Impact Factor ISSN: 1449-1907
Int J Med Sci 2012; 9(9):743-747. doi:10.7150/ijms.4862 This issue Cite
Research Paper
A Two-Step Control of Secondary Splenic Pedicles Using Ligasure during Laparoscopic Splenectomy
Department of Hepatobiliary and Pancreatic Surgery, the First Bethune Hospital, Jilin University, Jilin 130021, China.
Received 2012-7-12; Accepted 2012-10-15; Published 2012-10-18
Abstract
Objective: We modified the LigaSure vessel sealing into a two-step technique without using Endo-GIA stapler for the secondary splenic pedicle control in laparoscopic splenectomy (LS). This study evaluated the efficacy and safety outcomes of this technique.
Methods: Patients (n = 105) scheduled for elective LS were consecutively and prospectively enrolled, including 24 males and 81 females, with a mean age of 43.6 (range 11-75) years. Following the mobilization of the spleen, the splenic inflow was interrupted by applying a Hem-o-lock clip. LigaSure was used to seal and transect the secondary splenic pedicles adjacent to the pancreatic tail and subsequently in proximity to the spleen.
Results: Of 105 patients, 103 patients (98.1%) underwent successful LS, whereas two patients (1.9%) required the conversion to laparotomy. The mean operative time was 100 min, whilst the mean volume of blood loss was 500 mL. No clinically significant morbidities or mortality occurred following LS. An average of 8,000 RMB (range: 6900 to 9000; 1 USD = 6.5 RMB) was saved by using this two-step technique.
Conclusion: Secondary splenic pedicles can be successfully controlled in LS by using a two-step technique with the LigaSure vessel sealing system in an economically favorable way.
Keywords: Laparoscopic splenectomy, Secondary splenic pedicle, LigaSure vessel sealing system, Endo-GIA.
Introduction
Since Delaitre et al. 1 reported the first laparoscopic splenectomy (LS) in 1992, it has become the gold standard for the treatment of hematologic diseases of the spleen 2. Although it requires great technical care, the efficacy and safety of LS has justified its use, especially in less favorable cases such as splenomegaly and malignant tumors. The advantages of LS include less procedural pain, rapid postoperative recovery, and short hospital stay owing to its minimal invasiveness 3. LS is also thought to be financially preferable to open splenectomy 4.
The major procedural risk associated with LS is uncontrollable intraoperative bleeding requiring conversion to laparotomy5. Fundamental to LS is the division and management of the hilar vessels, which is frequently challenging in cases that involve splenic pedicles or main branches adherent to the pancreatic tail 6. For the hemostasis of splenic pedicels, the use of Endo-GIATM Universal (Endo-GIA) endoscopic vascular linear staplers prevails in current laparoscopic practice worldwide 7. However, the application of Endo-GIA staplers may subject patients to a higher risk of blood loss, conversion to laparotomy, and pancreatic injuries, as well as a greater financial burden compared to conventional ligation method, which compromise the otherwise intrinsic superiorities of LS8. The LigaSure vessel sealing system is a new surgical method that permanently fuses vessels ≤ 7 mm in diameter, by combining the effects of energy and pressure in a feedback-controlled response 9, 10. The feasibility, effectiveness, and safety of LigaSure in the sealing and dissection of splenic pedicels in LS has been investigated in multiple studies, including a comparative study with Endo-GIA11.
In our practice, we modified the LigaSure method into a two-step technique that does not require Endo-GIA staplers for the management of secondary splenic pedicels. A large series of patients underwent successful LS by using this technique in our institution. The primary objective of this study was to evaluate the feasibility, efficacy, safety, and cost-effectiveness of this two-step modified LigaSure procedure in LS.
Materials and methods
Patient enrollment
The study protocol was approved by the Institutional Review Board at the First Hospital of Norman Bethune Medical College, Jilin University. Patients (n = 105) who were scheduled for elective LS due to a wide range of splenic disorders (Table 1) were consecutively and prospectively enrolled between June 2006 and August 2010. The study cohort included 24 males and 81 females, with a mean age of 43.6 (range 11-75) years. All the patients volunteered to give informed consent in writing prior to the enrollment.
