Impact Factor ISSN: 1449-1907
Int J Med Sci 2011; 8(2):148-155. doi:10.7150/ijms.8.148 This issue Cite
Research Paper
The Value of Serum Biomarkers (Bc1, Bc2, Bc3) in the Diagnosis of Early Breast Cancer
1. İzmir Atatürk Training and Research Hospital 1st Surgical Clinic
2. İzmir Atatürk Training and Research Hospital 1st Biochemistry Clinic
3. Dokuz Eylül University Institute of Oncology
4. İzmir Atatürk Training and Research Hospital 1st Pathology Clinic
Received 2010-11-10; Accepted 2011-1-31; Published 2011-2-12
Abstract
Background: Surface enhanced laser desorption/ionization time-of-flight mass spectrometry (SELDI-TOF) is an approach to biomarker discovery that combines chromatography and mass spectrometry. We aimed to consider the efficacy of Bc1, Bc2, and Bc3 serum biomarkers on early detection of breast cancer (BC) in this study.
Study Design: In this prospective study, 91 patients who were admitted to our hospital between January 2007 and July 2008 were included. Serum samples from 91 women were stored at -80 °C until use. The cancer group included 27 cases of BC. The benign breast disease group included 24 women with benign breast diseases and control group 37 age-matched apparently healthy women. The data obtained for these three groups of patients was worked out for each serum biomarker (Bc1, Bc2, and Bc3) by using SELDI-TOF individually and compared with each other separately and evaluated statistically.
Results: Bc2 possesses the highest individual diagnostic power. Bc2 was statistically significant in comparison between the malignant disease group, control group and benign disease group. Bc1 was statistically significant in the malignant disease group compared to control group as well as in the benign disease group compared to control group. Thus Bc1, rather than showing malignant progression, it shows tumoral progression or inflammatory process. Bc3 was found upregulated in all malignant cases; however, it was not statistically significant compared to the benign disease group or the control group.
Conclusions: It has been shown that Bc2 profiles might be useful in clinical practice to improve BC diagnosis. However none of the proteomics reach reasonable AUC values for the discrimination of the BC. Additional confirmation in larger and similarly-designed prospective studies is needed to consider of the efficacy of Bc1 and Bc2 in early diagnosis of the BC.
Keywords: Breast Cancer diagnosis, Serum Biomarkers, Bc1, Bc2, and Bc3
INTRODUCTION
Breast cancer (BC) is the most common cancer among women in Westernized populations. In France, 41,000 new cases are diagnosed yearly and 25% of these women are below the age of 50 (1-4). BC has heterogeneous behavior and the frequency of metastasis in regional lymph nodules depends on tumor type (5).
Mammographic imaging is the most effective approach for diagnosing BC in women older than 50 years of age. Although new improvements are being made in the resolution of these imaging techniques, tumors smaller than 5 mm usually go undiagnosed. Moreover, as dense breast tissue decreases the mammographic sensitivity in young women, the effectiveness of mammography has not been established (7). Finally, high-grade tumors cannot be diagnosed with 1 to 2 years of regular mammography imaging. For these reasons, new approaches should be developed in order to improve diagnosis of BC and to increase the overall and disease free survival rates of patients who were diagnosed with this disease (8,9).
The high heterogeneity of BC warrants multiple biomarkers for early diagnosis of the disease. Many studies have shown that several proteins change in cancer. These changes may cause measurable alterations and secretion of marker proteins to body fluids. Among the available serum biomarkers, the most popular one is the cancer antigen (Ca). Ca 15.3 is used in monitoring BC and for early diagnosis of BC metastases. Ca 15.3 measurements, however, are not useful for diagnosis; it does not provide benefits in therapeutic decision-making in patients with BC (10). It is therefore essential to discover new biomarkers to manage different stages of BC development. Biomarkers may be promising in diagnosing development or progress of the disease and monitoring the treatment.
