Impact Factor ISSN: 1449-1907
Int J Med Sci 2014; 11(6):593-601. doi:10.7150/ijms.8096 This issue Cite
Research Paper
King Cobra (Ophiophagus hannah) Venom L-Amino Acid Oxidase Induces Apoptosis in PC-3 Cells and Suppresses PC-3 Solid Tumor Growth in a Tumor Xenograft Mouse Model
1. CENAR and Department of Molecular Medicine;
2. Department of Pharmacology;
3. Department of Pathology;
4. Department of Physiology; Faculty of Medicine, University of Malaya, 50603 Kuala Lumpur, Malaysia.
Received 2013-11-10; Accepted 2014-2-27; Published 2014-4-8
Abstract
King cobra (Ophiophagus hannah) venom L-amino acid oxidase (OH-LAAO), a heat stable enzyme, has been shown to exhibit very potent anti-proliferative activity against human breast and lung tumorigenic cells but not in their non-tumorigenic counterparts. We further examine its in vitro and in vivo anti-tumor activity in a human prostate adenocarcinoma (PC-3) model. OH-LAAO demonstrated potent cytotoxicity against PC-3 cells with IC50 of 0.05 µg/mL after 72 h incubation in vitro. It induced apoptosis as evidenced with an increase in caspase-3/7 cleavages and an increase in annexin V-stained cells. To examine its in vivo anti-tumor activity, we treated PC-3 tumor xenograft implanted subcutaneously in immunodeficient NU/NU (nude) mice with 1 µg/g OH-LAAO given intraperitoneally (i.p.). After 8 weeks of treatment, OH-LAAO treated PC-3 tumors were markedly inhibited, when compared to the control group (P <0.05). TUNEL staining analysis on the tumor sections showed a significantly increase of apoptotic cells in the LAAO-treated animals. Histological examinations of the vital organs in these two groups showed no significant differences with normal tissues, indicating no obvious tissue damage. The treatment also did not cause any significant changes on the body weight of the mice during the duration of the study. These observations suggest that OH-LAAO cytotoxic effects may be specific to tumor xenografts and less to normal organs. Given its potent anti-tumor activities shown in vitro as well as in vivo, the king cobra venom LAAO can potentially be developed to treat prostate cancer and other solid tumors.
Keywords: Ophiophagus hannah, L-amino acid oxidase, anti-proliferative, anti-PC-3 tumor, NU/NU mice.
Introduction
Prostate cancer is the second leading cause of cancer deaths after lung cancer worldwide [1]. Similar to other cancers, prostate cancer arise from prostate epithelial cells which display uncontrollable growth (beyond the normal limits), invasion, and metastasis capabilities [2]. Current chemotherapy regimen for prostate cancer usually involves the use of substances toxic to normal cells and hence the treatment is usually accompanied by a range of side effects such as vomiting, nausea and alopecia. Thus, there is continual need to search for potential natural-based cancer therapeutic agents that exhibit highly selective cytotoxicity against cancer cells over the normal cells.
Snake venom L-amino acid oxidase (LAAO, L-amino acid: O2 oxidoreductase, EC 1.4.3.2) has been reported to exhibit a wide range of pharmacological activities including anti-proliferative [3] and anti-bacterial activities [4]. It catalyzes oxidative deamination of L-amino acid to produce the corresponding α-keto acid, hydrogen peroxide and ammonia [5,6]. The biological activities are primarily caused by the hydrogen peroxide produced during the oxidative deamination and the cytotoxic action was believed to exert via apoptosis [3]. Unlike other venom LAAOs, king cobra (Ophiophagus hannah) venom LAAO, with a molecular weight of 130 kDa, has an unusual thermal stability. At pH 7.4, the enzyme retained 100% activity after incubation at 37 °C for 5 days. Unlike other snake venom LAAO, king cobra venom LAAO was stable at alkaline condition and was not inactivated by freezing [7]. Because of these favourable stability properties, king cobra venom LAAO has a greater potential, when compared to other venom LAAOs, to be developed as a cancer therapeutic agent. Earlier studies also showed that the king cobra venom enzyme exhibited extremely potent anti-proliferative activities in comparison with other venom LAAOs, and that its cytotoxicity was selective against tumorigenic cells [3].
