Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(6):2016-2026. doi:10.7150/ijms.130149 This issue Cite
Research Paper
Predictors of One-Year Mortality in Hospitalized Patients with Splenic Infarction: Survival Analysis of a Retrospective Cohort in Taiwan
1. Department of Emergency Medicine, Taipei Veterans General Hospital, Taipei, Taiwan.
2. Institute of Emergency and Critical Care Medicine, National Yang Ming Chiao Tung University, Taipei, Taiwan.
3. School of Medicine, College of Medicine, National Yang Ming Chiao Tung University, Taipei, Taiwan.
4. Department of Radiology, Taipei Veterans General Hospital, Taipei, Taiwan.
5. Department of Nuclear Medicine, Taipei Veterans General Hospital, Taipei, Taiwan.
6. Department of Pediatrics, Taipei Veterans General Hospital, Taipei, Taiwan.
7. Nursing Department, Taipei Veterans General Hospital, Taipei, Taiwan.
8. Institute of Clinical Nursing, National Yang Ming Chiao Tung University, Taipei, Taiwan.
9. Kinmen Hospital, Ministry of Health and Welfare, Kinmen, Taiwan.
Received 2025-12-18; Accepted 2026-4-15; Published 2026-4-23
Abstract
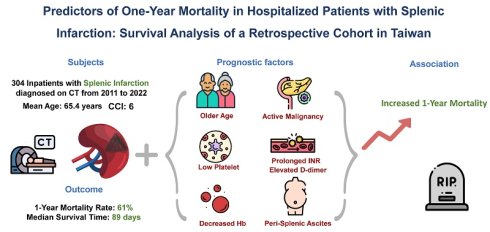
Background: Splenic infarction, an uncommon disease, has been demonstrated to be associated with substantial short-term mortality. Given the limited evidence on long-term mortality, this investigation examined the clinical and radiological characteristics of splenic infarction, its long-term prognosis, and predictors of mortality.
Methods: This retrospective cohort study at a tertiary care hospital in Taiwan enrolled adult inpatients with first-episode splenic infarction diagnosed on computed tomography from January 2011 to May 2022. The primary outcome was 1-year all-cause mortality. Kaplan-Meier analysis and multivariate weighted Cox regression were applied for survival analysis.
Results: The cohort included 304 individuals with an average age of 65.4 years. The mean CCI was 6, and active malignancy was observed in 49%. The estimated 1-year all-cause mortality rate was 61%, and the median survival time was 89 days. Compared with survivors, non-survivors exhibited significantly higher rates of left-sided pleural effusion, peri-splenic ascites, multiple or total infarction, and both portal and splenic vein thrombosis on CT imaging. Older age, active malignancy, decreased hemoglobin levels, low platelet counts, prolonged INR, elevated D-dimer, and peri-splenic ascites were independent predictors of mortality.
Conclusion: In splenic infarction, 1-year mortality is notably high. The identified independent prognostic factors included age, malignancy, laboratory findings, and radiologic features. These findings may assist clinicians in early risk stratification for hospitalized patients with splenic infarction.
Keywords: splenic infarction, survival analysis, mortality, risk factors, hospitalization
Introduction
Splenic infarction is an uncommon disease that arises from diverse conditions, most commonly arrhythmia, infection, malignancy, acute aortic syndrome, and coagulation abnormalities [1-3]. Patients typically exhibit a nonspecific presentation comprising left upper quadrant or left flank pain alongside leukocytosis, anemia, hypoalbuminemia, and elevations in lactate dehydrogenase and D-dimer [1, 4-6]. Short-term mortality after splenic infarction has been relatively well characterized, with reported rates reaching 17-33% [1-3, 7]. However, evidence on long-term prognosis remains limited.
Research regarding prognostic determinants of splenic infarction is also sparse. Our previous investigation focusing on short-term mortality in the emergency department (ED) population with splenic infarction identified uremia, malignancy, and high acuity as independent predictors [3]. Nevertheless, the prognostic validity of these short-term predictors for long-term mortality has not been established. One cohort study has reported that a history of stroke or cirrhosis is associated with higher long-term all-cause mortality, whereas anticoagulation is associated with improved survival [8]. However, these associations may lack robustness owing to the small sample size.
On contrast-enhanced computed tomography (CT), the hallmark feature is a wedge-shaped hypoattenuating parenchymal defect with its base abutting the splenic periphery. Other possible coexisting abnormalities include splenomegaly, ascites, portal vein thrombosis, splenic vein thrombosis, and infarction of other organs [9-11]. However, the CT phenotype of these patients has not been systematically characterized, and its relevance to prognosis remains underexplored.
In these cases, the leading causes of mortality were attributable predominantly to malignancy, sepsis, respiratory tract infection, and infective endocarditis [3]. While splenic infarction seldom directly accounts for death, numerous investigations have demonstrated its association with substantial subsequent mortality [1-3, 7]. Despite this, its clinical importance and prognostic implications are often underestimated by healthcare providers and investigators. Given the paucity and limitations of the existing literature, this research aims to investigate the clinical and radiological characteristics and long-term prognosis of splenic infarction, and to identify predictors of one-year mortality. We seek to enhance recognition of the clinical significance and prognostic relevance of this disease among clinicians and researchers.
Methods
Study design and setting
This was a retrospective observational cohort study conducted at Taipei Veterans General Hospital, a tertiary care medical center in Taiwan. This hospital provides approximately 2,900 beds and reports an annual total of about 820,000 inpatient bed-days. All work in this investigation adhered to the principles of the Declaration of Helsinki. Institutional Review Board 2 of this hospital approved the study (approval number 2023-01-028BC), and informed consent was waived because of the retrospective and observational nature of the study design.
Selection of participants
To screen the target population of interest, keywords related to splenic infarction were searched for in the CT report database. Inclusion criteria were hospitalized adult patients with first-episode splenic infarction diagnosed on CT during inpatient care or at the preadmission ED visit, with CT examination dates from January 2011 through May 2022. Exclusion criteria included the following: duplicate CT reports from different anatomical sites; CT performed outside the index hospitalization or the preadmission ED visit; age under 20 years; no evidence of splenic infarction; and traumatic splenic injury.
Data collection
The medical records of eligible participants were accessed for the study from March 1, 2023, to October 1, 2023. Data on patient demographics and clinical characteristics were comprehensively reviewed in the electronic health record system. Age, sex, ED visit, comorbidities, pharmacologic therapy, symptoms, laboratory tests, co-infarction of other organs during the same hospitalization, surgeries, intensive care unit (ICU) admission, lengths of ICU and hospital stay, admission date, CT diagnosis date, and the last follow-up date were retrieved. A co-author and radiologist, YK, meticulously examined all CT images, confirmed the diagnosis of splenic infarction, and extracted imaging features [9-11].
