCV with VG ventilation might have clinical benefits during laparoscopic surgery if oxygenation or Ppeak are superior with PCV with VG, compared with ERV.
Contrary to our expectations, neither PCV with VG nor ERV demonstrated superiority for improving oxygenation in patients undergoing RALP. Pmean, which is a major determinant of arterial oxygenation, was slightly, but significantly lower in the PCV with VG group (9 cm H2O) compared to the ERV group (10 cm H2O). However, PaO2 was comparable between groups at 30 min after initiation of the Trendelenburg position and pneumoperitoneum, as well as throughout the study period. A possible explanation for the lack of difference in PaO2 between groups is that ERV improves oxygenation by increasing Pmean only when alveoli are recruitable. As Lee et al. presented in their study, PaO2 improved in patients with higher physiological dead space and better baseline gas exchange [13]. In addition, there is no further beneficial effect of increasing Pmean when total PEEP is constant and alveoli are sufficiently inflated [13,24]. Thus, when oxygenation was improved beyond the alveolar capacity, further improvement does not occur by increasing Pmean. This is supported by our results, which showed that PaO2 did not decrease during Trendelenburg positioning and CO2 pneumoperitoneum and was somewhat higher than values reported in previous studies with a similar design. Previously, Kim et al.[11] and Choi et al.[6] reported lower PaO2 levels during the Trendelenburg position and CO2 pneumoperitoneum than at 10 min after induction. In Kim et al's study [11], PaO2 was lowest (153-155 cm H2O) during the Trendelenburg position in the 1:2 I/E ratio group with a higher Ppeak and lower Pmean, whereas in our study, PaO2 was highest during the Trendelenburg position and CO2 pneumoperitoneum with a relatively lower Ppeak and higher Pmean. Although Ppeak might not accurately reflect alveolar pressure when the flow pattern is modified [25], Ppeak is clinically a major determinant of alveolar pressure [26], and is related to the barotrauma. In our current study, PCV with VG reduced Ppeak as much as ERV. Therefore, the increase in PaO2 observed in the current study suggests that both PCV with VG and ERV are sufficient to recruit alveoli and reduce Ppeak during the Trendelenburg position and CO2 pneumoperitoneum.
A major issue in ventilatory strategies during RALP is maintaining a physiological arterial CO2 tension without using high airway pressures. During pneumoperitoneum, it may be difficult to continue to increase minute volume by increasing tidal volume or respiratory rate in response to an elevated ETCO2, as these maneuvers may causes lung hyperinflation or barotrauma [9,27]. Strikingly, no patients in the current study exhibited a Ppeak greater than 40 cm H2O and all patients maintained an ETCO2 between 35 and 45 mmHg during surgery. Although PaCO2 increased after the supine position was resumed at the end of surgery in the ERV group, the pH remained in the normal range and no clinical effects were observed. Together, our findings of improved oxygenation and maintained normocapnia without increased airway pressures suggest that both PCV with VG and ERV might be useful ventilator modalities during RALP.
Patients undergoing RALP are usually elderly, with multiple coexisting diseases and reduced cardiovascular reserve. These patients are vulnerable to hemodynamic changes, and even small changes in cardiac output may result in substantial hemodynamic effects. Thus, ventilatory strategies to minimize impairment of cardiac function are necessary. As mentioned previously, increases in Pmean during ERV improve oxygenation, but reduce venous return and cardiac output by increasing intrathracic pressure [24]. Although no cardiovascular collapse were noted in our patients, our findings suggest that PCV with VG may be a more clinically appropriate and easier mode of ventilation than ERV—especially for patients with cardiopulmonary disease—because PCV with VG maintains oxygenation effectively as ERV, without increasing Pmean.
Taken together, results of our study revealed that PCV with VG was similar to ERV in maintaining oxygenation with lower Pmean during RALP. These results are consistent with the previous studies that compared PCV with VG to PCV or VCV in that PCV with VG lowered Ppeak or Pmean while maintaining similar oxygenation [16-18]. In other words, use of PCV with VG provides tight control on tidal volume and adequate oxygenation with a better compromise towards peak inspiratory pressure [28]. Therefore, it is concluded that the use of PCV with VG might be helpful in patients vulnerable to changes in airway pressure and indicated for ERV. In particular, PCV with VG might be suitable for patients with underlying diseases such as COPD and patients undergoing laparoscopic surgery or one lung ventilation, without concerns of hemodynamic instability or possibility of autoPEEP.