Here we examined the enzyme's anti-tumor effect in a PC-3 tumor xenograft mouse model implanted in immunodeficient NU/NU mice, and evaluated its toxicity on vital organs. As yet, there are very few studies that demonstrated the in vivo anti-cancer action of snake venom LAAOs on tumors [8]. Human prostate adenocarcinoma (PC-3) cell line was used in this study because of its irresponsiveness to androgens, glucocorticoids, or epidermal or fibroblast growth factors [9].
Materials and methods
Ethics statement
All animal handling and procedures were performed according to the CIOMS guidelines [10]. The use of animals in this study was approved by the Animal Care and Use Committee (ACUC) of the University of Malaya (Ethical clearance letter No. 2013-06-07/MOL/R/FSY).
Animals
Four-week-old, male NU/NU mice (18-25 g) were purchased from BioLASCO, Taiwan (Certificate no: VP302S10023105). All mice were housed under specific pathogen free (SPF) conditions (3 mice per cage) and were fed with sterilized standard pellets and water ad libitum. To allow sufficient adaption to the new environment, these mice were housed for a week prior to the start of experiments.
Cell culture
Human prostate adenocarcinoma (PC-3) cell line was purchased from American Type Culture Collection (ATCC) (USA). The cells were grown in RPMI-1640 (Biowest, France) media supplemented with 10% foetal bovine serum (Sigma, USA). Cells were cultured in a humidified 37 °C incubator with 5% CO2 atmosphere.
Isolation of L-amino acid oxidase from king cobra venom and assay of enzyme activity
The venom, which was milked from several adult Malaysian king cobras, was obtained from Snake Valley (Seremban, Malaysia). The snakes were identified by one of the authors (N.H. Tan). King cobra snake venom LAAO was purified using a single-step Resource Q high performance liquid chromatography as described previously [4]. L-amino acid oxidase activity was determined according to Bergmeyer [11], using L-leucine as substrate, as described in Lee et al. [4]. Protein concentration was determined by the Bradford method [12] and the specific activity of the purified OH-LAAO obtained was 437.7 μmol/min/mg [4].
Cell viability assay
The cytotoxic effect of king cobra venom LAAO and the IC50 values were determined using MTT (3,(4,5-dimethythiazol-2-yl)-2,3-diphenyl tetrazolium bromide) method according to Ahn et al. [13] with modifications [3].
Caspase-3/7 and PE-Annexin V/7-AAD assays
Caspase-3/7 activity was measured using Caspase-Glo® 3/7 assay kit (Promega, USA) according to the manufacturer's protocol as described previously [3]. Human prostate adenocarcinoma (PC-3) cells were seeded into white-walled 96-well plates and treated with LAAO at its IC50. The luminescence of each sample was measured using Glomax-Multi detection system (Promega, USA). PE-Annexin V/7-AAD assay was performed using PE annexin V apoptosis detection kit I (BD Biosciences, USA) according to the manufacturer's instructions and as described in Lee at al. [3]. The cells were treated with 0.05 µg/mL of LAAO (IC50 of PC-3) for 72 h. Data analysis were performed using FCS express 4 flow software (De Novo software, CA) and results were expressed in density plots and plotted as 7-AAD versus PE-Annexin V.
PC-3 tumor xenograft mouse model
Prostate adenocarcinoma cells (PC-3) were grown to 80% - 90% confluency before harvested using trypsin-EDTA (Sigma, USA). The cells were washed twice with PBS followed by centrifugation. The cell pellet was re-suspended in PBS and the number of viable cells was determined using trypan blue exclusion method and counted with a haemocytometer. The cell suspension was then mixed with an equal volume of Matrigel TM (BD Biosciences, USA). Each mouse was inoculated subcutaneously (s.c) with 200 µL of the cells-MatrigelTM mixture which contained approximately 0.6 x 107cells. Mice were examined daily for tumor growth at the injection site. Tumor volumes (mm3) were measured weekly using a digital calliper and were calculated as length x width x height x π/6, to approximate the volume of a spheroid. When the tumor volume had reached about 100 - 200 mm3 (usually 1 - 2 weeks after injection of the PC-3 cells), mice were randomly assigned into two groups (n = 6/group). Mice were treated i.p daily for 8 weeks with either vehicle (PBS) or LAAO at a dose of 1 µg/g/day/200 µL. The tumor volume and body weight of the individual animals were monitored and recorded weekly over the 8-week treatment period.