Splenic disorders of patients scheduled for elective laparoscopic splenectomy (n = 105).
| Diagnosis | n (%) |
|---|---|
| Hematologic diseases | 76 (72.4) |
| Idiopathic thrombocytopenic purpura | 49 (64.5) |
| Hereditary spherocytosis | 17 (22.4) |
| Autoimmune hemolytic anemia | 9 (11.8) |
| Leukemia | 1 (1.3) |
| Hypersplenism | 16 (15.2) |
| Post-hepatitis B cirrhosis | 1 (6.3) |
| Post-hepatitis C cirrhosis | 5 (31.3) |
| Primary hypersplenism | 10 (62.5) |
| Others | 13 (12.4) |
| Splenic cyst | 7 (53.8) |
| Traumatic splenic rupture | 3 (23.1) |
| Splenic abscess | 2 (15.4) |
| Splenic benign tumor | 1 (7.7) |
Preoperative Preparation and Work-up
The patients received routine hematological tests, as well as a coagulation study in cases of infiltrative diseases such as myelofibrosis or lymphoma to exclude compromised hepatic function. Patients who exhibited splenomegaly were further assigned for computed tomographic scan to determine the size and position of the spleen for the purpose of planning patient positioning and trocar placement. Platelet transfusion was indicated for thrombocytopenic patients with a platelet count < 30 × 109/L 12.
Patient Positioning and Port Placement
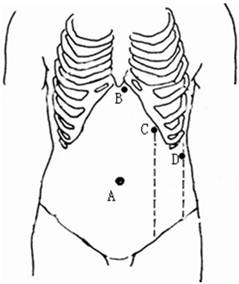
Nasogastric decompression and urethral catheterization were given as routine, and intravenous prophylactic antibiotics were administered 30 min preceding the induction of general anesthesia. Patients were positioned supine with their chests and pelvises fixed with straps and cloth cushions to allow the tilt of the operating table. Following the establishment of pneumoperitoneum, a 10.5-mm port was placed at the umbilicus, and a 30° laparoscope (Karl Storz Endoskope, Tuttlingen, Germany) was introduced. A 5-mm port was positioned on the left of the falciform ligament below the xiphoid, allowing the exposure of the splenic hilum by using a grasper. A 10.5-mm port was placed in the left midclavicular as the main manipulation port 13. Another 5 mm port was at the inferior pole of the spleen on the left anterior axillary line to retract the spleen (Figure 1). In addition, sometimes the positions will vary slightly and naturally according to dimensions of the spleen.
Patient positioning and trocar placement for laparoscopic splenectomy. A: 10.5-mm port for laparoscope; B: 5-mm port for a grasper exposing the splenic hilum; C: 10.5-mm port for the main manipulation port; and D: additional 5-mm port for retraction.
Laparoscopic Mobilization of Spleen
Laparoscopy was performed through the anterior approach. The gastrocolic ligament was transected by using LigaSure (Covidien, Boulder, CO) to expose the lesser omental bursa. The short gastric vessels were divided by using LigaSure or SonoSure (Olympus, Tokyo, Japan), beginning from the inferior pole of the spleen. The lienocolic ligament was subsequently transected adjacent to the spleen to avoid injuries to the colon. Following the mobilization of the spleen, the splenic artery was identified along the upper margin of the pancreatic body. The arterial inflow was interrupted by applying a polymer Hem-o-lock clip (Autosuture, Norwalk, USA) to reduce the spleen size and minimize the blood loss from splenectomy12. The splenic artery was further skeletonized with caution for approximately one centimeter, preferably proximal to the bifurcation, and disrupted by using a clip. The retroperitoneal and diaphragmatic attachments were dissected by using LigaSure, SonoSure, laparoscopic clips, or a combination of these depending on the thickness of the ligaments.
Two-step Control of Secondary Splenic Pedicles
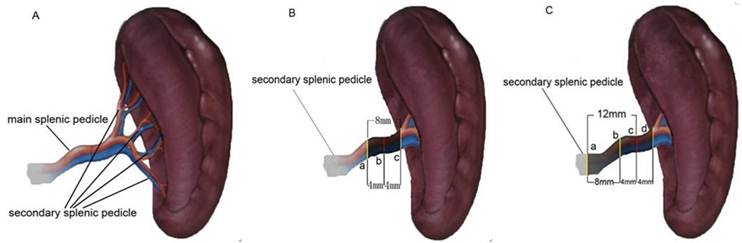
The secondary splenic pedicles (not the main splenic pedicle) were sealed and divided by using LigaSure rather than Endo-GIA staplers. The main splenic pedicle was sufficiently mobilized for the division of secondary splenic pedicles by using a two-step technique (Figure 2). The pedicular vessels were sealed adjacent to the pancreatic tail. Subsequently, the vascular segments were fused and transected in proximity to the spleen. The length of the fused secondary splenic pedicles extended from 4 to 12 mm. Any minor oozing from the perisplenic adipose, splenic capsule or diaphragm was controlled by using SonoSure or clips, especially in the case of severe thrombocytopenia.