Active malignancy was defined as any of the following: (1) a new or recently diagnosed cancer; (2) receipt of oncologic therapy in the prior six months; or (3) advanced-stage disease for which treatment options are constrained or infeasible [12, 13]. The Charlson Comorbidity Index (CCI) was calculated based on their medical history [14].
Outcomes
The primary outcome was one-year all-cause mortality. For analytic purposes, subjects were grouped as survivors versus non-survivors based on the last follow-up within one year.
Statistical analysis
Continuous variables were reported as mean ± standard deviation (SD) and compared using Student's t-test. Categorical variables were presented as frequencies and percentages, with comparisons assessed by chi-square or Fisher's exact test. If missing values were present in laboratory variables, the variables were log-transformed and subjected to multiple imputation using the multivariate imputation by chained equations (MICE) framework with predictive mean matching. A total of m = 30 imputations were generated based on the proportion of missing data, with 20 iterations to ensure convergence and a fixed random seed to ensure reproducibility [15, 16]. Relevant covariates, including clinical characteristics, laboratory tests, imaging findings, treatments, and prognosis from observed cases, were incorporated to predict missing values using a regression model-based approach. The imputed values were then back-transformed, and the resulting datasets were utilized for subsequent statistical modeling and analyses.
For survival analysis, Kaplan-Meier analysis was used to estimate the one-year all-cause mortality rate and median survival time. In addition, Schoenfeld residuals were used to evaluate the proportional hazards assumption. Weighted Cox regression using the Average Hazard Ratio (AHR) weighting approach, which assigns weights based on individuals' time at risk across event times to yield a time-averaged hazard ratio accommodating non-proportional hazards, was employed to identify prognostic factors for the primary outcome [17, 18]. The time origin for survival analysis was defined as the date of CT diagnosis. A sensitivity analysis was additionally conducted using the date of hospital admission as the time origin, with adjustment for the interval from admission to CT diagnosis, to assess the robustness of the findings. For patients who received CT diagnosis in the emergency department before hospital admission, the admission-to-CT interval was defined as 0 days. In the univariate analysis, variables with p values below 0.05 were incorporated into the multivariate analysis employing backward elimination. Results were expressed in terms of hazard ratios (HRs) with 95% confidence intervals (CIs). The AHR can be interpreted as the average relative hazard over the entire follow-up period rather than a constant hazard ratio at any single time point. Because the proportional hazards assumption was not fully satisfied, AHRs provide a summary measure of the association between covariates and mortality over the follow-up period.
With respect to missing data, differences in the proportions of missing data between survivors and non-survivors were assessed using the chi-square test. Mortality rates were evaluated after stratification by missingness. Variables with missing values were analyzed prior to imputation. Furthermore, another sensitivity analysis based on complete-case Cox regression models was performed.
To address potential confounding by indication in the association between anticoagulant therapy and 1-year mortality, inverse probability of treatment weighting (IPTW) based on propensity scores was performed after multiple imputation as a sensitivity analysis. Propensity scores were estimated using baseline covariates that could influence both treatment allocation and mortality risk.
The dataset was collected with Microsoft Office Excel 2019. All statistical analyses were conducted using R (version 4.5.1) in the Google Colab environment. Multiple imputation was performed with the mice package, and survival analysis with the coxphw package. Statistical significance was defined as a two-sided p value < 0.05 in all analyses. The study was performed and documented in compliance with STROBE guidelines.
Results
During the study period, 615 individuals were initially identified as eligible through keyword screening of the CT report database, and 304 patients were included in the final cohort after exclusions (Figure 1). As shown in Table 1, the study cohort had a mean age of 65.4 years, with 63.2% male. Admissions from the ED accounted for 21.4%. Common comorbidities included hypertension (51.3%), active malignancy (49.3%), and diabetes mellitus (35.5%), with a mean CCI of 6. In comparison to survivors, non-survivors showed significantly older age, greater prevalence of diabetes mellitus and active malignancy, and higher CCI. The pancreas, hematologic cancers, liver, and colorectum were the predominant sites of active malignancy among hospitalized patients with splenic infarction (Table 2).
Study participant enrollment flowchart.
Baseline characteristics among hospitalized patients with splenic infarction
| Characteristics | All (n = 304) | Survivors (n = 126) | Non-survivors (n = 178) | p value |
|---|---|---|---|---|
| Age | 65.4 ± 16.1 | 62.4 ± 17.6 | 67.6 ± 14.8 | 0.007 |
| Male sex | 192 (63.2) | 82 (65.1) | 110 (61.8) | 0.559 |
| Admission from ED | 65 (21.4) | 32 (25.4) | 33 (18.5) | 0.151 |
| Comorbidities | ||||
| Dyslipidemia | 39 (12.8) | 16 (12.7) | 23 (12.9) | 0.954 |
| Hypertension | 156 (51.3) | 69 (54.8) | 87 (48.9) | 0.312 |