Toxicity study of LAAO in tumor-bearing mice
Mice from both treated and control group were sacrificed by cervical dislocation under anesthesia at the end of the 8 weeks study. Vital organs (lung, liver, heart, spleen, kidneys) and solid tumor tissues were harvested and fixed in 10% buffered formalin for 48 h. The tissues were dehydrated by ethanol and cleared with xylene followed by paraffin embedding and sectioning processes. Histological sections were stained with hematoxylin and eosin and examined using light microscopy.
TdT-mediated dUTP nicked-end labelling (TUNEL) immunohistochemical staining
Terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nicked-end labelling (TUNEL) for the detection of apoptotic cells were performed using ApopTag® plus peroxidase in situ apoptosis detection kit (Millipore, USA) according to the manufacturer's protocols. Briefly, 5 µm thin tumor sections mounted on the silanized slides were dewaxed, washed with PBS and pre-treated with proteinase K (20 mg/mL) (Millipore, USA) for 15 min. Sections were then quenched in 3% hydrogen peroxide for 5 min and an adequate amount of the equilibration buffer was applied onto each section and incubated for 10 sec. Subsequently, tumor sections were incubated with TdT enzyme at 37 °C for 1 h in a humidified chamber. After labelling, sections were washed and allowed to bind to anti-digoxigenin conjugate in a humidified chamber for 30 min. Slides were then stained with 3, 3'-diaminobenzidine (DAB) peroxidase substrate for 5 min, counter stained with 0.5% methyl green for 10 min and finally mounted under the coverslips. Slides were examined under light microscope and the pictures of 5 random fields of view per section at 200X magnification were taken. The percentage of apoptotic cells were scored as an average of the ratio of TUNEL-positive cells to the total number of cells presence in each field.
Statistical analysis
Results for cytotoxicity, caspase-3/7 and PE-annexin V/7-AAD were expressed as mean ± S.D. The in vivo anti-tumor activity was expressed as mean ± S.E.M. The significance of the differences of the means was determined by one way ANOVA, followed by Tukey's post hoc multiple comparison test. The statistical analysis was conducted using SPSS 17.0 (SPSS Inc., Chicago, IL, USA).
Results
Cytotoxicity of king cobra venom LAAO (OH-LAAO) against PC-3 cells
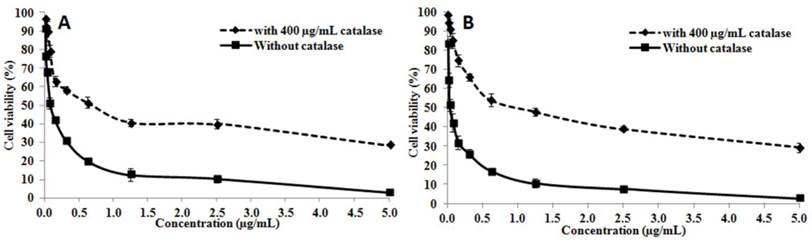
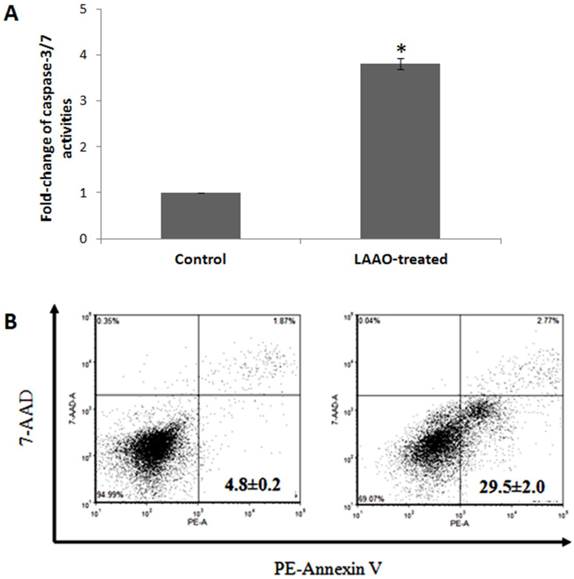
The cytotoxic activity of OH-LAAO on PC-3 cell line was dose-dependent (Figure 1). The cell kill curves were similar between 24 h and 72 h, yielding IC50 values 0.08 ± 0.02 and 0.05 ± 0.01 µg/mL, respectively. LAAO-treated cells also demonstrated a 3.8-fold increase in caspase-3/7 activity, when compared with vehicle-treated cells (Figure 2A). Low levels of caspase-3/7 activity was also detected in the control cells, which was presumably due to the small amount of apoptotic cells present in the growing cell population. PE-annexin V/7-AAD analysis also showed greater number of apoptotic cells by OH-LAAO treatment (29.5%) compared to vehicle group (4.8%) (Figure 2B). These data suggest that LAAO-mediated cell death in PC-3 cells is via induction of apoptosis
The cytotoxic actions of king cobra venom LAAO (OH-LAAO) on PC-3 cells. The PC-3 cells were treated with different concentrations of OH-LAAO (0.01 - 5.00 µg/mL) for 24 h (A) or 72 h (B), with or without catalase (400 µg/mL). The viability of the cells was measured by MTT assay. Results given are mean ± S.D (n = 3). Solid line —■—: without catalase; Broken line ---♦---: with catalase.