Briefly, mass spectrometry and 2D gel electrophoresis technology coupled with advanced bioinformatics (11) enhanced the capacity of characterizing new biomarkers (12). Surface-enhanced laser desorption/ionization time-of-flight (SELDI-TOF) mass spectrometry is another approach that integrates chromatography and mass spectrometry. SELDI-TOF is an appropriate method to monitor protein changes in complex cellular extracts or body fluids (serum, plasma, urea, nipple aspiration material, etc) (13). Various selective chips to which biomaterials may stick are used. Each of the different chip surfaces grip a proteins sub-line analyzed by the TOF mass spectrometry. Several comparative studies have described marked and different forms of protein in prostate, bladder, breast, melanoma, and ovarian cancers (14-20). Li et al. (21) should be congratulated for a valiant effort to validate 3 previously identified serum BC biomarkers by surface-enhanced laser desorption/ionization time-of-flight mass spectrometry (SELDI-TOF MS). They observed three serum peaks to distinguish BC patients from controls by SELDI-TOF. They called the serum breast cancer biomarkers as Bc1, Bc2, and Bc3. The present study aims to evaluate the effectiveness of Bc1, Bc2 and Bc3 in the early diagnosis of the BC in a prospective clinical trial.
METHODS
Patients
This prospective study was performed in Atatürk Training and Research Hospital First surgical department between January 2007 and July 2008. The patients who consented to be in the study and were between 18-75 years of age were included in the study. The patients were divided into three groups. Group 1: BC group, Group 2: Benign breast disease group, and Group 3: Healthy women group. The pathological diagnoses were based on excisional biopsy or segmental mastectomy in group 1 and 2. The Healthy women group was the women who had no complaints about their breast and the mammography and ultrasound study were normal. The patients with diabetes mellitus, chronic obstructive pulmonary disease, and other site malignancy were excluded from the study. The patients who had chemotherapy or radiotherapy previously were also excluded from the study. The patients whose pathological result was ductal carcinoma in situ in group 1 were excluded from the study. The patients with metastatic and locally advanced BC (stage IIIA, IIIB, IIIC, and IV) were not included in the study. All women signed a consent form before serum collection for this institutional review board (IRB)-approved study. Consent of subjects and the ethics board of Izmir Ataturk Training and Research Hospital were obtained. Serum samples were obtained from the patients who were included in the study. Blood sampling was performed after surgery in the patients who were underwent surgical intervention (in group1 and 2). The serum samples in group 3 were obtained after the mammography and ultrasonography examination. Sera collected from these patients were stored in the laboratory of Izmir Hıfzısıhha Institute at -80°C. The blood samples were analyzed for Bc1, Bc2, and Bc3 serum proteins using SELDI-TOF analysis method. Results were compared within three groups and 3 biomarkers (Bc1, Bc2, and Bc3) individually and were evaluated statistically.
SELDI Analysis
Sample Preparation
Blood samples were collected before operation and cure, following 12 hours of fasting, in sitting position in 8-ml vacuum tubes containing gel (BD™ P100 Blood Collection System for Plasma Protein Preservation). Samples were centrifuged at 1500 g for 15 minutes to separate sera. Serum samples were divided in 250 µL units and were stored at -80ºC until the time of analyses.
Serum Protein Profile Determination
Immobilized metal affinity capture arrays (IMAC30) protein chips loaded with Cu2+ metal were used to profile proteins in serum analyses. The samples were loaded to defined locations using a bioprocessor. IMAC 30 protein chips, with 50 µL 100mM CuSO4 on each sample, were incubated at room temperature for 5 minutes. The samples were then rinsed with distilled water and washed with 200 µL binding solution (500mM NaCl, 100mM NaH2PO4/NaOH, pH 7.0) three times for 10 minutes. All sera were first diluted with dilution solution (9 M urea, 50mM Tris/HCl, pH 9.0,2 % (wv1) CHAPS) at the ratio of 5:1. Sera were then diluted again with binding solution at the ratio of 10:1 and were applied in 100 µL amounts in the wells on the chip. Protein chips on which the samples were loaded were kept in the horizontal shaker for one hour (at 900 rpm and room temperature) to ensure protein binding. Chips were washed 4 times with 200 µL binding solution (for 10 minutes each on horizontal shaker), rinsed with distilled water, and then dried at room temperature. One µL Matrix solution (50% saturated solution of sinapinic acid in 50% acetonitrile, 0.5% trifluoroacetic acid) was added to each well and dried at room temperature. One µL Matrix solution was added and dried again. Chips were loaded automatically PBS IIc SELDI-TOF (Ciphergen, Biosystems Inc., Fremont, CA, USA) device for “surface-enhanced laser desorption/ionization time-of flight mass spectrometry” SELDI-TOF-MS analysis. For protein mass analyses the spectra were collected at 0-20 kDa range. 192 pulse rate, positive direction, and 220 intensity were used for laser application. For protein mass determination, external calibration, pure peptide standards (All-in-one peptide molecular mass standard -Ciphergen Biosystems, Inc.) were used. This is the same technique previously described by Li et al (21).
