Impact Factor ISSN: 1449-1907
Int J Med Sci 2012; 9(6):480-487. doi:10.7150/ijms.4583 This issue Cite
Research Paper
Osteogenic Differentiation of Dental Follicle Stem Cells
1. Department of Biomedical Sciences, Medical School, University of Foggia, ITALY;
2. Department of Gynecology, Obstetrics and Neonatology, Faculty of Medicine and Surgery, University of Bari “Aldo Moro”, ITALY;
3. Department of Basic Medical Sciences, Section of Human Anatomy and Histology, University of Bari “Aldo Moro”, ITALY.
Received 2012-5-12; Accepted 2012-7-31; Published 2012-8-13
Abstract
Background: Stem cells are defined as clonogenic cells capable of self-renewal and multi-lineage differentiation. A population of these cells has been identified in human Dental Follicle (DF).
Dental Follicle Stem Cells (DFSCs) were found in pediatric unerupted wisdom teeth and have been shown to differentiate, under particular conditions, into various cell types of the mesenchymal tissues.
Aim: The aim of this study was to investigate if cells isolated from DF show stem features, differentiate toward osteoblastic phenotype and express osteoblastic markers.
Methods: We studied the immunophenotype of DFSCs by flow cytometric analysis, the osteoblastic markers of differentiated DFSCs were assayed by histochemical methods and real-time PCR.
Results: We demonstrated that DFSCs expressed a heterogeneous assortment of makers associated with stemness. Moreover DFSCs differentiated into osteoblast-like cells, producing mineralized matrix nodules and expressed the typical osteoblastic markers, Alkaline Phosphatase (ALP) and Collagen I (Coll I).
Conclusion: This study suggests that DFSCs may provide a cell source for tissue engineering of bone.
Keywords: dental follicle, stem cells, osteogenic markers, bone tissue engineering, human postnatal dental tissue.
INTRODUCTION
The DF is a loose connective tissue sac that surrounds the unerupted tooth and plays different roles in the tooth development. It is an ectomesenchymal tissue surrounding the enamel organ and the dental papilla of the developing tooth germ prior of the eruption. Its presence is required for eruption [1,2], indeed it appears to regulate the bone remodelling, influencing osteoblast and osteoclast activity during eruption [3]. As the tooth pierces the gingiva, the DF differentiates into the Periodontal Ligament (PDL) that anchors the tooth to the surrounding alveolar bone in its socket. It has been postulated that DFSCs as well as differentiate in PDL fibroblasts, may differentiate in tooth cementoblasts, and in alveolar bone osteoblasts [4]: this observation is consistent with the hypothesis of the presence of Mesenchymal Stem Cells (MSCs) with osteoblastic potential in DF.
Dental pulp (DP) has been identified as a promising source of MSCs for tissue engineering [5-8]. MSCs are self-renewable and can differentiate into all cell lineages that form mesenchymal and connective tissues [5]. DFSCs are either more undifferentiated than Dental Pulp Stem Cells (DPSCs), thus dental bud is a more productive tissue of MSCs than mature DP and would have alternative applications in bone and periodontal tissue engineering.
Therefore, the interest for tissue engineering research has been increasing rapidly among dental researchers and institutes. One reason for this is that dentists are familiar with tissue regeneration techniques, that are now common in dental hospitals [9,10]. Dentists are obviously the surgeons more involved and motivated in collecting the dental buds from which the biotechnologists can isolate DFSCs.
Furthermore, the development of techniques to generate whole teeth and bone is ongoing [11,12].
A key factor that can improve this technology within the context of modern tissue engineering is the use of adult stem cells. After birth, mesenchymal cells are called 'mesenchymal stem cells'.
In the adult, MSCs maintain the physiological tissue turnover and, upon injury or disease, they differentiate to allow tissue regeneration. MSCs have been differentiated, in vitro, into many specialized connective tissues [13].
The ability to generate human tissues and organs from cells grown in vitro is a big task of regenerative medicine, reproducing organism structures from stem cells was an insurmountable effort until several unrelated disciplines converged into the field of tissue engineering [12]. To engineer a functional biological structure, cells must be induced to differentiate and to synthesize the appropriate extracellular matrix molecules in the overall shape and dimensions of the diseased or missing tissues/organs.