Apoptotic effects of king cobra venom LAAO (OH-LAAO) on PC-3 cells. A) Caspase-3/7 activities of OH-LAAO treated cells. The PC-3 cells were treated with OH-LAAO at IC50 dosage (0.05 µg/mL) for 72h. Caspase-3/7 activities were determined using Caspase-Glo 3/7 assay kit (n= 3). The fold change of the control is considered as 1 and the error bars indicate standard deviations. * P < 0.05 in the LAAO-treated versus control cells. B) PE-Annexin V/7-AAD analysis of the effect of OH-LAAO treatment on the PC-3 cells. The cells were treated with 0.05 µg/mL of OH-LAAO for 72 h and were then stained with Annexin V conjugated to Phycoerythrin (PE) and 7-Amino-Actinomycin D (7-AAD) prior to the FACS analysis. Cells (10,000 events) were analyzed by fluorescent-activated cell scanner. The lower left quadrant contains the viable (Annexin V negative; 7-AAD negative) population, the upper left quadrant contains the damage (Annexin V negative; 7-AAD positive) population, the upper right quadrant contains the late apoptotic (Annexin V positive; 7-AAD positive) cells and the lower right quadrant contains the early apoptotic (Annexin V positive; 7-AAD negative) cells. Left: PC-3 cells control; Right: OH-LAAO treated PC-3 cells. Total apoptotic cells (%) are shown in each figure. Figures are representatives of duplicate analysis from 2-independent experiments.
Anti-tumor effects of OH-LAAO in PC-3 tumor xenograft mouse model
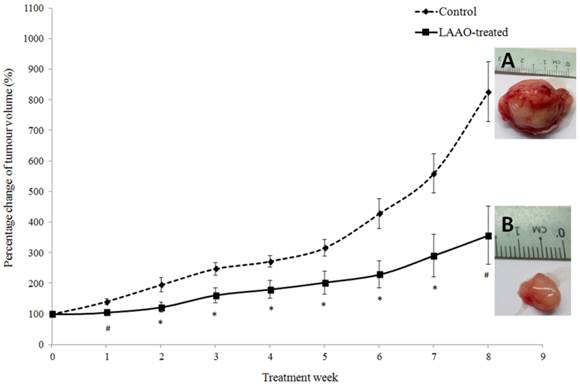
PC-3 tumor-bearing nude mice were treated with 1 µg/g/day of OH-LAAO for 8 weeks. As shown in Figure 3, LAAO-treated tumors grew markedly slower than those treated with vehicle. A significant difference in mean tumor size was seen between vehicle-treated and LAAO-treated group (P <0.01), as early as 7 days post treatment. By the 8th week, tumors in control mice had grown to greater than 700% of the original size (size of the tumor at the start of treatment = 100%), whereas for the treated mice, the average tumor size was 300% of their original size (P <0.01, between treated and control). All tumors were harvested at the end of the study (week 8). Interestingly, one of the tumors achieved complete regression with no sign of tissue left for harvest.
Anti-tumor effect of OH-LAAO on solid PC-3 prostrate tumor in nude mice. PC-3 tumor-bearing mice were treated i.p. daily with either vehicle (control, broken line---♦---, n = 6) or OH-LAAO (1 µg/g, solid line —■—, n = 6). Tumors were measured weekly, and the percentage of tumor volume was shown after comparison with those at week 0. Data was expressed as mean ± SEM. (P <0.05 for data points marked with *, P <0.01 for data points marked with #). Insert A: Representative tumor image of the control group. Insert B: Representative tumor image of the LAAO-treated group.