Schematic illustrations of splenic pedicels. (A) The main splenic pedicle branches off into secondary pedicles; (B) in the conventional approach, the segment between a and c (8 mm) was coagulated by using LigaSure, followed by the transection along line b; (C) by using our two-step technique, the segment between a and b was coagulated, preceding the further coagulation of the segment between b and d and followed by the transection along line c, resulting in a 12 mm-long coagulation segment.
Specimen Retrieval
Following the transection of the spleen, the port on the left midclavicular line was extended to 5 cm for the retrieval of a spleen specimen. A self-prepared polymer bag was folded and inserted through the trocar hole. The spleen was retracted towards the pelvis, followed by the placement of the retrieval bag onto the splenic bed. Patients were placed in the Trendelenburg position on their left side, whilst the resection specimen, as well as any accessory spleen if applicable, was maneuvered into the retrieval bag by using a second grasper. The specimen bag was fastened and withdrawn from the peritoneal cavity by using a pair of sponge forceps under laparoscopic visualization. The peritoneal cavity was irrigated and examined for any active hemorrhage, especially from short gastric and splenic vessels, following the removal of the spleen specimen. In the case of suspected pancreatic tail injuries, a closed suction drain was placed through the left anterior axillary port.
Postoperative Care
A complete blood cell count test was performed every two days to monitor the platelet count. Aspirin or heparin was administered in the case of thrombocytosis more than 300 × 109/L to prevent thrombotic events. Targarona 14 also suggested that the anticoagulation therapy should be administered to paitients who have the risk factors.
Results
Out of 105 patients, 103 (98.1%) underwent successful LS. Due to the potential of uncontrollable bleeding, laparoscopy was converted to laparotomy in two patients (1.9%) complicated with portal hypertension and splenic tumor, respectively. Concomitant laparoscopic cholecystectomy was performed in four biologically related patients deriving from a single family and suffering from autoimmune hemolytic anemia. The mean duration of operation was 100 ± 15 min, and the mean volume of blood loss was 500 ± 94 mL.
The pathological outcomes were consistent with the preoperative diagnoses, whilst congestive splenomegaly had the highest frequency (95/105, 90.5%). The majority of patients resumed oral intake on postoperative day 2. No transfusion of any blood product was given. No clinically significant morbidities or mortality occurred following LS. Post-splenectomy hyperthrombocytosis was present for 0.5-2 months following the surgery, but it did not cause any thrombotic event in the patients having received prophylactic anticoagulatants.
The mean duration of hospital stay was 7.5 d (range: 6-9 d). An estimated average of USD 1230 (range, 1060-1380) was saved by patients undergoing LS by using LigaSure versus Endo-GIA. Patients were followed up for 3 months to 2 years, showing no occurrence of major complications.
Discussion
The secure control of splenic pedicles is crucial for the successful performance of LS, especially in high-risk patients such as those complicated with idiopathic thrombocytopenic purpura. Uncontrollable hemorrhage when it occurs requires the conversion to laparotomy in most cases. Multiple hemostases are available for the vascular management of LS, including clips, ligatures, and bipolar and ultrasonic coagulation. However, these methods lack consistent and reliable hemostatic effect, prolonging the time required for pedicular division and increasing the risk of bleeding 15. Therefore, the use of these approaches is usually limited to the transection of vessels <3 mm in diameter.
The Endo-GIA stapler is an effective alternative instrument, but its application for pedicular control requires proper positioning as close as possible to the hilum, accurate vascular dissection, and cautious skeletonization. Bleeding from the transection line has been frequently reported in previous studies 16. It has been found that in 70% of cases the pancreatic tail is less than one centimeter from the splenic hilum, and closely adjacent in the remaining cases. The transection of the main splenic pedicle by using the linear stapler is likely to injure the pancreatic tail, which is then subject to pancreatic fistula and iatrogenic pancreatitis. Additionally, the costliness of the delicate Endo-GIA staplers also restricts its use in our practice, and is a heavy burden on the public health care system.
We herein report a modification of the technique of pedicle control in LS, in which hilar vessels are transected using LigaSure alone, based on the anatomy of splenic pedicles. Unlike the main pedicle, the diameters of secondary pedicles are <7 mm, and are more easily controlled by using the LigaSure vessel sealing system. Our two-step coagulation of secondary splenic pedicles, including both the pancreatic and hilar ends, also greatly improves the safety of pedicle control, as it widens the coagulation length from the conventional 4 mm to 12 mm. Moreover, the division of secondary pedicels with LigaSure minimizes the risk of pancreatic injuries.