This study has several limitations. First, duration of PCV with VG or ERV were as short as 60 minutes because duration of Trendelenburg position was about 60 minutes. Considering that alveolar recruitment does not occur immediately after application of a specific ventilator mode and oxygenation improvement may be time dependent [24], a longer operative time may have produced different results. Second, our study did not include patients with respiratory disease or obesity, both of which are important factors for compromising oxygenation and respiratory mechanics. Third, patients were ventilated without the use of extrinsic PEEP, and we could not measure auto-PEEP during surgery, because measurement of auto-PEEP requires an end-expiratory hold [29].
In conclusion, during RALP, PCV with VG is an acceptable alternative ventilatory strategy to ERV for achieving similar levels of oxygenation. Indeed, oxygenation improved with both types of ventilation, suggesting that both ventilatory methods are suitable for RALP. However, PCV with VG produced lower Pmean values, suggesting that it may be more useful than ERV in patients with reduced cardiovascular function. Regardless of ventilation mode, careful monitoring is necessary to maintain adequate oxygenation, ventilation, and airway pressures during the Trendelenburg position and CO2 pneumoperitoneum phase of RALP.
Acknowledgements
Assistance with the study: The authors would like to thank Dr. Young Deuk Choi in the Department of Urology at the Yonsei university college of medicine for his helpful advice for this manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Menon M, Shrivastava A, Tewari A. Laparoscopic radical prostatectomy: conventional and robotic. Urology. 2005;66(5 Suppl):101-4
2. Hu JC, Gu X, Lipsitz SR, Barry MJ, D'Amico AV, Weinberg AC. et al. Comparative effectiveness of minimally invasive vs open radical prostatectomy. JAMA. 2009;302(14):1557-64
3. Falabella A, Moore-Jeffries E, Sullivan MJ, Nelson R, Lew M. Cardiac function during steep Trendelenburg position and CO2 pneumoperitoneum for robotic-assisted prostatectomy: a trans-oesophageal Doppler probe study. Int J Med Robot. 2007;3(4):312-5
4. Kalmar AF, Foubert L, Hendrickx JF, Mottrie A, Absalom A, Mortier EP. et al. Influence of steep Trendelenburg position and CO2 pneumoperitoneum on cardiovascular, cerebrovascular, and respiratory homeostasis during robotic prostatectomy. British journal of anaesthesia. 2010;104(4):433-9
5. Lestar M, Gunnarsson L, Lagerstrand L, Wiklund P, Odeberg-Wernerman S. Hemodynamic perturbations during robot-assisted laparoscopic radical prostatectomy in 45 degrees Trendelenburg position. Anesth Analg. 2011;113(5):1069-75
6. Choi EM, Na S, Choi SH, An J, Rha KH, Oh YJ. Comparison of volume-controlled and pressure-controlled ventilation in steep Trendelenburg position for robot-assisted laparoscopic radical prostatectomy. J Clin Anesth. 2011;23(3):183-8
7. Andersson LE, Baath M, Thorne A, Aspelin P, Odeberg-Wernerman S. Effect of carbon dioxide pneumoperitoneum on development of atelectasis during anesthesia, examined by spiral computed tomography. Anesthesiology. 2005;102(2):293-9
8. Ogurlu M, Kucuk M, Bilgin F, Sizlan A, Yanarates O, Eksert S. et al. Pressure-controlled vs volume-controlled ventilation during laparoscopic gynecologic surgery. J Minim Invasive Gynecol. 2010;17(3):295-300
9. Kim WH, Hahm TS, Kim JA, Sim WS, Choi DH, Lee EK. et al. Prolonged inspiratory time produces better gas exchange in patients undergoing laparoscopic surgery: A randomised trial. Acta Anaesthesiol Scand. 2013;57(5):613-22
10. Kim SH, Choi YS, Lee JG, Park IH, Oh YJ. Effects of a 1:1 inspiratory to expiratory ratio on respiratory mechanics and oxygenation during one-lung ventilation in the lateral decubitus position. Anaesth Intensive Care. 2012;40(6):1016-22