| Diabetes mellitus | 108 (35.5) | 34 (27.0) | 74 (41.6) | 0.009 |
| Coronary artery disease | 60 (19.7) | 24 (19.0) | 36 (20.2) | 0.799 |
| Valvular heart disease | 27 (8.9) | 12 (9.5) | 15 (8.4) | 0.741 |
| Myocardial infarction | 8 (2.6) | 4 (3.2) | 4 (2.2) | 0.722 |
| Congestive heart failure | 37 (12.2) | 15 (11.9) | 22 (12.4) | 0.905 |
| Active malignancy | 150 (49.3) | 38 (30.2) | 112 (62.9) | < 0.001 |
| CVA or TIA | 38 (12.5) | 12 (9.5) | 26 (14.6) | 0.187 |
| Uremia requiring renal replacement therapy | 27 (8.9) | 8 (6.3) | 19 (10.7) | 0.192 |
| Liver cirrhosis | 35 (11.5) | 14 (11.1) | 21 (11.8) | 0.853 |
| COPD | 19 (6.3) | 9 (7.1) | 10 (5.6) | 0.588 |
| PAOD | 12 (3.9) | 4 (3.2) | 8 (4.5) | 0.767 |
| CCI | 6 ± 4 | 5 ± 3 | 7 ± 3 | < 0.001 |
| Medications use before hospitalization | ||||
| Antiplatelet drugs | 48 (15.8) | 24 (19.0) | 24 (13.5) | 0.190 |
| Anticoagulants | 31 (10.2) | 18 (14.3) | 13 (7.3) | 0.047 |
ED: emergency department; CVA: cerebrovascular accident; TIA: transient ischemic attack; COPD: chronic obstructive pulmonary disease; PAOD: peripheral arterial occlusive disease; CCI: Charlson Comorbidity Index
Location of active malignancy among hospitalized patients with splenic infarction
| Location | All (n = 150) | Survivors (n = 38) | Non-survivors (n = 112) | p value |
|---|---|---|---|---|
| Head and neck1 | 5 (3%) | 1 (3%) | 4 (4%) | 1.000 |
| Esophagus | 4 (3%) | 3 (8%) | 1 (1%) | 0.050 |
| Stomach | 6 (4%) | 2 (5%) | 4 (4%) | 0.643 |
| Small intestine | 2 (1%) | 1 (3%) | 1 (1%) | 0.444 |
| Colon or rectum | 13 (9%) | 6 (16%) | 7 (6%) | 0.094 |
| Liver3 | 17 (11%) | 3 (8%) | 14 (13%) | 0.562 |
| Biliary tract cancer | 10 (7%) | 2 (5%) | 8 (7%) | 1.000 |
| Ampulla of Vater | 3 (2%) | 1 (3%) | 2 (2%) | 1.000 |
| Pancreas | 30 (20%) | 5 (13%) | 25 (22%) | 0.222 |
| Urinary tract | 6 (4%) | 2 (5%) | 4 (4%) | 0.643 |
| Prostate | 4 (3%) | 1 (3%) | 3 (3%) | 1.000 |
| Uterus or ovary | 8 (5%) | 5 (13%) | 3 (3%) | 0.025 |
| Leukemia2 | 9 (6%) | 0 (0%) | 9 (8%) | 0.112 |
| Lymphoma1 | 18 (12%) | 6 (16%) | 12 (11%) | 0.398 |
| Multiple myeloma | 2 (1%) | 0 (0%) | 2 (2%) | 1.000 |
| Lung3 | 11 (7%) | 0 (0%) | 11 (10%) | 0.066 |
| Others (skin, soft tissue, vessel)2 | 4 (3%) | 0 (0%) | 4 (4%) | 0.572 |
| Unknown primary origin | 1 (1%) | 0 (0%) | 1 (1%) | 1.000 |
1One patient had concurrent tonsillar cancer and lymphoma.
2Another patient had concurrent fibromyxosarcoma of the limb and acute myeloid leukemia.
3Another patient had concurrent lung cancer and hepatocellular carcinoma.
The Kaplan-Meier analysis indicated an estimated 1-year all-cause mortality of 61% (95% CI: 55-66%), with a median survival time of 89 days (Figure S1). With respect to etiology and associated conditions, non-survivors exhibited higher proportions of active malignancy and sepsis, but lower proportions of acute aortic syndrome and unknown causes (Table 3).
Etiology and associated conditions of splenic infarction
| Etiology and associated conditions | All (n = 304) | Survivors (n = 126) | Non-survivors (n = 178) | p value |
|---|---|---|---|---|
| Active malignancy1 | 150 (49.3) | 38 (30.2) | 112 (62.9) | <0.001 |
| Abdominal solid tumor | 99 (32.6) | 28 (22.2) | 71 (39.9) | 0.001 |
| Non-abdominal solid tumor | 25 (8.2) | 4 (3.2) | 21 (11.8) | 0.007 |
| Hematologic malignancy | 29 (9.5) | 6 (4.8) | 23 (12.9) | 0.017 |
| Cardioembolic diseases | 98 (32.2) | 39 (31.0) | 59 (33.1) | 0.687 |
| Atrial fibrillation | 34 (11.2) | 15 (11.9) | 19 (10.7) | 0.737 |
| Infective endocarditis | 17 (5.6) | 10 (7.9) | 7 (3.9) | 0.134 |
| Others2 | 82 (27.0) | 32 (25.4) | 50 (28.1) | 0.602 |
| Sepsis3 | 68 (22.4) | 19 (15.1) | 49 (27.5) | 0.010 |
| Nonmalignant abdominal conditions4 | 118 (38.8) | 41 (32.5) | 77 (43.1) | 0.059 |
| Hypercoagulable state5 | 17 (5.6) | 10 (7.9) | 7 (3.9) | 0.134 |
| Atherosclerosis of abdominal aorta, celiac trunk, or splenic artery | 24 (7.9) | 14 (11.1) | 10 (5.6) | 0.080 |
| Aortic dissection or aneurysm | 24 (7.9) | 19 (15.1) | 5 (2.8) | <0.001 |
| Iatrogenic6 | 22 (7.2) | 6 (4.8) | 16 (9.0) | 0.161 |
| Autoimmune disease | 11 (3.6) | 4 (3.2) | 7 (3.9) | 1.000 |
| Unknown cause | 11 (3.6) | 10 (7.9) | 1 (0.6) | <0.001 |
1 Three patients had concurrent malignancies: (1) tonsillar cancer and lymphoma; (2) fibromyxosarcoma of the limb and acute myeloid leukemia; and (3) lung cancer and hepatocellular carcinoma.
2 Other cardioembolic diseases included arrhythmias, severe heart failure, myocardial infarction, nonbacterial thrombotic endocarditis, and pulmonary arterial hypertension.
3 Sepsis in this table excluded infections limited to infective endocarditis and intra-abdominal sources.
4 Nonmalignant abdominal conditions included pancreatitis, liver cirrhosis, bowel ischemia, recent abdominal surgery, severe gastrointestinal bleeding, severe intra-abdominal bleeding, peritonitis, severe colitis, cholangitis, and cholecystitis.
5 Hypercoagulable states included antithrombin III deficiency, myeloproliferative neoplasms, essential thrombocytosis, thrombotic thrombocytopenic purpura, and simultaneous vascular thrombosis (e.g., splenic vein thrombosis, portal vein thrombosis, deep vein thrombosis, and pulmonary embolism) in the absence of other identifiable causes.
6 Iatrogenic causes included recent aortic intervention, extracorporeal membrane oxygenation (ECMO), or embolization of the celiac trunk or its branches.
Among cases, 7.2% experienced left upper quadrant abdominal pain and 34.2% reported abdominal pain in other locations. Fever (26.6%) and nausea/vomiting (14.1%) were also observed. Symptom distributions did not differ significantly between groups (Table 4).