Pathology
The pathological specimens were evaluated at the pathology laboratory of Atatürk training and research hospital. In malignant subjects, the most descriptive block was selected for each subject and Estrogen receptor (ER) (Novocastra RT4-ER-6F11 7 ml, UK), Progesterone receptor (PR) (Neomarkers RM 9102-S, USA), P 53 (Dako Clone Do7; Denmark), c erbB-2 (Labvision Clone SP 3 7 ml USA), and the Ki 67 proliferation marker (Dako Clone MIB1, Denmark) were applied with the Strept-Avidin-Biotin method.
PR, P 53 and materials used for Ki 67 proliferation were applied after diluting at 1/100, 1/25 and 1/75, respectively, since they were concentrated materials. Diaminobenzidine (DAB) was used as chromogen material and Mayer's hematoxyline was used as opposite staining.
In ER, PR, and P53 evaluation, the percentage and intensity (+ → weak, 2+→moderate, 3+→ intense) of nuclear staining were considered and calculated.
Cerb B-2 was scored at 4 levels based on the membranous staining in invasive tumors:
Score 0: No staining
Score 1: Stainings not surrounding the cell membrane, the presence of which are hardly detected and which are not completely membranous (+)
Score 2: Presence of moderate staining completely surrounding cytoplasmic membrane in at least 10% of invasive carcinoma cells or presence of membranous staining in less than 30% provided that the staining is intense (++)
Score 3: Presence of intense staining surrounding the whole cytoplasmic membrane in at least 30% of invasive cells (+++)
Scores 0 and 1 were accepted as negative, and score 3 as positive. The tumors with score 2 were evaluated with fluorescence in situ hybridization (FISH) method and accepted as positive if the result was correlated by FISH method.
Ki 67 was calculated by counting the areas where the nuclear staining was the highest.
Statistical Analysis
Spectra data were transferred to a data processor and software capable of analyzing univariate statistical analysis (ProteinChip Data Manager Software). Mass calibrations of all spectra were performed internally and peak intensities were normalized according to total ion flow.
Peak aggregation and selection were performed by excluding the very low mass region (0-1500 Da) overlapping with single-photon absorptiometry (SPA) peaks. Each peak cluster was compared using the one way Mann-Whitney U test for inter-group comparisons and p values of the group were calculated. Statistical significance was set at p<0.05. Areas under the receiver-operator characteristic curve (ROC) (AUC) were calculated for each peak cluster. All peaks that present statistically significant difference in one-way statistical analysis were checked and confirmed until there were no incorrect peaks.
RESULTS
The descriptive characteristics of the groups were showed in Table 1. The BC group included 27 patients (18 invasive ductal carcinoma, 6 invasive lobular carcinoma, and 3 mixed type breast carcinoma; age range 37-73; mean age 52.6). ER was positive in 20 (74.0%), PR was positive in 18 (66.6%), and C-erb B2 was positive in 8 (29.6%) patients with immunohistochemical analysis. Breast conserving surgery was performed on 16 (59.2%) patients and the remaining 11 (40.8%) patients underwent modified radical mastectomy. The benign breast disease group included 24 patients, of whom 7 had fibrocystic disease, 3 lipoma, 3 sclerosing adenosis, 9 fibroadenoma, 1 breast abscess, and 1 fat necrosis (age range 21-57; mean age 40). The third group (the control group) included 37 healthy female subjects (age range 23-71; mean age 39.1).