Toxicity study of OH-LAAO in tumor-bearing mice
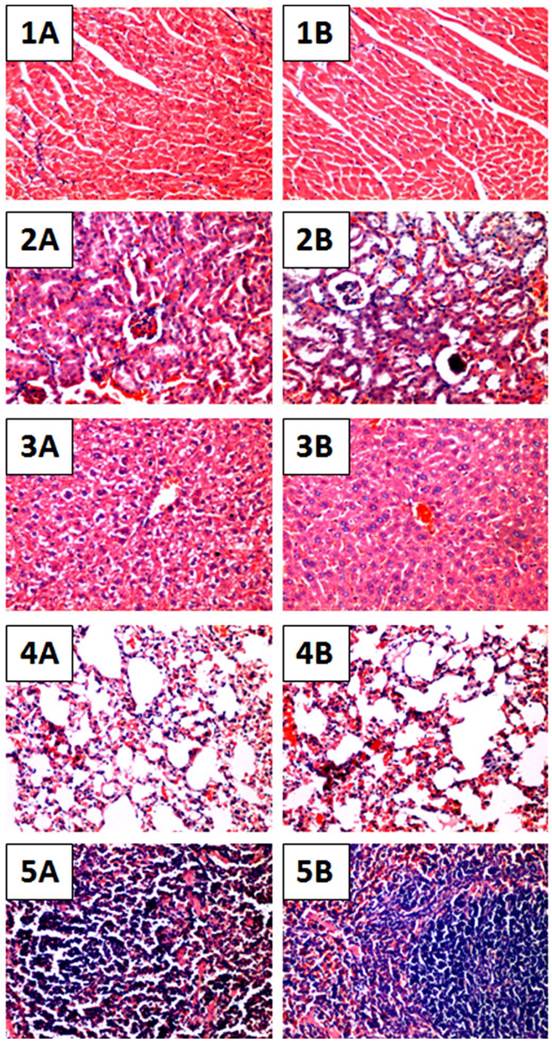
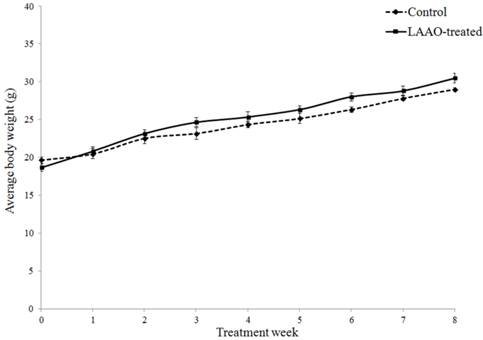
To further evaluate whether daily treatment of OH-LAAO in tumor-bearing nude mice for 8 weeks affected the vital organs, we performed detailed histological examinations on the organs harvested at the end of study. Heart, kidney, liver, lung and spleen in LAAO-treated mice did not show any abnormal pathological changes when compared to those in vehicle-treated mice (Figure 4). Also, the body weight of the LAAO-treated group did not differ (P >0.05) from the vehicle-treated mice throughout the treatment period (Figure 5).
Histological sections of major organs of OH-LAAO treated mice. Hematoxylin and eosin stained sections of heart, kidney, liver, lung and spleen tissues of vehicle-treated mice (1A - 5A) as well as mice treated with i.p. administration of 1 µg/g of OH-LAAO daily for 8 weeks (1B - 5B). The treatment did not reveal significant pathological changes. Images were shown at 100X magnification.
OH-LAAO treatment did not affect body weight of mice. Changes in body weight of the mice in OH-LAAO-treated group and control group were determined weekly. There was no significant differences in the average body weight of the two groups over the 8 weeks treatment period (P > 0.05). Solid line —■—: Treated mice; Broken line ---♦---: Control mice. Data was expressed as mean ± SEM.