Bone marrow has been studied since many years as a valid source of stem cells both of the hematopoietic and mesenchymal lineage. However many other MSCs sources, that could be also more convenient, have been identified.
To date, in human postnatal dental tissues, five different sources of MSCs have been already identified: dental pulp [5-8], periodontal ligament [5], exfoliated deciduous teeth [14], dental follicle [15] and root apical papilla [16].
Harvesting DFSCs from teeth is mostly noninvasive, although tooth extraction itself is an invasive technique.
Naturally, to obtain DFSCs, the extraction of a tooth before its eruption is required; but the early (age 7-12) removal of the third molar gives excellent stem cells without losing a useful tooth. Thus patients selected for the extraction are usually subjects that will undergo dental overcrowding which is worsened by wisdom teeth eruption.
Since DFSCs are capable to differentiate into different cell types other than PDL fibroblasts, we can assume that cells with stem features are present within the DF.
Given the above background, the objectives of this study were to investigate if cells isolated from DF show stem features, differentiate toward osteoblastic phenotype and express osteoblastic markers.
PATIENTS, MATERIALS and METHODS
Patients and Cell cultures
Human DFSCs were isolated and cultivated as previously reported in our previous experience with DPSCs [5-8].
Normal human third molars follicles were collected from tooth buds of healthy pediatrics patients, aged 8-12 years, that underwent to extractions for orthodontics reasons, after obtaining informed consent from each patient parents.
DFs were digested for 1 hour at 37° C in agitation in a solution of 3 mg/ml type I collagenase plus 4 mg/ml dispase (Gibco Ltd., Uxbridge, UK). Single-cell suspensions were obtained by passing the cells through a 70µm BD Falcon strainer (Falcon) (Becton & Dickinson, Sunnyvale, CA).
After filtration, single cell suspensions were seeded in a 25 cm2 culture flask and cultured with α-MEM supplemented with 5% fetal bovine serum (FBS), 100 U/ml penicillin-G, 100 μg/ml streptomycin and 0.25 μg/ml fungizone (Gibco Limited, Uxbridge, United Kingdom).
Flasks were incubated at 37° C and 5% CO2 and the medium was changed every 3 days.
In order to study DFSCs proliferation rate, 2 x 105 cells were plated in a six well plate, detached and counted after 3, 5 and 7 days of culture. These values were compared to those obtained culturing human Bone Marrow Stromal Cells (hBMSCs) obtained as previously described [17].
For the induction of osteoblastic differentiation, cells were grown in an osteogenic medium consisting of α-MEM supplemented with 2% FBS, 10-8 M dexamethasone and 50 µg/ml ascorbic acid (Sigma Aldrich, Milan, Italy).
For the evaluation of DFSCs' ability to form mineralized nodules in vitro, we cultured them for 30 days in the osteogenic medium supplemented with 10 mM β -glycerophosphate (Sigma Aldrich, Milan, Italy).
Immunophenotypic Analysis
The following fluorescein isothiocyanate (FITC)-conjugated or phycoerythrin (PE)-conjugated mAbs were used for immunofluorescent staining of DFSCs: PE-CD73, PE-CD90, PE-CD146, isotype control (all BD-Pharmingen, Milano, Italy), and FITC-CD44, FITC-CD105, FITC-HLA-I and FITC-CD45 (Beckman Coulter, Inc., Milano, Italy). Cells were washed in FACS buffer (phosphate-buffered saline pH 7.2, 0.2% bovine serum albumin, and 0.02% sodium azide) containing 3% human serum, incubated with fluorochrome-conjugated mAbs for 15 minutes and then washed with the same buffer before flow cytometric analysis. Data were acquired using a FC500 (Beckmann Coulter, Inc.) flow cytometer and analyzed using Kaluza software (Beckmann Coulter, Inc.). The area of positivity was determined using an isotype-matched mAb.
Von Kossa Stainining
The capacity of DFSCs to form mineralized nodules in vitro, a feature normally attributed to osteoblasts, was assessed by performing a Von Kossa staining on cells cultivated for thirty days in the appropriate medium. Thus cells were washed two times with distilled water and then fixed with a 3% formaldehyde, 2% sucrose solution for 10'.