Clinical manifestations among hospitalized patients with splenic infarction
| Variables | All (n = 304) | Survivors (n = 126) | Non-survivors (n = 178) | p value |
|---|---|---|---|---|
| Symptoms | ||||
| Abdominal pain in LUQ location | 22 (7.2) | 13 (10.3) | 9 (5.1) | 0.081 |
| Abdominal pain in other location | 104 (34.2) | 46 (36.5) | 58 (32.6) | 0.477 |
| Left flank pain | 10 (3.3) | 3 (2.4) | 7 (3.9) | 0.531 |
| Fever | 81 (26.6) | 32 (25.4) | 49 (27.5) | 0.679 |
| Nausea or vomiting | 43 (14.1) | 17 (13.5) | 26 (14.6) | 0.784 |
| Laboratory examination | ||||
| White blood cell count (/μL) | 12562 ±11341 | 12193 ± 11038 | 12823 ± 11574 | 0.631 |
| Hemoglobin (g/dL) | 10.0 ± 2.5 | 10.8 ± 2.5 | 9.4 ± 2.2 | <0.001 |
| Platelet (x103/μL) | 180 ± 155 | 233 ± 182 | 142 ± 120 | <0.001 |
| INR | 1.4 ± 0.8 | 1.2 ± 0.4 | 1.5 ± 1.0 | <0.001 |
| APTT (seconds) | 35 ± 20 | 33 ± 16 | 37 ± 22 | 0.099 |
| D-dimer (μg/mL) | 13.1 ± 19.8 | 9.4 ± 14.9 | 15.8 ± 22.2 | 0.014 |
| Albumin (g/dL) | 3.0 ± 0.6 | 3.2 ± 0.6 | 2.9 ± 0.6 | <0.001 |
| BUN (mg/dL) | 30 ± 27 | 26 ± 21 | 33 ± 30 | 0.011 |
| Creatinine (mg/dL) | 1.5 ± 1.6 | 1.6 ± 1.8 | 1.5 ± 1.4 | 0.421 |
| Total bilirubin (mg/dL) | 2.9 ± 5.1 | 1.9 ± 3.9 | 3.6 ± 5.7 | 0.003 |
| Lactate dehydrogenase (IU/L) | 590 ± 1083 | 544 ± 1025 | 623 ± 1118 | 0.559 |
| C-reactive protein (mg/dL) | 12.1 ± 42.6 | 8.1 ± 9.9 | 14.9 ± 54.5 | 0.124 |
| Findings in computed tomography | ||||
| Left-sided pleural effusion | 189 (62.2) | 69 (54.8) | 120 (67.4) | 0.025 |
| Peri-splenic ascites | 146 (48.0) | 50 (39.7) | 96 (53.9) | 0.014 |
| Infarction pattern | 0.027 | |||
| Single splenic infarction | 96 (31.6) | 50 (39.7) | 46 (25.8) | |
| Multiple splenic infarction | 193 (63.5) | 72 (57.1) | 121 (68.0) | |
| Total splenic infarction | 15 (4.9) | 4 (3.2) | 11 (6.2) | |
| Infarction size (largest diameter, mm) | 43 ± 27 | 44 ± 31 | 43 ± 25 | 0.776 |
| Spleen size | ||||
| Axial (mm) | 111 ± 29 | 113 ± 30 | 110 ± 28 | 0.335 |
| Coronal oblique (mm) | 115 ± 34 | 119 ± 41 | 112 ± 28 | 0.121 |
| Cirrhotic liver | 73 (24.0) | 24 (19.0) | 49 (27.5) | 0.088 |
| Main portal vein thrombosis | 22 (7.2) | 4 (3.2) | 18 (10.1) | 0.021 |
| Splenic vein thrombosis | 32 (10.5) | 5 (4.0) | 27 (15.2) | 0.002 |
| Spleen rupture | 2 (0.7) | 1 (0.8) | 1 (0.6) | 1.000 |
| Admission-to-CT Interval (days) | 11.8 ± 21.2 | 7.9 ± 11.8 | 14.5 ± 25.5 | 0.002 |
LUQ: left upper quadrant; INR: international normalized ratio; APTT: activated partial thromboplastin time; BUN: Blood urea nitrogen; CT: computed tomography
Laboratory variables, including INR, APTT, D-dimer, albumin, BUN, total bilirubin, lactate dehydrogenase, and C-reactive protein, exhibited missing values. The proportion of missing data was highest for D-dimer, followed by lactate dehydrogenase and C-reactive protein. The proportion of missing albumin data was significantly higher in survivors than in non-survivors (p < 0.001), whereas no significant differences were observed for the other variables with missing values (Table S1 and Table S2). After multiple imputation, compared with survivors, non-survivors were characterized by lower levels of hemoglobin, platelet count, and albumin, and higher levels of INR, D-dimer, BUN, and total bilirubin (Table 4). In addition, an analysis of missing laboratory data prior to imputation is provided in Table S3, which showed similar findings and between-group differences compared with the results after multiple imputation.
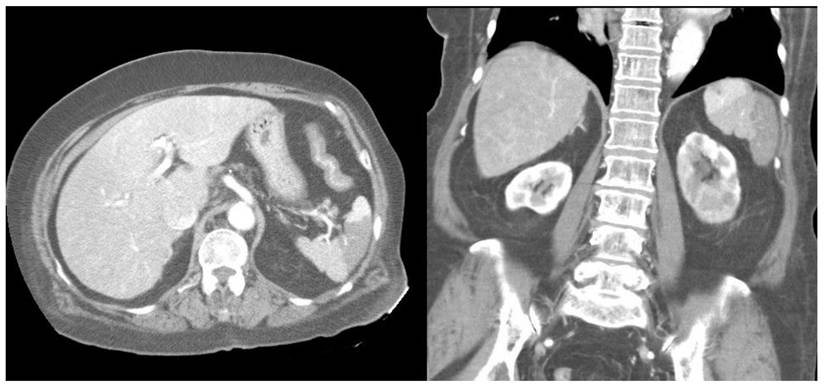
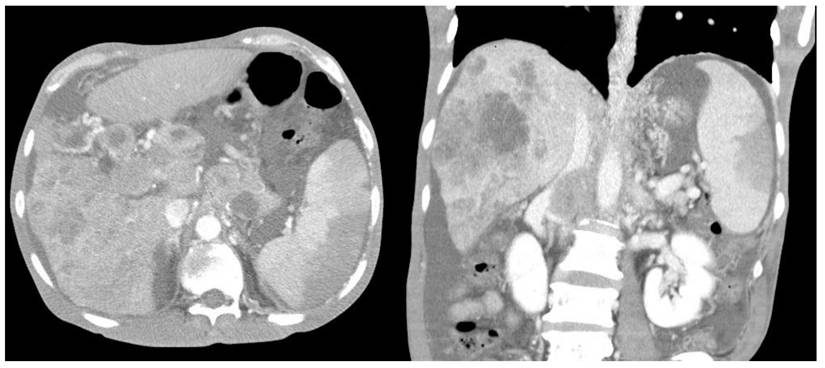
Furthermore, compared with survivors, the non-survivors exhibited significantly higher rates of left-sided pleural effusion, peri-splenic ascites, multiple or total infarction, main portal vein thrombosis, and splenic vein thrombosis on CT imaging. Co-infarctions most commonly involved the kidneys, brain, and intestines. At least one organ co-infarction occurred in 30.6% of cases, while 11.5% experienced two or more, but no significant group differences were observed (Table S4). Figure 2 shows concurrent splenic and left renal infarction, whereas Figure 3 shows splenic infarction with portal vein thrombosis and ascites in a patient with hepatocellular carcinoma.