OH-LAAO induced apoptosis in PC-3 tumor cells
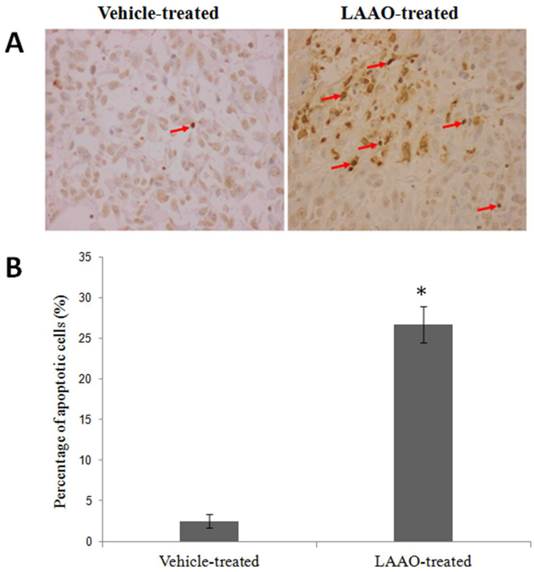
To determine whether LAAO-mediated tumor size reduction is due to induction of apoptosis, we examined the DNA fragmentation in these tumors using TUNEL assay. Representative tumor sections stained with TUNEL are shown in Figure 6A. Significant greater apoptotic cells were observed in LAAO-treated tumors (26.7%) than in vehicle-treated tumors (2.5%) (Figure 6B).
Apoptotic effect of OH-LAAO in PC-3 solid tumor.(A) Representative TUNEL stained of tumor sections harvested from tumor-bearing nude mice treated with vehicle (PBS, the controls) and OH-LAAO, at 200X magnifications. Apoptosis were evidenced in cells with dark brown-stained nuclei. (B) Percentage of TUNEL-positive cells in tumor sections from the vehicle- (n = 6) and LAAO-treated mice (n = 5). One of the tumors from the LAAO-treated group achieved complete regression, with no tumor tissue left after 8-weeks treatment. Data was expressed as mean ± SEM. * P < 0.05 in the LAAO-treated versus control group.
Discussion
We previously showed that king cobra venom LAAO exerts potent cytotoxicity against breast cancer and lung cancer cells [3]. In this study, we further showed that OH-LAAO is similarly potent in inhibiting the growth of human prostate cancer cells, PC-3, in vitro and in vivo. Its cytotoxicity is due to induction of apoptosis, as in agreement with previous reports [14-20]. In vivo studies using PC-3 tumor xenograft in nude mice showed that 8 weeks of OH-LAAO treatment at a dose of 1 μg/g (i.p.) markedly inhibited the growth of the solid tumor.
The dose of 1 μg/g OH-LAAO was chosen for experimental treatment as earlier work reported that the enzyme has an LD50 of 5.0 µg/g [7]. The 8-week OH-LAAO treatment at this dose indeed did not cause any significant differences in the body weight between the treated and control mice, nor any significant pathological abnormalities in the major organs of the treated animals. These observations, together with the high IC50 of OH-LAAO determined in non-tumorigenic breast and lung cells as reported earlier [3], strongly suggest that this enzyme is relatively non-toxic to normal tissues. The selective cytotoxicity of OH-LAAO could be due to the differences in the metabolism of the cells (tumor cells versus non-tumorigenic cells) or the selective binding of the enzyme to tumorigenic cells. Wei et al. [21] reported that at dosage up to 50 µg Bungarus faciastus LAAO did not induce significant organ damage including lung, heart, kidneys, liver and spleen. Bregge-Silva et al. [22] reported that 100 µg of Lachesis muta LAAO did not cause morphological changes in the heart, lung and kidney tissues. However, Wei et al. [23] reported that 50 µg of Agkistrodon blomhoffii ussurensis LAAO caused severe lung lesions after 6 and 24 h injections, indicating that LAAOs from different venoms may differ in their toxicity.
The detailed mechanism of cytotoxic action of snake venom LAAO on cancer cells has not been established. A possibility is that the injected OH-LAAO acts by binding to the tumor cell surface and generates a highly localized (and hence concentrated) H2O2. The H2O2 may have direct cytotoxic action or may act via the oxidative modifications of signalling proteins. The dose of OH-LAAO chosen in the in vivo anti-tumor studies (1 µg/g body weight) was estimated to give a theoretical level of LAAO of about 5 µg/mL in the extracellular fluid (ECF) of the mice, assuming that ECF was 20% of body weight [24], if all the injected LAAO were absorbed and distributed throughout the entire ECF compartment instantaneously. This level would far exceed the IC50 dose of PC-3 cells in vitro. However, it should be noted that absorption of LAAO injected is not likely to be instantaneous, and not all injected LAAO would be absorbed. Further, a certain quantity of LAAO may be degraded or excreted before it reaches the target cells. Also, the rate and ability of the LAAO to permeate the capillary wall and penetrate to the tumor cells are likely to be limited. Thus, the exact level of LAAO that the tumor cells in the treated mice are exposed to is likely to be much lower than 5 µg/mL. Nevertheless, at this dose of treatment (1 µg/g body weight) the LAAO in the animals was shown to be able to inhibit growth of the tumor effectively, with no visible deleterious effect on the major organs or reducing the body weight of the animals.