After being rinsed in distilled water, cells were treated with a 5% silver nitrate solution in dark for 30' and then exposed to bright light (a 100W lamp) for 1 hour, laying them over a white background.
Alkaline Phosphatase
Expression of ALP, a typical osteoblast marker, was assessed in DFSCs, grown in osteogenic medium, with Leukocyte Alkaline Phosphatase Kit, a commercial kit based on naphthol AS-BI and fast red violet LB (Sigma Aldrich).
Cells were fixed with a citrate-acetone-formaldehyde fixative for 30'' at room temperature.
After being gently rinsed with distilled water, cells were incubated for 15' in dark with alkaline-dye mixture (NaNO2, FRV-Alkaline Solution, Naphthol AS-BI Alkaline Solution) and finally washed with water.
RNA Isolation and Real-Time PCR
To examine the genic expression of specific osteoblastic markers such as ALP, Coll I and RUNX2, DFSCs were subjected to real-time PCR: all the results were normalized versus actin.
Total RNA was extracted from DFSCs cultured in osteoblastic medium for ten and seventeen days and undifferentiated cells using spin columns (RNeasy, QIAGEN, Hilden, Germany), according to the manufacturer's instructions.
RNA was reverse-transcribed using the Super Script First-Stand Synthesis System kit for RT-PCR (Invitrogen, Carlsbard, CA, USA): an RT mixture containing 1µg total RNA, dNTPs (deoxyribonucleoside triphosphates), Oligo(dT), RT buffer, MgCl2, DTT (dithiothreitol), RNaseOUT, SuperScript II RT, DEPC (diethyl pyrocarbonate)-treated water to final volume 100µL was prepared, according to the manufacturer's instructions. cDNA was amplified with the iTaq SYBR Green supermix with ROX kit (Bio-Rad Laboratories, Bio-Rad Laboratories Inc., Hercules, CA, USA) and the PCR amplification was performed using the Chromo4 Real-Time PCR Detection System (Bio-Rad Laboratories).
The following primer pairs were used for the real Time PCR amplification: sense ALP 5'-CGCACGGAACTCCTGACC-3'; antisense ALP 5'-GCCACCACCACCATCTCG-3'; sense Coll I 5'-CGTGGCAGTGATGGAAGTG-3'; antisense Coll I 5'-AGCAGGACCAGCGTTACC-3'; sense RUNX2 5'-GGAATGCCTCTGCTGTTATG-3'; antisense RUNX2 5'-TTCTGTCTGTGCCTTCTGG-3'; sense Actin 5'-AATCGTGCGTGACATTAAG-3'; antisense Actin 5'-GAAGGAAGGCTGGAAGAG-3'.
The running conditions were as follows: incubation at 95°C for 3', and 40 cycles of incubation at 95°C for 15'' and 60°C for 30''.
After the last cycle, the melting curve analysis was performed into 55-95°C interval by incrementing the temperature of 0.5°C. The fold change values were calculated by Pfaffl's method [18].
Statistical Analyses
Statistical analyses were performed by Student's t-test with the Statistical Package for the Social Sciences (spssx/pc) software (SPSS, Chicago, IL, USA). The results were considered statistically significant for p<0.05.
RESULTS
Immunophenotype of DFSCs
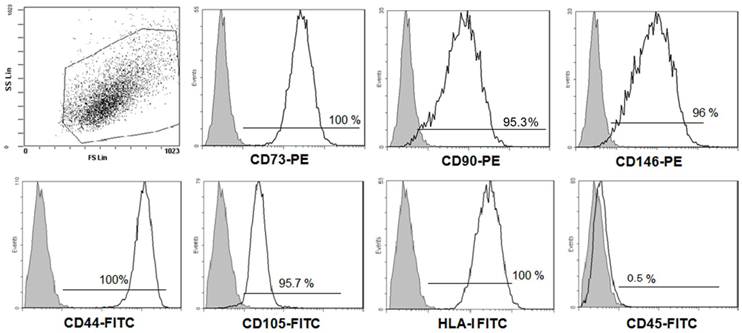
Firstly, to ensure that our DFSCs culture met minimal criteria to be called MSC [19], three DFSC cultures (passages 1-2 at the time of testing) were characterized in more details. Flow cytometric analysis of research accepted MSC cell surface markers was performed. Figure 1 shows flow cytometry histograms from one representative DFSC culture. In sum, all DFSC cultures exhibited ≥95% expression of CD73, CD90, CD146, CD44, CD105 and HLA-I, while they were negative for CD45, a common leukocyte antigen.