Concurrent splenic and left renal infarction. Contrast-enhanced abdominal CT images showing wedge-shaped hypodense regions within the spleen on the axial view (left panel) and within both the spleen and the left kidney on the coronal view (right panel).
Splenic infarction in a patient with hepatocellular carcinoma. Contrast-enhanced abdominal CT reveals a wedge-shaped splenic hypodensity consistent with infarction, an ill-defined heterogeneous hypodense mass in the right hepatic lobe, portal vein thrombosis, and ascites.
During hospitalization, the survivor group received more anticoagulants (Table 5). Overall, three patients (1%) underwent splenectomy. Study participants demonstrated prolonged ICU and hospital stays averaging 13 and 50 days, respectively. The leading causes of death included solid tumor malignancy (46%), hematologic malignancy (11%), sepsis or infectious disease (15%), intestinal or colonic ischemia (6%), and acute myocardial infarction or heart failure (5%) (Table S5).
Therapy and prognosis among hospitalized patients with splenic infarction
| Variables | All (n = 304) | Survivors (n = 126) | Non-survivors (n = 178) | p value |
|---|---|---|---|---|
| Pharmacologic therapy* | ||||
| Anticoagulants | 71 (23.4) | 37 (29.4) | 34 (19.1) | 0.037 |
| Antiplatelet agents | 87 (28.6) | 41 (32.5) | 46 (25.8) | 0.203 |
| Analgesics | 179 (58.9) | 66 (52.4) | 113 (63.5) | 0.053 |
| Splenectomy | 3 (1.0) | 3 (2.4) | 0 (0.0) | 0.070 |
| Length of stay | ||||
| ICU (days) | 13 ± 32 | 10 ± 20 | 16 ± 39 | 0.067 |
| Hospitalization (days) | 50 ± 216 | 65 ± 329 | 39 ± 55 | 0.389 |
*Medications encompassed both the patients' existing treatments and newly initiated pharmacological therapies.
ICU: intensive care unit
The global test for the proportional hazards assumption, assessed using Schoenfeld residuals, was significant (p = 0.001), indicating that the assumption may not hold for the overall model. Several variables, including active malignancy, total bilirubin, splenic vein thrombosis, peri-splenic ascites, and anticoagulant use, showed evidence of violation of the proportional hazards assumption (Table S6). Weighted Cox regression using the AHR weighting approach was therefore performed to address the violation of the proportional hazards assumption. Univariate weighted Cox regression revealed several prognostic factors for 1-year mortality in splenic infarction. Multivariate weighted Cox regression analysis identified independent risk factors, including older age, active malignancy, decreased hemoglobin levels, low platelet counts, prolonged INR, elevated D-dimer, and peri-splenic ascites (Table 6).
Weighted Cox regression analysis of factors associated with 1-year mortality among hospitalized patients with splenic infarction
| Predictive Variables | Univariate analysis | Multivariate analysis* | ||
|---|---|---|---|---|
| HR** (95% CI) | p value | HR** (95% CI) | p value | |
| Age | 1.011 (1.002-1.020) | 0.021 | 1.019 (1.008-1.030) | < 0.001 |
| Diabetes mellitus | 1.327 (0.985-1.788) | 0.063 | ||
| Active malignancy | 1.855 (1.354-2.542) | < 0.001 | 1.873 (1.360-2.580) | < 0.001 |
| Modified CCI*** | 1.055 (1.003-1.109) | 0.038 | ||
| Sepsis | 1.402 (1.019-1.930) | 0.038 | ||
| Unknown cause | 0.170 (0.022-1.310) | 0.089 | ||
| Hemoglobin | 0.869 (0.816-0.927) | < 0.001 | 0.911 (0.852-0.974) | 0.006 |
| Platelet | 0.997 (0.995-0.998) | < 0.001 | 0.997 (0.996-0.999) | < 0.001 |
| INR | 1.214 (1.075-1.371) | 0.002 | 1.198 (1.063-1.349) | 0.003 |
| D-dimer | 1.011 (1.003-1.019) | 0.007 | 1.010 (1.002-1.017) | 0.010 |
| Albumin | 0.651 (0.514-0.823) | < 0.001 | ||
| BUN | 1.007 (1.003-1.011) | 0.001 | ||
| Total bilirubin | 1.052 (1.018-1.088) | 0.003 | ||
| Left-sided pleural effusion | 1.546 (1.125-2.123) | 0.007 | ||
| Peri-splenic ascites | 1.740 (1.294-2.341) | < 0.001 | 1.714 (1.275-2.305) | < 0.001 |
| Multiple splenic infarction | 1.507 (1.068-2.127) | 0.020 | ||
| Total splenic infarction | 2.856 (1.337-6.136) | 0.007 | ||
| Main portal vein thrombosis | 1.814 (1.137-2.894) | 0.012 | ||
| Splenic vein thrombosis | 1.294 (0.931-1.800) | 0.125 | ||
| Anticoagulant therapy | 0.747 (0.504-1.108) | 0.147 | ||
* Variables with p < 0.05 in univariate analyses were entered into multivariate analysis using backward elimination.
**HR denotes the average hazard ratio (AHR) estimated using weighted Cox regression that accommodates potential non-proportional hazards.
***Because malignancy is included in the CCI, a modified CCI was calculated by excluding malignancy-related components to avoid conceptual overlap and potential collinearity with the active malignancy variable in the model.