In some unfavorable cases such as splenomegaly, the laparoscopic procedure is technically demanding due to limited space for manipulation 17-20. The clipping of the splenic artery with Hem-o-lock ligating clip as in our study interrupts splenic inflow, decreases spleen size, and reduces the bleeding risk. Additionally, our technique results in a very low conversion rate (1.9%), whereas the conversion to open splenectomy is caused by extensive perisplenic adhesion or vascular hyperplasia.
Conclusion
Secondary splenic pedicles can be successfully controlled in LS by using a two-step technique with the LigaSure vessel sealing system. The use of LigaSure leads to a shorter time in surgery, less blood loss, and lower medical expense as compared to the Endo-GIA stapler.
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Delaitre B, Maignien B, Icard P. Laparoscopic splenectomy. Br J Surg. 1992;79:1334
2. Torelli P, Cavaliere D, Casaccia M. et al. Laparoscopic splenectomy for hematological diseases. Surg Endosc. 2002;16:965-71
3. Canda AE, Sucullu I, Ozsoy Y. et al. Hospital experience, body image, and cosmesis after laparoscopic or open splenectomy. Surg Laparosc Endosc Percutan Tech. 2009;19:479-83
4. Heslin MJ, Winzeler AH, Weingarten JO. et al. Laparoscopic adrenalectomy and splenectomy are safe and reduce hospital stay and charges. Am Surg. 2003;69:377-81
5. Brody FJ, Chekan EG, Pappas TN. et al. Conversion factors for laparoscopic splenectomy for immune thrombocytopenic purpura. Surg Endosc. 1999;13:789-91
6. Katkhouda N, Waldrep DJ, Feinstein D. et al. Unresolved issues in laparoscopic splenectomy. Am J Surg. 1996;172:585-90
7. Vargün R, Göllü G, Fitöz S. et al. En-bloc stapling of the splenic hilum in laparoscopic splenectomy. Minim Invasive Ther Allied Technol. 2007;16:360-2
8. Kercher KW, Novitsky YW, Czerniach DR. et al. Staple line bleeding following laparoscopic splenectomy: intraoperative prevention and postoperative management with splenic artery embolization. Surg Laparosc Endosc Percutan Tech. 2003;13:353-6
9. Romano F, Caprotti R, Franciosi C. et al. Laparoscopic splenectomy using Ligasure. Preliminary experience. Surg Endosc. 2002;16:1608-11
10. Gelmini R, Romano F, Quaranta N. et al. Sutureless and stapleless laparoscopic splenectomy using radiofrequency: LigaSure device. Surg Endosc. 2006;20:991-4
11. Romano F, Gelmini R, Caprotti R. et al. Laparoscopic splenectomy: ligasure versus EndoGIA: a comparative study. J Laparoendosc Adv Surg Tech A. 2007;17:763-7
12. Grahn SW, Alvarez J 3rd, Kirkwood K. Trends in Laparoscopic Splenectomy for Massive Splenomegaly. Arch Surg. 2006;141:755-62
13. Machado MA, Makdissi FF, Herman P. et al. Exposure of splenic hilum increases safety of laparoscopic splenectomy. Surg Laparosc Endosc Percutan Tech. 2004;14:23-5
14. Targarona EM. Portal vein thrombosis after laparoscopic splenectomy: the size of the risk. Surg Innov. 2008;15:266-70
15. Poulin EC, Mamazza J, Schlachta CM. Splenic artery embolization before laparoscopic splenectomy. An update. Surg Endosc. 1998;12:870-5
16. Targarona EM, Espert JJ, Bombuy E. et al. Complications of laparoscopic splenectomy. Arch Surg. 2000;135:1137-40
17. Terrosu G, Donini A, Baccarani U. et al. Laparoscopic versus open splenectomy: In the management of splenomegaly: our preliminary experience. Surgery. 1998;124:839-43
18. Targarona EM, Espert JJ, Balagué C. et al. Splenomegaly should not be considered a contraindication for laparoscopic splenomegaly. Ann Surg. 1998;228:35-9
19. Kercher KW, Matthews BD, Walsh RM. et al. Laparoscopic splenectomy for massive splenomegaly. Am J Surg. 2002;183:192-6
20. Wang Y, Zhan X, Zhu Y. et al. Laparoscopic splenectomy in portal hypertension: a single-surgeon 13-year experience. Surg Endosc. 2010;24:1164-9
Author contact
Corresponding author: Guangyi Wang, MD, PhD, Department of Hepatobiliary and Pancreatic Surgery, the First Hospital, Jilin University, Jilin 130021, China (Tel: 86-431-88783331; E-mail:wgymdcom).

 Global reach, higher impact
Global reach, higher impact