Phenotypes of DFSCs. The expression of the indicated mesenchymal stem cell markers on DFSCs was analyzed using flow cytometry. Results from one representative DFSC culture are shown. The grey histograms signify staining with isotype controls, and the white histograms represent staining with the specified surface marker antibody.
Proliferation rate of DFSCs Clonogenic Populations
It is well documented that osteoprogenitor cells can be isolated from bone marrow aspirates [13-14,17], and from dental pulp tissue [20] due to their ability to adhere to a plastic substrate, and, under appropriate conditions, begin to proliferate.
As compared to bone marrow-derived MSCs, DFSCs were easily isolated from impacted teeth and immediately grown in culture.
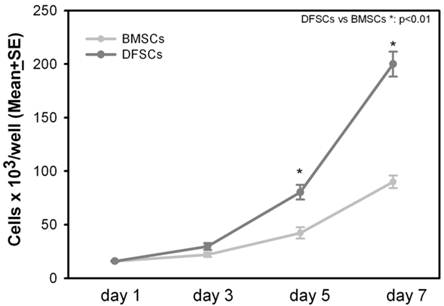
Cell proliferation rate of DFSCs was measured at the first passage after the extraction from the original tissue. DFSCs and BMSCs were seeded at the same cell density (2x105 cells/well): DFSCs proliferation rate was significantly higher in comparison to BMSCs, indeed after seven days of culture, DFSCs reached the average number of 2x106 cells/well compared to 0,9x106 cells/well of BMSCs [Figure 2].
DFSCs proliferationin vitro. Proliferation of DFSCs and BMSCs after three, five and seven days of culture.
ALP positivity and mineral matrix nodules deposition in DFSCs
We investigated whether, DFSCs properly isolated from three different patients and cultivated in osteogenic medium, were able to differentiate in osteoblast-like cells.
Thus, using a histochemical assay, we evaluated the expression of the osteoblast marker ALP.
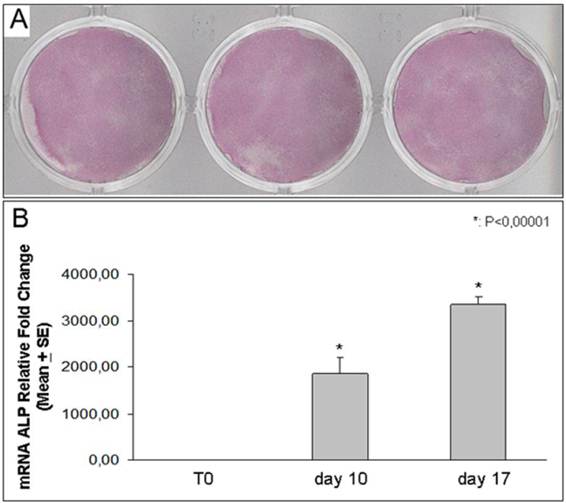
We found that DFSCs harvested from all three different dental follicles, and treated with osteogenic medium, expressed high levels of ALP expression [Figure 3]. In addition ALP expression was increased over the time starting from point 0 to 17 days of culture.
ALP expression. Histochemical staining for ALP in DFSCs incubated in osteogenic medium for 10 days (samples from three different patients) (A). Real Time-PCR analysis shows ALP expression in DFSCs cultured for 10 and 17 days in osteogenic medium: the expression of the enzyme progressively increased up to 3 thousand of times (B).
Moreover, long-term cultures (thirty days) of the three different DFSCs clones, demonstrated the capacity to form mineralized matrix nodules [Figure 4].
Mineralized nodules formation. Von Kossa staining shows mineralized nodules formation by DFSCs (from three patients) incubated for thirty days in osteogenic medium.