CI: confidence interval; INR: international normalized ratio; BUN: blood urea nitrogen
In the sensitivity analysis using hospital admission as the time origin and adjusting for diagnostic delay, the main findings remained largely consistent. Active malignancy, age, platelet count, INR, D-dimer, and peri-splenic ascites remained significant predictors of 1-year mortality. In addition, BUN, multiple splenic infarctions, and total splenic infarction emerged as significant factors, whereas hemoglobin became insignificant (Table S7). In another sensitivity analysis using complete-case Cox regression models, active malignancy, hemoglobin, BUN, platelet count, and peri-splenic ascites were significantly associated with 1-year mortality (Table S8).
The unadjusted complete-case Cox regression and unadjusted weighted Cox regression using hospital admission as the time origin both identified anticoagulant use as a significant factor (HR 0.685, 95% CI 0.471-0.996, and HR 0.657, 95% CI 0.448-0.963, respectively). However, anticoagulant use was no longer statistically significant (HR 0.747, 95% CI 0.504-1.108) in the unadjusted weighted Cox regression when the CT diagnosis date was used as the time origin. Furthermore, IPTW analysis based on propensity scores after multiple imputation also showed no significant association (HR 1.143, 95% CI 0.758-1.720) (Table S9).
Discussion
This retrospective cohort study demonstrated the clinical manifestations of splenic infarction in hospitalized patients, with a 1-year all-cause mortality rate of 61%. Multivariate weighted Cox regression analysis identified several independent mortality predictors, including older age, active malignancy, decreased hemoglobin levels, low platelet counts, prolonged INR, elevated D-dimer, and peri-splenic ascites.
A remarkably high 1-year mortality rate (61%) was observed in this study, together with prolonged ICU and hospital stays. These findings are likely attributable to multiple comorbidities (mean CCI 6) and a high prevalence of active malignancy (49.3%) in this cohort. Consistent with our findings, markedly high short-term mortality has been documented in several published articles [1-3, 7]. The long-term mortality rate was described as relatively low, at 6.86 per 100 patient-years, in a multicenter study in Taiwan; however, that cohort excluded cases with active malignancy and hematologic illness and enrolled relatively younger participants than ours (61.0 vs. 65.4 years) [8]. In addition, the leading causes of death, as shown in Table S5, were malignancy, severe infection, bowel ischemia, and acute heart disease among patients with splenic infarction. In this context, splenic infarction may act as a sentinel sign of catastrophic systemic microcirculatory failure or advanced abdominal conditions, rather than the direct cause of death.
Regarding the location of active malignancy, Table 2 shows that hepatobiliary, ampullary, and pancreatic malignancies accounted for about 40% of cases. This distribution may be related to local vascular compression caused by advanced tumors or regional lymphadenopathy. Besides, the pathophysiology of malignancy-related hypercoagulability (Trousseau syndrome) involves tumor cell-induced tissue factor production, mucin-producing activity, P- and L-selectin-mediated platelet-rich micro-thrombosis, tissue hypoxia, and oncogene activation, ultimately resulting in thrombin generation and fibrin deposition [19, 20]. Both arterial and venous thromboembolism are prevalent in patients with cancer, a condition referred to as cancer-associated thrombosis (CAT), especially for pancreatic cancer, gastric cancer, lung cancer, and lymphoma. This thromboembolic diathesis significantly leads to morbidity and mortality [19, 21].
The classification of etiologies and associated conditions in Table 3 was based on published landmark literature and incorporated comorbidities, active illnesses, and imaging findings [1, 2, 4, 22]. Apart from active malignancy, other etiologies and associated conditions, including cardioembolic diseases, sepsis, nonmalignant abdominal conditions, acute aortic syndromes or aneurysms, and connective tissue diseases, also appear to play important roles in patients with splenic infarction. In addition, because patients with acute aortic syndrome who survived to hospitalization may be subject to confounding by indication, this variable was not included in the regression analysis.
Our weighted Cox regression model demonstrated active malignancy as an independent predictor of mortality. Active malignancy was most commonly located in the pancreas, hematologic system, liver, and colorectum. Splenic infarction is possibly attributable to hypercoagulation or local invasion into the hepatobiliary system or splenic vasculature among cancer patients [10, 23, 24]. Consistently, malignancy has also been described as a prognostic factor for short-term mortality, and this research extended the evidence by validating its relevance to long-term prognosis [3, 22]. Because malignancy is included in the CCI, a modified CCI was calculated by excluding malignancy-related components to avoid conceptual overlap and potential collinearity with the active malignancy variable in the regression models. After adjustment for the modified CCI, active malignancy remained an independent prognostic factor. Accordingly, our findings indicate that cancer patients presenting with splenic infarction should be considered for earlier prognostic evaluation, with timely management of the underlying malignancy may help improve clinical outcomes.
Given that the proportional hazards assumption was not satisfied for several variables in this study, a weighted Cox regression model using the AHR weighting approach was employed. This approach yields a time-averaged hazard ratio, enabling valid inference under non-proportional hazards. AHRs were used to summarize the association between covariates and mortality over the entire 1-year follow-up period. For instance, active malignancy was associated with an AHR of 1.873, indicating that patients with active malignancy had an approximately 1.9-fold higher average hazard of death during the 1-year follow-up period. Consequently, the reported hazard ratios represent weighted averages over the full follow-up period, rather than assuming a constant effect over time. While this approach improves model validity, the temporal patterns of risk associated with certain variables may not be fully captured. Future well-designed studies incorporating time-dependent covariates or flexible survival models are warranted to better elucidate these dynamic relationships.
The missingness of albumin differed significantly between survivors and non-survivors, suggesting that its missing mechanism may not be completely random. This may reflect clinical practice in which albumin is more frequently measured in critically ill patients. This suggests that albumin has clinical importance. Although the assumption of missing at random (MAR) may not fully hold for this variable, the missingness of most other variables can reasonably be considered consistent with the MAR assumption. Given that albumin is likely associated with disease severity, its missingness may introduce bias. Nevertheless, the use of multiple imputation to handle missing values and the inclusion of these variables as potential prognostic factors in multivariable regression for adjustment are considered appropriate. Complete-case analysis may reduce statistical power and introduce selection bias due to missing data, whereas multiple imputation makes more efficient use of available data. The analysis of laboratory data following multiple imputation yielded results consistent with those observed prior to imputation, particularly regarding between-group differences and statistical significance (Table S3), thereby supporting the validity of the multiple imputation approach. In the sensitivity analysis using a complete-case Cox regression model (Table S8), the overall findings were generally consistent with the main results from the multivariate weighted Cox regression model (Table 6), supporting the robustness of our findings.