11. Kim MS, Kim NY, Lee KY, Choi YD, Hong JH, Bai SJ. The impact of two different inspiratory to expiratory ratios (1:1 and 1:2) on respiratory mechanics and oxygenation during volume-controlled ventilation in robot-assisted laparoscopic radical prostatectomy: a randomized controlled trial. Can J Anaesth. 2015;62(9):979-87
12. Park JH, Lee JS, Lee JH, Shin S, Min NH, Kim MS. Effect of the Prolonged Inspiratory to Expiratory Ratio on Oxygenation and Respiratory Mechanics During Surgical Procedures. Medicine (Baltimore). 2016;95(13):e3269
13. Lee SM, Kim WH, Ahn HJ, Kim JA, Yang MK, Lee CH. et al. The effects of prolonged inspiratory time during one-lung ventilation: a randomised controlled trial. Anaesthesia. 2013;68(9):908-16
14. Brower RG, Matthay MA, Morris A, Schoenfeld D, Thompson BT, Wheeler A. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. New England Journal of Medicine. 2000;342(18):1301-8
15. Kim H. Protective strategies for one-lung ventilation. Korean journal of anesthesiology. 2014;67(4):233-4
16. Dion JM, McKee C, Tobias JD, Sohner P, Herz D, Teich S. et al. Ventilation during laparoscopic-assisted bariatric surgery: volume-controlled, pressure-controlled or volume-guaranteed pressure-regulated modes. Int J Clin Exp Med. 2014;7(8):2242-7
17. Song SY, Jung JY, Cho MS, Kim JH, Ryu TH, Kim BI. Volume-controlled versus pressure-controlled ventilation-volume guaranteed mode during one-lung ventilation. Korean J Anesthesiol. 2014;67(4):258-63
18. Pu J, Liu Z, Yang L, Wang Y, Jiang J. Applications of pressure control ventilation volume guaranteed during one-lung ventilation in thoracic surgery. Int J Clin Exp Med. 2014;7(4):1094-8
19. Marcy TW, Marini JJ. Inverse ratio ventilation in ARDS. Rationale and implementation. Chest. 1991;100(2):494-504
20. Zavala E, Ferrer M, Polese G, Masclans JR, Planas M, Milic-Emili J. et al. Effect of inverse I:E ratio ventilation on pulmonary gas exchange in acute respiratory distress syndrome. Anesthesiology. 1998;88(1):35-42
21. Yanos J, Watling SM, Verhey J. The physiologic effects of inverse ratio ventilation. Chest. 1998;114(3):834-8
22. Lee K, Oh YJ, Choi YS, Kim SH. Effects of a 1:1 inspiratory to expiratory ratio on respiratory mechanics and oxygenation during one-lung ventilation in patients with low diffusion capacity of lung for carbon monoxide: a crossover study. J Clin Anesth. 2015;27(6):445-50
23. Marini JJ, Ravenscraft SA. Mean airway pressure: physiologic determinants and clinical importance-Part 2: Clinical implications. Crit Care Med. 1992;20(11):1604-16
24. Lessard MR, Guerot E, Lorino H, Lemaire F, Brochard L. Effects of pressure-controlled with different I:E ratios versus volume-controlled ventilation on respiratory mechanics, gas exchange, and hemodynamics in patients with adult respiratory distress syndrome. Anesthesiology. 1994;80(5):983-91
25. Milic-Emili J, Tantucci C, Chassé M, Corbeil C. Introduction with special reference to Ventilator-associated Barotrauma. Pulmonary Function in Mechanically Ventilated Patients: Springer. 1991:1-8
26. Kilpatrick B, Slinger P. Lung protective strategies in anaesthesia. Br J Anaesth. 2010;105(Suppl 1):i108-16
27. Park EY, Koo BN, Min KT, Nam SH. The effect of pneumoperitoneum in the steep Trendelenburg position on cerebral oxygenation. Acta Anaesthesiol Scand. 2009;53(7):895-9
28. Ball L, Dameri M, Pelosi P. Modes of mechanical ventilation for the operating room. Best Pract Res Clin Anaesthesiol. 2015;29(3):285-99
29. Mughal MM, Culver DA, Minai OA, Arroliga AC. Auto-positive end-expiratory pressure: mechanisms and treatment. Cleve Clin J Med. 2005;72(9):801-9
Author contact
Corresponding author: Jin Ha Park, MD. PhD. Department of Anesthesiology and Pain Medicine and Anesthesia and Pain Research Institute, Yonsei University College of Medicine, 50 Yonsei-ro, Seodaemun-gu, Seoul, 03722, Republic of Korea. Phone: 82-2-2228-2420; Fax: 82-2-312-7185; E-mail: realsummerac

 Global reach, higher impact
Global reach, higher impact