Descriptive characteristics of the patients
| Breast Cancer n(%) | benign | control | |
|---|---|---|---|
| Number of patients | 27 | 24 | 37 |
| Age | |||
| Range | 37-73 | 21-57 | 23-71 |
| Median | 52.6 | 40 | 39.1 |
| Pathology | |||
| Invasive lobuler carcinoma | 18 | - | |
| Invasive ductal carcinoma | 6 | - | |
| Mixed type breast carcinoma | 3 | - | |
| Fibrocystic disease | - | 7 | - |
| Lipoma | - | 3 | - |
| Sclerosing adenosis | - | 3 | - |
| Fibroadenoma | - | 9 | - |
| Breast abscess | - | 1 | - |
| Fat necrosis | - | 1 | - |
| ER (+) | 20 (74.0%) | - | - |
| PR (+) | 18 (66.6%) | - | - |
| C-erb B2 (+) | 8 (29.6%) | - | - |
| Surgery | |||
| Excisional biopsy | 0 | 24 | - |
| Breast conserving surgery | 16 (59.2%) | - | - |
| Mastectomy | 11 (40.8%) | - | - |
Complex protein profiles of sera of 27 women with BC, 24 women with benign breast disease and 37 healthy women were obtained by SELDI-TOF MS analyses using IMAC30-NI beams. The spectra were normalized. As was expected, peaks were identified at 4.3 (Bc1), 8.1 (Bc2) and 8.9 (Bc3) kDA. The results of protein profiles and the statistical differences between the groups were showed in Tables 2.
The marker levels in the groups.
| Bc1 4300kDa | Bc2 8100 kDa | Bc3 8900 kDa | |
|---|---|---|---|
| Control (mean+sd) | 162.44±95* | 39.93+25 | 152.93+62 |
| Benign (mean+sd) | 217.9+137 | 31.46+23 | 148.29+58 |
| Malignant (mean+sd) | 250.24±167 | 97.65+101† | 185.28+95 |
*: Bc1 level is statistically lower in the control group than the others.
†: Bc2 level is significantly higher in the malignant group tan the others.
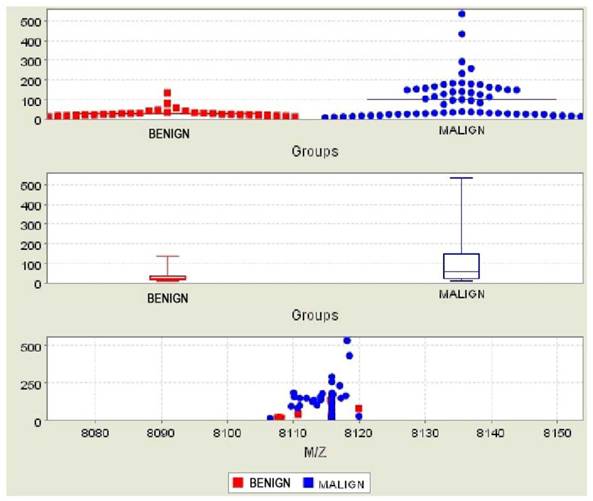
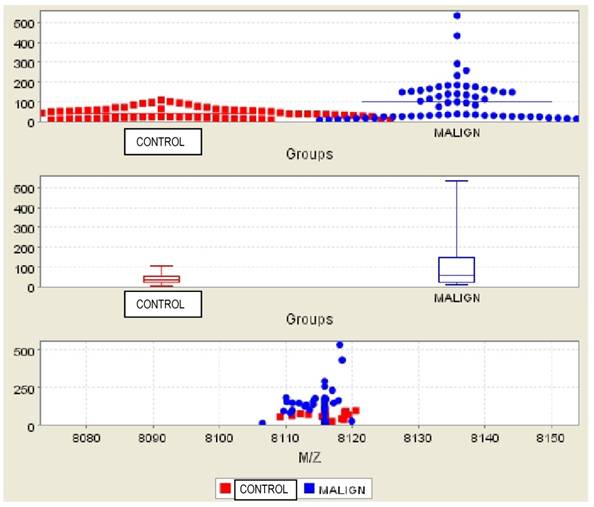
Bc2 was found significantly higher in both the comparison of malignant and benign patients (Figure 1) and malignant patients versus patients in the control group (Figure 2) (p=0.002 and p=0.003, respectively) (Table 2). The AUC values did not reach at 0.70 for the Bc2 in the groups. Bc1 was statistically significantly higher in the comparison of malignant patients to those in the control groups, as well as in the comparison of benign patients and those in the control group (p=0.006 and p=0.015, respectively). The AUC values were below the 0.70 for Bc1 in all groups. Although Bc3 was high in all malignant patients, the comparison of the benign and control groups did not yield a statistically significant difference (p=0.098 and p=0.134, respectively).