Low platelet counts, prolonged INR, and elevated D-dimer were identified as independent prognostic factors in this investigation. Taken together, these three factors suggest coagulopathy, with a tendency toward liver failure or disseminated intravascular coagulation (DIC) [25]. With elevated serum total bilirubin (mean 2.9 mg/dL) and a high prevalence of ascites (41.8%) on CT imaging in our cohort, liver decompensation might be considered one of the predictors. Nevertheless, serum total bilirubin was not an independent risk factor in our final model, and the role of liver failure remains to be clarified. DIC, frequently induced by critical illnesses such as trauma, malignancy, sepsis, and obstetric complications, results in the paradoxical manifestation of both thrombosis and bleeding owing to coagulation factor consumption and leads to a critical clinical condition and poor prognosis [26, 27].
Anticoagulant use has been reported to be associated with reduced long-term mortality in patients with splenic infarction in prior research [8], which is consistent with our findings from unadjusted complete-case Cox regression and unadjusted weighted Cox regression using hospital admission as the time origin. However, this apparent association in these models may be subject to bias, including missing data, selection bias, immortal time bias, and confounding by indication. To reduce potential bias related to missing data, selection bias, and immortal time bias, we used the CT diagnosis date as the time origin after multiple imputation. In this analysis, unadjusted weighted Cox regression showed no significant association between anticoagulant use and 1-year mortality. Patients receiving anticoagulants may have had more favorable baseline characteristics, such as lower disease severity or reduced bleeding risk, which could contribute to better outcomes; thus, confounding by indication remains a potential concern. Therefore, IPTW based on propensity scores was applied as a sensitivity analysis to better estimate the effect of anticoagulation on survival, and no statistically significant association was observed. This analysis supports the robustness of our main finding that anticoagulant use was not an independent prognostic factor.
In the sensitivity analysis using the date of hospital admission as the time origin and adjusting for the interval from hospital admission to CT diagnosis (Table S7), the main findings remained largely consistent, supporting the robustness of the identified prognostic factors in the primary analysis. Although non-survivors exhibited a longer admission-to-CT interval in descriptive analyses (Table 4), this association was reversed in time-to-event analyses, including univariate weighted Cox regression. This discrepancy likely arises from differences in analytical approaches, as descriptive analyses ignore event timing and censoring, whereas Cox regression accounts for time-to-event dynamics and risk sets. The admission-to-CT interval varied across patients and likely reflected differences in clinical severity. Patients with more severe presentations may undergo earlier imaging, whereas those with less specific or milder symptoms may experience delayed diagnosis. Nevertheless, the association between hemoglobin level and mortality was attenuated after accounting for diagnostic delay, suggesting potential confounding by diagnostic timing and disease severity. Patients with severe anemia may undergo earlier imaging, potentially introducing bias if not accounted for. The identification of multiple or total splenic infarction as significant predictors in the sensitivity analysis may also reflect improved adjustment for disease severity through inclusion of the admission-to-CT interval. These findings highlight the importance of accounting for diagnostic timing in observational studies of acute conditions and may help explain the observed differences in certain variables between the primary and sensitivity analyses, particularly given the influence of time-to-event dynamics and potential confounding.
Our results revealed no significant association between fatal outcomes and serum creatinine, while a significant association was observed with serum BUN in the sensitivity analysis. Previous evidence has suggested that a higher BUN-to-creatinine ratio may be linked to longer ICU stays and increased in-hospital mortality [28, 29]. Serum BUN elevation could primarily represent muscle protein catabolism in the context of prolonged critical illness, but it may also arise from dehydration, gastrointestinal hemorrhage, severe infection, or impaired renal and hepatic function [29, 30]. Cardiorenal syndrome may also contribute to increased circulating BUN levels. In severe heart disease or cardiac decompensation, reduced cardiac output and impaired renal perfusion may activate the renin-angiotensin-aldosterone system and stimulate the sympathetic nervous system. These neurohormonal responses enhance renal tubular urea reabsorption, which may subsequently result in elevated serum BUN levels [31]. The comorbidities and associated diseases of splenic infarction—such as cancer, infective endocarditis, sepsis, and severe heart failure—may help elucidate the underlying causes of BUN elevation [3]. Furthermore, this finding emphasizes that splenic infarction in the context of severe underlying disease should prompt increased clinical awareness and monitoring.
Our study comprehensively reviewed the radiological features of splenic infarction on CT. Our observations were consistent with prior research, demonstrating comparable prevalences of peri-splenic ascites, left-sided pleural effusion, and multiple infarcts [6]. In univariate weighted Cox regression analysis, left-sided pleural effusion, peri-splenic ascites, multiple or total splenic infarction, and main portal vein thrombosis were significant. Furthermore, our research demonstrated that the presence of peri-splenic ascites was significantly and independently correlated with mortality after adjustment. The presence of ascites typically indicates an underlying advanced systemic or local disorder, pointing to critical derangements in fluid homeostasis, portal hypertension, or peritoneal integrity, primarily including cirrhosis, malignant ascites, right-sided heart failure, and nephrotic syndrome [32]. Consequently, the prognostic relevance of this radiological finding highlights the critical role of peri-splenic ascites in splenic infarction and emphasizes it as a key feature warranting attention in future imaging evaluations.
In this study, concomitant infarction of other organs was observed in 30.6% of patients with splenic infarction during the same hospitalization, particularly renal infarction, mesenteric ischemia, and cerebral infarction. Additionally, 11.5% of patients had infarctions in two or more other organs. The high prevalence of co-infarction observed in our study is consistent with prior literature [4, 9]. These concurrent thrombotic events may indicate systemic embolization or a thrombotic storm and may be attributable to severe sepsis, DIC, advanced malignancy, or aortic dissection [2, 13, 33].
Although conducted at a single medical center in Taiwan, this research enrolled 304 individuals over approximately 12 years. To our knowledge, it represents the largest comprehensively analyzed cohort to date. Bewersdorf et al focused on 281 cancer patients out of 591 splenic infarction cases screened in the USA [23]. Of 353 splenic infarction patients identified in Korea, Im and colleagues concentrated on 101 infected subjects [33]. Furthermore, our investigation thoroughly reviewed clinical and radiological characteristics, addressed long-term prognosis, and examined predictors of mortality after multiple imputation and multivariate adjustment. For clinical application, early risk identification may be considered for patients characterized by advanced age, active malignancy, coagulopathy, anemia, or ascites. From a research perspective, additional prospective multicenter investigations are required to explore reversible prognostic factors for splenic infarction and to improve patient outcomes.