Osteoblast markers expression in DFSCs
To confirm ALP expression and its time-dependent increasing evidenced with the histochemical assay, we evaluated the enzyme RNA expression using Real Time-PCR. We demonstrated that the ALP expression was progressively up-regulated in DFSCs after ten and seventeen days of differentiation obtained with osteogenic medium [Figure 3].
Based on the results obtained on ALP expression and mineral nodules formation in DFSCs, we also evaluated the expression of osteoblast master gene RUNX2. RUNX2 directly regulates the expression of many osteoblastic markers such as Coll I which is the most abundant protein produced by osteoblasts [21,22].
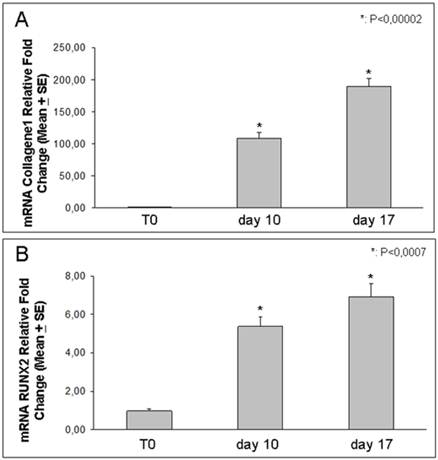
Using real-time PCR, we demonstrated that the expression of either RUNX2 or Coll I, increased by the time after 10 and 17 days of osteoblast differentiation [Figure 5]. In particular RUNX2 expression showed a 6-7 fold increase respectively at 10 and 17 days relative to T0, while Coll I increased even more, showing a strong 100-200 fold increase respectively at 10 and 17 days relative to T0.
Coll I and RUNX2 expression. Real Time-PCR analysis shows RUNX2 and Coll I increased expression in DFSCs cultured for 10 and 17 days in osteogenic medium: Coll I increased progressively up to two hundred times and RUNX2 increased up to seven times.
DISCUSSION
Our data indicate that DFSCs express a heterogeneous assortment of makers associated with stemness, and have a higher proliferation rate, compared to BMSCs. Moreover the tooth bud is easy to access and is a considerably larger tissue than mature tooth pulp. We demonstrate the presence in DFSCs, of a highly proliferative clonogenic cell population, which suggests their remarkable capacity of self-renewal and propagation. Each colony originates from a single progenitor cell (colony-forming unit-fibroblast, CFU-F) and displays a wide variation in cell morphology and growth potential [5, 13, 17, 20]. The cells within each colony are characterized by a typical fibroblast-like morphology analogous to the progeny of human bone marrow and human dental pulp tissue [16, 17, 20].
Their ability to differentiate into a wide variety of cell types, together with the possibility of obtaining a high number of cells, make them good candidates for the repair and regeneration of a large variety of tissues, [12]. Two of the main characteristics of stem cells are self-renewal or self-propagation and competency for multipotent differentiation.
Differentiation into osteogenic lineage is considered to be a trademark event of BMSCs [13] and DPSCs [5-8, 17, 20]. In this study we demonstrated a functional osteogenic differentiation of DFSCs towards an osteoblastic phenotype.
We showed that DFSCs, cultured in opportune conditions, highly increased the osteoblast main markers, RUNX2, Coll I and ALP, and in vitro formed mineralized matrix nodules.
The osteogenic commitment capacity of DFSCs is comparable to that of BMSCs [13,17] and DPSCs [17, 20].
The use of mesenchymal stem cells derived from dental tissues, which are easy to access for dentists and oral surgeons, with their high proliferation features and capacity to differentiate into osteoblasts, offers a great potential for the future of clinical dentistry [17].
However, these cells would need a good scaffold to facilitate their integration, differentiation, and matrix synthesis and to promote multiple specific interactions between cells [10,12,17].
In this scenario, DFSCs have to be considered stem cells perfect to be used for tissue engineering since they are characterized by a high proliferation rate, multidifferentiation ability, easy accessibility, high viability, opportunity to be safely cryopreserved and expression of mesenchymal markers.