Comparison of subjects in the malignant and benign groups for Bc2
Comparison of subjects in the malignant and control groups for Bc2
DISCUSSION
Diagnosing early-stage BC before it becomes symptomatic provides the opportunity to achieve complete cure and reduces the mortality of BC. Unfortunately, the data pooled between 1992 and 1999 in the United States show that 63% of the BC patients go undiagnosed during the early-stage (22). Small lesions are frequently missed and may not be visible, even by mammography, particularly in young women and in those with dense breast tissue (23). Molecular markers that can potentially be used to identify small lesions that are invisible to imaging techniques could provide an opportunity to treat a neoplasm before it invades tissue. In particular, markers that could be detected during the ductal carcinoma in situ (DCIS) stage may prove useful, since 100% of women with BC who are diagnosed during the early stage may be treated.
Most of the molecular-based approaches investigating methods for early diagnoses of BC have specific targets such as oncogenes, tumor suppressor genes, growth factors, tumor antigens, and other gene products. These approaches, however, have poor sensitivities and specificities since none of them alone is useful for the majority of the BC and none of them is specific for cancer or breast tissues. No biomarker has been suggested to date for the early diagnosis of BC (24). Tumor markers approved by the American Food and Drug Administration (FDA) such as CA 15.3 and CA 27.29 are recommended only for monitoring of advanced or recurrent breast cancers (25).
Common “change patterns” associated with the disease status are identified using approaches that are based on genomics and proteomics instead of targeting a specific anomaly that may occur in a small subgroup of patients. Both genomic and proteomic approaches accumulate multidimensional data which may be analyzed by multivariate statistics and by powerful pattern algorithms. A possible regression in these approaches is not a direct result of corresponding pathologies but it rather tends to explore the patterns among multiple variables that may be the result of a pre-analysis of a specific series of sample. Therefore, it is more possible to obtain high classification rates in single-centered studies. An independent analysis of a separate series of sample pooled from different patient groups and hospitals is a method to evaluate the actual performance of these markers.
Jinang Li, from the John Hopkins Hospital, is the first to explore proteomics in early diagnosis of BC, who also enabled the advance of proteomics and conducted the first clinical trial in this field (26). In their first study in 2002, Li et al. investigated the 3 serum biomarkers Bc1 (4,3 kDa), Bc2 (8,1 kDa), and Bc3 (8,9 kDa) using SELDI-TOF technology in a BC group and a non-cancer control group. While Bc1 did not yield a very significant result in this study, Bc2 and Bc3 were found in increasing values. Bc3 had the highest independent diagnostic power (26). However, the patients in the study by Li et al. were categorized as malignant and control groups and women with benign breast disease and health women were in the same group. No statistical subgroup analyses were performed in this study.
In the present study, statistical analyses for Bc1, Bc2, and Bc3 were carried out individually in women with malignant disease, in those with benign breast disease and in healthy women; that is, subgroup analyses were performed. According to the results of the present study, Bc2 had the highest independent diagnostic power. There were statistically significant differences between the subjects with malignant disease and those with benign disease as well as between subjects with malignant disease and the control group (healthy women). However the AUC values were not reaching at 0.70 for the Bc2 in the groups. Bc3 was high in all malignant patients but individual comparison of Bc3 between malignant subjects and those with benign disease and between malignant subjects and healthy controls did not result in significant differences. The most interesting finding of the present study different from the three previous studies conducted on this subject relates to Bc1, which yielded a statistically significant difference between the malignant and control groups (p=0.006) as well as between the women with benign disease and the control group (p=0.015). Unfortunately again the AUC values were below the 0.70 for Bc1 in all groups. The relevance of this finding is that it was the result of the first subgroup analysis for these biomarkers. Bc1 should therefore be studied in terms of tumoral development and inflammatory response rather than malignancy.