It is well established that many anticancer drugs cannot penetrate solid tumor tissue efficiently to reach the target cancer cells due to the tumor microenvironment [25] and this is particularly likely also for a large molecules such as snake venom LAAOs (with molecular weight of 130 kDa), which possess low diffusion coefficient. In our studies, we demonstrated that despite the large molecular size, king cobra venom LAAO could effectively inhibit solid tumor growth in vivo. Compared to the control, treatment with OH-LAAO caused 39% reduction on PC-3 tumor size in the mice (P <0.01) after just one week of treatment. While substantial individual variation in anti-tumor effect was observed in the LAAO-treated mice during the 8-week treatment. Our data demonstrate that king cobra venom LAAO could indeed effectively inhibit tumor growth in vivo. The ability of king cobra venom LAAO to effectively inhibit the tumor growth is presumably due to its very high cytotoxicity towards PC-3 cells (IC50 = 0.05 µg/mL, 72 h incubation). Earlier, Zhang and Wu [8] reported that 10 days treatment with 1.5 µg/g of Agkistrodon acutus venom LAAO inhibited tumor growth of Hepatoma 22, Sarcoma 180 and Ehrlich ascites carcinoma tumors (established in Kun Ming mice) by 21.6%, 12.9% and 14.2%, respectively. However, it is not known whether the mice used were immunocompromised and hence the validity of results difficult to assess. In this study, we used immunocompromised mice, and the king cobra venom LAAO yielded greater level of growth inhibition, including one complete regression after 8 weeks of treatment.
In conclusion, the effectiveness of king cobra venom LAAO in suppressing the growth of solid tumors in vivo without causing much significant toxic effects supports its potential as an anti-cancer agent. However, its high molecular weight may hinder its penetration into the tumor; therefore further development in delivery system may be required. For example, magnetic nanoparticle-enzyme system may allow the use of lower amount of LAAO to target specifically the tumor area, while maintaining maximal cytotoxicity. This approach has been used in the application D-amino acid oxidase for cancer treatment [26]. In addition, the enzyme is probably more effective against cancer cell suspensions such as leukemias and ascites, than in solid tumors. Alternatively, the enzyme could be cloned and transfected to tumor cells to induce cell death in situ, an approach suggested by Sun et al. [17].
Acknowledgements
We thank Ms Kavita S. Subramaniam for her assistance in FACS analysis. This work was supported by UMRG 075/12BIO and PV 069/2011B from University of Malaya.
Competing Interests
The authors have declared that no competing interest exists.
References
1. American Cancer Society. Cancer Facts & Figures 2009. Atlanta: ACS. 2009:19-20
2. Smart RC. Chemical carcinogenesis and mutagenesis. In: (ed.) Hodgson E. A textbook of modern toxicology. New Jersey: John Wiley & Sons. 2010:237-264
3. Lee ML, Chung I, Fung SY, Kanthimathi MS, Tan NH. Anti-proliferative activity of king cobra (Ophiophagus Hannah) venom L-amino acid oxidase. Basic and Clinical Pharmacology and Toxicology. 2013 DOI: 10.1111/bcpt.12155
4. Lee ML, Tan NH, Fung SY, Shamala DS. Antibacterial action of a heat-stable form of L-amino acid oxidase isolated from king cobra (Ophiophagus hannah) venom. Comp Biochem Physiol C. 2011;153:237-242
5. Du XY, Clemetson KJ. Snake venom L-amino acid oxidases. Toxicon. 2002;40:659-665
6. Tan NH, Fung SY. Snake venom L-amino acid oxidase. Mackessy, S. (Ed.), Handbook of Venoms and Toxins of Reptiles. CRC Press, New York. 2009:219-232
7. Tan NH, Saifuddin MN. Isolation and characterization of an unusual form of Lamino acid oxidase from cobra (Ophiophagus hannah) venom. Biochem Int. 1989;19:937-944