In addition, the stem cells derived from teeth are large spindle-shaped cells with a large central nucleus, abundant cytoplasm, and cytoplasmic extensions in culture. These adherent cells are morphologically identical to the mesenchymal stem cells obtained from bone marrow (BMSCs) [17,20]. Moreover stem cells, derived from different dental tissues and obtained from variable stages of tooth development, can be used to produce partial or total tissue regeneration.
In literature there was demonstrated the possibility of generating a piece of pulp/dentin complex in vitro as a filling material [27-29].
DFSCs share many peculiarities with periodontal ligament stem cells (PDLSCs), although they are easier to isolate. PDLSCs come from the mature tooth compartment. However, as recently reported, PDLSCs have been used in autologous transplantation therapy of periodontium regenerations [30]. Considering that periodontal disease causes alveolar bone resorption and tooth loss, regenerating periodontal tissues would represent the ideal therapeutic aid in dentistry [31-33].
Autologous PDLSCs showed the best regenerating capacity for PDL, alveolar bone and cementum, as well as peripheral nerve and blood vessel, which was evaluated by conventional and immune histology [31]. DFSCs could be a more proliferative and abundant source of stem cells to be easily isolated from wisdom tooth bud of young patients, stored and used in cases of dental tissues regeneration necessity that could occur in the adult life.
CONCLUSIONS
We demonstrated in our work that DFSCs met criteria to be called MSCs and have a remarkable osteogenic potential.
Indeed the ability of these stem cells to differentiate into osteoblasts is certain: once they are cultured in osteogenic induction medium, several clusters of cells adhere, assume an osteoblast-like morphology and express the typical osteoblastic marker such as ALP and Coll I. Moreover DFSCs formed mineralized matrix nodules showing a mature osteoblast exclusive feature.
DFSCs have a similar, if not identical, potential of BMSCs, but they come from a dental bug that could generate a problematic wisdom tooth and proliferate at a higher rate. Therefore DFSCs could be used for bone regeneration of stomatognathic and other systems, and represent an excellent cell source for tissue engineering therapy. Moreover DFSCs can differentiate in the other cells of the connective tissues, thus, more diversified clinical applications should be investigated.
As a potential stem cell source, dental stem cell banking may be a necessary step and further progresses on establishing individualized induced pluripotent stem cells for dental tissue regeneration should be imminent.
Acknowledgements
Funding for this work was awarded from Ministero dell'Istruzione, dell'Università e della Ricerca -PRIN 2009 Mori (Progetto di Ricerca d'Interesse Nazionale- Grant 2009).
CONFLICT OF INTEREST
The authors have declared that no competing interest exists.
References
1. Cahill DR, Marks SC Jr. Tooth eruption: evidence for the central role of the dental follicle. J Oral Pathol. 1980;9:189-200
2. Marks SC Jr, Cahill DR. Experimental study in the dog of the non-active role of the tooth in the eruptive process. Arch Oral Biol. 1984;29:311-22
3. Wise GE, Yao S. Regional differences of expression of bone morphogenetic protein-2 and RANKL in the rat dental follicle. Eur J Oral Sci. 2006;114:512-6
4. Bosshardt DD, Schroeder HE. Cementogenesis reviewed: a comparison between human premolars and rodent molars. Anat Rec. 1996;245:267-92
5. Ballini A, De Frenza G, Cantore S. et al. In vitro stem cell cultures from human dental pulp and periodontal ligament: new prospects in dentistry. Int J Immunopathol Pharmacol. 2007;20(1):9-16
6. Mori G, Brunetti G, Oranger A. et al. Dental pulp stem cells: osteogenic differentiation and gene expression. Ann N Y Acad Sci. 2011;1237(1):47-52
7. Mori G, Centonze M, Brunetti G. et al. Osteogenic properties of human dental pulp stem cells. J Biol Regul Homeost Agents. 2010;24(2):167-75