Another study by Mathelin et al. included a total of 89 patients. Bc1a and Bc1b defined by the authors corresponded to the Bc1 and Bc3a and Bc3b corresponded to Bc3 of Li. However, the order of efficacy in the study by Mathelin et al. was Bc1a>Bc1b>Bc3b>Bc3a (27).
Li et al. conducted a study in cooperation with John Hopkins Milan National Cancer Institute which included 176 subjects. Similar to their first study, they identified significant differences for Bc3 and Bc2 (21).
The four studies including the present study conclude that the available 3 biomarkers reflect the malignant nature of the tumor rather than indicating tumoral progress, as presence of metastases or tumor diameters were not affected by the lymph node dissemination.
The results of the present study and the other three studies did not fully confirm each other. Several hypotheses may be suggested to explain these differences. First of all, Li's study was a retrospective one and therefore samples might have been prepared and converted in different ways. In the present study, the samples were treated and processed the same way and were frozen at most one hour after they were collected. As in Li's self-criticism in his evaluation of study results (21), the fact that the sera was frozen or stored for extended periods of time might have resulted in changes in the protein content, affecting especially the results of Bc1. The present study and the study by Mathelin et al. (27) demonstrated that freezing times longer than one hour results in modifications of several protein peaks. Moreover, the sera used in the present study were frozen only once. Perhaps even more importantly, the samples of the present study were completely free from hemolysis: it is well-known that hemolysis greatly ruins protein profiles. Another factor may be the differences in statistical analyses. Direct analyses on linear data were used in the present study and the study by Mathelin et al. (27). Li et al., on the other hand, used logarithmic transformation of peak intensity (25).
The most important point is that the present study performed subgroup analyses while Li et al. (26, 21) and Mathelin et al. (27) did not. Their control groups included women with benign breast disease and healthy women together, whereas the present study examined these three groups separately. Alongside BC, broad studies are being performed on ovarian, prostate, colon, lung, pancreatic, and bladder cancer with proteomics with the SELDI-TOF method (28-37).
Proteomics were listed among BC tumor markers recommendations first in the American Society of Clinical Oncology (ASCO) 2007 guidelines. Further prospective studies were recommended in this field particularly with the SELDI-TOF method (38).
In conclusion, it can not be said that proteomics studied with SELDI-TOF method for early diagnosis of BC is useful in the clinical practice. Bc2 had the highest independent diagnostic power on BC on the base of the p value. Bc1 did not yield statistical significance in the comparison of malignant subjects and the control subjects, although a statistical significance was found in the comparison of benign subjects and the control group. Bc1 should therefore be studied in terms of tumoral development and inflammatory response rather than malignancy. Although Bc3 was high in all malignant subjects, the comparison of the benign and control groups did not yield a statistically significant difference. None of the proteomics reach reasonable AUC values for the discrimination of the BC. However, larger prospective studies and subgroup analyses are needed on this subject to say that it can be used in the clinical practice.