8. Zhang L, Wu WT. Isolation and characterization of ACTX-6: a cytotoxic L-amino acid oxidase from Agkistrodon acutus snake venom. Natural Product Research. 2008;22:554-563
9. Kaighn ME, Narayan KS, Ohnuki Y, Lechner JF, Jones LW. Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Investigative Urology. 1979;17:16-23
10. Howard-Jones NA. A CIOMS ethical code for animal experimentation. WHO Chronicle. 1995;39:51-56
11. Bergmeyer HU. L-amino acid oxidase. Methods in Enzymatic Analysis, Vol 2. Verlag Chimie, Gmbh, Weinheim. 1983:149-151
12. Bradford MM. A rapid method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Ana Biochem. 1976;72:248-254
13. Ahn MY, Lee BM, Kim YS. Characterization and cytoxicity of L-amino acid oxidase from the venom of king cobra (Ophiophagus hannah). Int J Biochem Cell Biol. 1997;29:911-919
14. Alves RM, Antonucci GA, Paiva HH, Cintra AC, Franco JJ. et al. Evidence of caspase-mediated apoptosis induced by L-amino acid oxidase isolated from Bothrops atrox snake venom. Comparative Biochemistry and Physiology, Part A. 2008;151:542-550
15. Alves MR, Figueiredo RF, Antonucci GA, Paiva HH, Pires LB. et al. Cell cycle arrest evidence, parasiticidal and bactericidal properties induced by L-amino acid oxidase from Bothrops atrox snake venom. Biochemie. 2011;93:941-947
16. Samel M, Vija H, Rönnholm G, Siigur J, Kalkkinen N, Siigur E. Isolation and characterization of an apoptotic and platelet aggregation inhibiting L-amino acid oxidase from Vipera berus berus (common viper) venom. Biochim Biophys Acta. 2006;1764:707-714
17. Sun LK, Yoshii Y, Hyodo A, Tsurushima H, Saito A. et al. Apoptotic effect in the glioma cells induced by specific protein extracted from Okinawa Habu (Trimeresurus flavoviridis) venom in relation to oxidative stress. Toxicol in vitro. 2003;17:169-177
18. Stabeli RG, Sant'Ana CD, Ribeiro PH, Costa TR, Ticli FK. et al. Cytotoxic L-amino acid oxidase from Bothrops moojeni: biochemical and functional characterization. Intn J Biol Macromol. 2007;41:132-140
19. Zhang L, Cui L. A cytotoxin isolated from Agkistrodon acutus snake venom induces apoptosis via Fas pathway in A549 cells. Toxicol in vitro. 2007;21:1095-1103
20. Zhang L, Wei LJ. ACTX-8, a cytotoxic L-amino acid oxidase isolated from Agkistrodon acutus snake venom, induces apoptosis in Hela cervical cancer cells. Life Sci. 2007;80:1189-1197
21. Wei JF, Yang HW, Wei XL, Qiao LY, Wang WY, He SH. Purification, characterization and biological activities of the L-amino acid oxidase from Bungarus fasciatus snake venom. Toxicon. 2009;54:262-271
22. Bregge-Silva C, Nonato MC, de Albuquerque S, Ho PL, Junqueira de Azevedo IL. et al. Isolation and biochemical, functional and structural characterization of a novel L-amino acid oxidase from Lachesis muta snake venom. Toxicon. 2012;60:1263-1276
23. Wei XL, Wei JF, Li T, Qiao LY, Liu YL. et al. Purification, characterization and potent lung lesion activity of an L-amino acid oxidase from Agkistrodon blomhoffii ussurensis snake venom. Toxicon. 2007;50:1126-1139
24. Chapman ME, Hu L, Plato CF, Kohan DE. Bioimpedance spectroscopy for the estimation of body fluid volumes in mice. Am J Physiol Renal Physiol. 2010;299(F):280-283
25. Minchinton AI, Tannock IF. Drug penetration in tumors: mechanisms, role in drug resistance, and methods for modifications. Nat Rev Cancer. 2006;6:583-592
26. Bava A, Gornati R, Cappellini F, Caldinelli L, Pollegioni L. and Bernardini, G. D- amino acid oxidase-nanoparticle system: a potential novel approach for cancer enzymatic therapy. Nanomedicine. 2013;8:1797-1806
Author contact
Corresponding author: Nget Hong Tan, Department of Molecular Medicine, Faculty of Medicine, University of Malaya, Kuala Lumpur, Malaysia. Fax: 603-79674957 Tel: 603-79674912 E-mail address: tanngethongcom.sg.

 Global reach, higher impact
Global reach, higher impact