8. Ballini A, Capodiferro S, Cantore S. et al. Dental Pulp Stem Cells Curriculum Vitae. Journal Of Osteology And Biomaterials. 2010;1(1):23-27
9. Shayesteh YS, Khojasteh A, Soleimani M. et al. Sinus augmentation using human mesenchymal stem cells loaded into a beta-tricalcium phosphate/hydroxyapatite scaffold. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106:203-9
10. Annunziata M, Guida L, Perillo L. et al. Biological response of human bone marrow stromal cells to sandblasted titanium nitride-coated implant surfaces. J Mater Sci Mater Med. 2008;19(12):3585-91
11. Honda MJ, Tsuchiya S, Sumita Y. et al. The sequential seeding of epithelial and mesenchymal cells for tissue-engineered tooth regeneration. Biomaterials. 2007;28:680-9
12. Boccaccio A, Ballini A, Pappalettere C. et al. Finite element method (FEM), mechanobiology and biomimetic scaffolds in bone tissue engineering. Int J Biol Sci. 2011;7(1):112-32
13. Pittenger MF, Mackay AM, Beck SC. et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143-7
14. Miura M, Gronthos S, Zhao M. et al. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci U S A. 2003;100:5807-12
15. Yao S, Pan F, Prpic V. et al. Differentiation of stem cells in the dental follicle. J Dent Res. 2008;87:767-71
16. Sonoyama W, Liu Y, Yamaza T. et al. Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: a pilot study. J Endod. 2008;34:166-71
17. Colucci S, Brunetti G, Oranger A. et al. Myeloma cells suppress osteoblast through sclerostin secretion. Blood Cancer Journal. 2011;1(6):e27
18. Pfaffl MW. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001;29:e45
19. Dominici M Le Blanc K, Mueller I. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006;8:315-7
20. Huang GT, Gronthos S, Shi S. Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res. 2009;88:792-806
21. Gronthos S, Mankani M, Brahim J. et al. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A. 2000;97:13625-30
22. Hassan MQ, Gordon JA, Beloti MM. et al. A network connecting Runx2, SATB2, and the miR-23a~27a~24-2 cluster regulates the osteoblast differentiation program. Proc Natl Acad Sci U S A. 2010;107(46):19879-84
23. Brunetti G, Oranger A, Mori G. et al. Sclerostin is overexpressed by plasma cells from multiple myeloma patients. Ann N Y Acad Sci. 2010;1237:19-23
24. Seo BM, Miura M, Gronthos S. et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004;364:149-55
25. Morsczeck C, Götz W, Schierholz J. et al. Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol. 2005;24:155-65
26. Sonoyama W, Liu Y, Fang D. et al. Mesenchymal stem cell mediated functional tooth regeneration in swine. PLoS One. 2006;1:e79
27. Iohara K, Zheng L, Ito M. et al. Side population cells isolated from porcine dental pulp tissue with self-renewal and multipotency for dentinogenesis, chondrogenesis, adipogenesis, and neurogenesis. Stem Cells. 2006;24(11):2493-503
28. Nakashima M, Akamine A. The application of tissue engineering to regeneration of pulp and dentin in endodontics. J Endod. 2005;31:711-8
29. Iohara K, Nakashima M, Ito M. et al. Dentin Regeneration by Dental Pulp Stem Cell Therapy with Recombinant Human Bone Morphogenetic Protein 2. J Dent Res. 2004;83(8):590-5
30. Iohara K, Imabayashi K, Ishizaka R. et al. Complete pulp regeneration after pulpectomy by transplantation of CD105+ stem cells with stromal cell-derived factor-1. Tissue Eng Part A. 2011;17(15-16):1911-20
31. Park JY, Jeon SH, Choung PH. Efficacy of periodontal stem cell transplantation in the treatment of advanced periodontitis. Cell Transplant. 2011;20:271-85
32. Mori G, Brunetti G, Colucci S. et al. Osteoblast apoptosis in periodontal disease: role of TNF-related apoptosis-inducing ligand. Int J Immunopathol Pharmacol. 2009;22(1):95-103
33. Mori G, Brunetti G, Colucci S. et al. Alteration of activity and survival of osteoblasts obtained from human periodontitis patients: role of TRAIL. J Biol Regul Homeost Agents. 2007;21(3-4):105-14
Author contact
Corresponding author: Andrea Ballini, Address: P.zza G. Cesare N. 11, Bari (70124) Italy. Phone: (+39) 3492432155 E-mail: g.moriit.

 Global reach, higher impact
Global reach, higher impact