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Li J, Zhang Z, Rosenzweig J, Wang YY, Chan DW. Proteomics and bioinformatics approaches for identification of serum biomarkers to detect breast cancer. Clin Chem. 2002;48(8):1296-1304
2. Chung M, Chang HR, Bland KI, Wanebo HJ. Younger women with breast carcinoma have a poorer prognosis than older women. Cancer. 1996;77(1):97-103
3. Jmor S, Al-Sayer H, Heys SD, Payne S, Miller I, Ah-See A, Hutcheon A, Eremin O. Breast cancer in women aged 35 and under: prognosis and survival. J R Coll Surg Edinb. 2002;47(5):693-699
4. Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365(9472):1687-1717
5. Ermilova VD. [International histological classification of breast cancer by the WHO (1968) and its prognostic value]. Arkh Patol. 1980;42(4):13-19
6. Singletary SE, Greene FL. Revision of breast cancer staging: the 6th edition of the TNM Classification. Semin Surg Oncol. 2003;21(1):53-59
7. Rosenberg RD, Hunt WC, Williamson MR, Gilliland FD, Wiest PW, Kelsey CA, Key CR, Linver MN. Effects of age, breast density, ethnicity, and estrogen replacement therapy on screening mammographic sensitivity and cancer stage at diagnosis: review of 183,134 screening mammograms in Albuquerque, New Mexico. Radiology. 1998;209(2):511-518
8. Ugnat AM, Xie L, Morriss J, Semenciw R, Mao Y. Survival of women with breast cancer in Ottawa, Canada: variation with age, stage, histology, grade and treatment. Br J Cancer. 2004;90(6):1138-43
9. Sant M, Allemani C, Capocaccia R, Hakulinen T, Aareleid T, Coebergh JW, Coleman MP, Grosclaude P, Martinez C, Bell J, Youngson J, Berrino F. Stage at diagnosis is a key explanation of differences in breast cancer survival across Europe. Int J Cancer. 2003;106(3):416-422
10. Lumachi F, Basso SM, Brandes AA, Pagano D, Ermani M. Relationship between tumor markers CEA and CA 15-3, TNM staging, estrogen receptor rate and MIB-1 index in patients with pT1-2 breast cancer. Anticancer Res. 2004;24(5):3221-3224
11. Stein RC, Zvelebil MJ. The application of 2D gel-based proteomics methods to the study of breast cancer. J Mammary Gland Biol Neoplasia. 2002;7(4):385-393
12. Hondermarck H. Breast cancer: when proteomics challenges biological complexity. Mol Cell Proteomics. 2003;2(5):281-291
13. Issaq HJ, Veenstra TD, Conrads TP, Felschow D. The SELDITOF MS approach to proteomics: protein profiling and biomarker identification. Biochem Biophys Res Commun. 2002;292(3):587-592
14. Sauter ER, Shan S, Hewett JE, Speckman P, Du Bois GC. Proteomic analysis of nipple aspirate fluid using SELDI-TOF-MS. Int J Cancer. 2005;114(5):791-796
15. Liu W, Guan M, Wu D, Zhang Y, Wu Z, Xu M, Lu Y. Using Three Analysis Pattern and SELDI-TOF-MS to Discriminate Transitional Cell Carcinoma of the Bladder Cancer from Noncancer Patients. Eur Urol. 2005;47(4):456-462
16. Wilson LL, Tran L, Morton DL, Hoon DS. Detection of differentially expressed proteins in early-stage melanoma patients using SELDI-TOF mass spectrometry. Ann N Y Acad Sci. 2004;1022:317- 322
17. Tong W, Xie Q, Hong H, Shi L, Fang H, Perkins R, Petricoin EF. Using decision forest to classify prostate cancer samples on the basis of SELDI-TOF MS data: assessing chance correlation and prediction confidence. Environ Health Perspect. 2004;112(16):1622- 1627
18. Meric-Bernstam F. Serum proteomics for BRCA1-associated breast cancer. Ann Surg Oncol. 2004;11(10):883-884
19. Gretzer MB, Chan DW, van Rootselaar CL, Rosenzweig JM, Dalrymple S, Mangold LA. et al. Proteomic analysis of dunning prostate cancer cell lines with variable metastatic potential using SELDI-TOF. Prostate. 2004;60(4):325-331
20. Laronga C, Becker S, Watson P, Gregory B, Cazares L, Lynch H, Perry RR, Wright GLJr, Drake RR, Semmes OJ. SELDI-TOF serum profiling for prognostic and diagnostic classification of breast cancers. Dis Markers. 2003;19(4-5):229-238
21. Li J, Orlandi R, White CN, Rosenzweig J, Zhao J, Seregni E, Morelli D, Yu Y, Meng X-Y, Zhang Z, Davidson NE, Fung ET, Chan DW. Independent Validation of Candidate Breast Cancer Serum Biomarkers Identified by Mass Spectrometry. Clinical Chemistry. 2005;51:2229-2235
22. Jemal A. Tiwari RC, Murray T, Ghafoor A, Samuels A, Ward E, et al. Cancer statistics, 2004. CA Cancer J Clin. 2004;54:8-29
23. Antman K, Shea S. Screening mammography under age 50. JAMA. 1999;281:1470-2
24. Smith RA, Cokkinides V, Eyre HJ. American Cancer Society guidelines for the early detection of cancer, 2004. CA Cancer J Clin. 2004;54:41-52
25. Chan DW, Sell S. Tumor markers. In: (ed.) Burtis CA, Ashwood ER. Tietz textbook of clinical chemistry, 3rd ed. Philadelphia: WB Saunders. 1999:390-413
26. Li J, Zhang Z, Rosenzweig J, Wang YY, Chan DW. Proteomics and bioinformatics approaches for identification of serum biomarkers to detect breast cancer. Clin Chem. 2002;48(8):1296-1304
27. Mathelin C. Cromer A, Wendling C, Tomasetto C, Rio MC: Serum biomarkers for detection of breast cancers: a prospective study. Breast Cancer Research and Treatment. 2006;96:83-90
28. 20. Adam BL, Qu Y, Davis JW, Ward MD, Clements MA, Cazares LH, et al. Serum protein fingerprinting coupled with a pattern-matching algorithm distinguishes prostate cancer from benign prostate hyperplasia and healthy men. Cancer Res. 2002;62:3609-14
29. Adam PJ, Boyd R, Tyson KL, Fletcher GC, Stamps A, Hudson L. et al. Comprehensive proteomic analysis of breast cancer cell membranes reveals unique proteins with potential roles in clinical cancer. J Biol Chem. 2003;278:6482-9
30. Clarke W, Silverman BC, Zhang Z, Chan DW, Klein AS, Molmenti EP. Characterization of renal allograft rejection by urinary proteomic analysis. Ann Surg. 2003;237:660-4
31. Koopmann J, Zhang Z, White N, Rosenzweig J, Fedarko N, Jagannath S. et al. Serum diagnosis of pancreatic adenocarcinoma using surface-enhanced laser desorption and ionization mass spectrometry. Clin Cancer Res. 2004;10:860-8
32. Paweletz CP, Trock B, Pennanen M, Tsangaris T, Magnant C, Liotta LA. et al. Proteomic patterns of nipple aspirate fluids obtained by SELDI-TOF: potential for new biomarkers to aid in the diagnosis of breast cancer. Dis Markers. 2001;17:301-7
33. Petricoin I, Emanuel F, Ardekani AM, Hitt BA, Levine PJ, Fusaro VA. et al. Use of proteomic patterns in serum to identify ovarian cancer. Lancet. 2002;359:572-7
34. Rosty C, Christa L, Kuzdzal S, Baldwin WM, Zahurak ML, Carnot F. et al. Identification of hepatocarcinoma-intestine-pancreas/pancreatitis- associated protein I as a biomarker for pancreatic ductal adenocarcinoma by protein biochip technology. Cancer Res. 2002;62:1868-75
35. Vlahou A, Laronga C, Wilson L, Gregory B, Fournier K, McGaughey D. et al. A novel approach toward development of a rapid blood test for breast cancer. Clin Breast Cancer. 2003;4:203-9
36. Vlahou A, Schellhammer PF, Mendrinos S, Patel K, Kondylis FI, Gong L. et al. Development of a novel proteomic approach for the detection of transitional cell carcinoma of the bladder in urine. Am J Pathol. 2001;158:1491-502
37. Vlahou A, Schorge JO, Gregory BW, Coleman RL. Diagnosis of ovarian cancer using decision tree classification of mass spectral data. J Biomed Biotechnol. 2003;2003:308-14
38. Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S, Somerfield MR, Hayes DF, Bast RCJr. American Society of Clinical Oncology 2007 Update of Recommendations for the Use of Tumor Markers in Breast Cancer. J Clin Oncol. 2007;25(33):5287- 5312
Author contact
Corresponding author: Kemal Atahan, 6342 sok. No:44 Ayşe Kaya 2 Apt. Kat:3, Daire:6 35540 Bostanlı/İzmir/TURKEY. Phone: +905324126805; Fax: +902322445624 ; e-mail: kemalatahancom.tr

 Global reach, higher impact
Global reach, higher impact