This research had several limitations. First, this study was retrospective in design and involved missing laboratory data. The missingness of certain laboratory variables, particularly albumin, may not fully satisfy the assumption of missing at random, thereby introducing potential bias even after multiple imputation. Second, eligible participants were derived exclusively from a single hospital. Third, this study focused on hospitalized patients with CT-confirmed splenic infarction, excluding outpatients as well as mild or asymptomatic cases that may have been managed conservatively in the ED settings without hospital admission or imaging confirmation. Consequently, our study population may represent patients with relatively more severe clinical presentations, which may limit the generalizability of our findings to the broader population with splenic infarction. Given the above limitations, well-designed multicenter prospective studies with comprehensive data collection are warranted to minimize bias, identify key prognostic factors, and enhance the generalizability.
Conclusion
In summary, the 1-year mortality rate among hospitalized adult inpatients with splenic infarction was considerably high, reaching 61%. For clinical practice, early risk assessment and prognostic counseling should be considered for hospitalized patients with advanced age, active malignancy, coagulopathy, anemia, or peri-splenic ascites. For investigators, prospective multicenter studies with robust designs are warranted to enable comprehensive analysis, identify modifiable factors, and ultimately improve clinical outcomes.
Abbreviations
ED: emergency department; CT: computed tomography; ICU: intensive care unit; CCI: Charlson comorbidity index; SD: standard deviation; MICE: multivariate imputation by chained equations; AHR: average hazard ratio; HR: hazard ratio; CI: confidence interval; STROBE: strengthening the reporting of observational studies in epidemiology; INR: international normalized ratio; BUN: blood urea nitrogen; DIC: disseminated intravascular coagulation.
Supplementary Material
Supplementary figure and tables.
Acknowledgements
We gratefully acknowledge the Department of Emergency Medicine at Taipei Veterans General Hospital, Taiwan, for its generous support and permission to conduct this study. We also extend our sincere gratitude to Miss Yi-Wen Liu for her dedicated support of this research.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
Author contributions
J-WL: conception and study design, recruitment of participating patients, data collection, statistical analysis, manuscript drafting and revision. YK: conception and study design, recruitment of participating patients, data collection. M-JJ: conception and study design, result validation. C-CT: recruitment of participating patients, data collection. C-KH: statistical analysis. T-FH: statistical advice, result validation. DH-TY: statistical advice, result validation. H-HH: conception and study design, statistical analysis, result validation, manuscript drafting and revision. All authors approved the final version to be published and confirmed their accountability for the study.
Ethics approval
Institutional Review Board 2 of Taipei Veterans General Hospital approved the study (approval number 2023-01-028BC).
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work the authors used ChatGPT (OpenAI) in order to enhance the manuscript's readability. After using this tool, the authors have reviewed and edited the content as needed and take full responsibility for the content of the publication.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Hakoshima M, Kitakaze K, Adachi H, Katsuyama H, Yanai H. Clinical, Hematological, Biochemical and Radiological Characteristics for Patients with Splenic Infarction: Case Series with Literature Review. J Clin Med Res. 2023;15:38-50
2. Brett AS, Azizzadeh N, Miller EM, Collins RJ, Seegars MB, Marcus MA. Assessment of Clinical Conditions Associated with Splenic Infarction in Adult Patients. JAMA Intern Med. 2020;180:1125-8
3. Lin JW, Chen CT, Kuo Y, Jeng MJ, How CK, Huang HH. Risk factors for mortality among patients with splenic infarction in the emergency department. J Formos Med Assoc. 2025;124:375-80
4. Wand O, Tayer-Shifman OE, Khoury S, Hershko AY. A practical approach to infarction of the spleen as a rare manifestation of multiple common diseases. Ann Med. 2018;50:494-500
5. Jaroch MT, Broughan TA, Hermann RE. The natural history of splenic infarction. Surgery. 1986;100:743-50
6. Antopolsky M, Hiller N, Salameh S, Goldshtein B, Stalnikowicz R. Splenic infarction: 10 years of experience. Am J Emerg Med. 2009;27:262-5
7. Goerg C, Schwerk WB. Splenic infarction: sonographic patterns, diagnosis, follow-up, and complications. Radiology. 1990;174:803-7
8. Yen CC, Wang CK, Chaou CH, Chen SY, Lin JP, Ng CJ. Anticoagulant Therapy Is Associated with Decreased Long-Term Mortality in Splenic Infarction Patients: A Multicenter Study. Front Med (Lausanne). 2021;8:778198
9. Romano S, Scaglione M, Gatta G, Lombardo P, Stavolo C, Romano L. et al. Association of splenic and renal infarctions in acute abdominal emergencies. Eur J Radiol. 2004;50:48-58
10. Cox M, Li Z, Desai V, Brown L, Deshmukh S, Roth CG. et al. Acute nontraumatic splenic infarctions at a tertiary-care center: causes and predisposing factors in 123 patients. Emerg Radiol. 2016;23:155-60
11. Balcar I, Seltzer SE, Davis S, Geller S. CT patterns of splenic infarction: a clinical and experimental study. Radiology. 1984;151:723-9
12. Carney BJ, Wang TF, Ren S, George G, Al Homssi A, Gaddh M. et al. Anticoagulation in cancer-associated thromboembolism with thrombocytopenia: a prospective, multicenter cohort study. Blood Adv. 2021;5:5546-53
13. McBane Ii R, Loprinzi CL, Ashrani A, Perez-Botero J, Leon Ferre RA, Henkin S. et al. Apixaban and dalteparin in active malignancy associated venous thromboembolism. The ADAM VTE Trial. Thromb Haemost. 2017;117:1952-61
14. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373-83
15. Austin PC, White IR, Lee DS, van Buuren S. Missing Data in Clinical Research: A Tutorial on Multiple Imputation. Can J Cardiol. 2021;37:1322-31
16. van Buuren S, Groothuis-Oudshoorn K. mice: Multivariate Imputation by Chained Equations in R. Journal of Statistical Software. 2011;45:1-67
17. Schemper M. Cox Analysis of Survival Data with Non-Proportional Hazard Functions. Journal of the Royal Statistical Society Series D (The Statistician). 1992;41:455-65
18. Schemper M, Wakounig S, Heinze G. The estimation of average hazard ratios by weighted Cox regression. Stat Med. 2009;28:2473-89
19. Khorana AA, Mackman N, Falanga A, Pabinger I, Noble S, Ageno W. et al. Cancer-associated venous thromboembolism. Nat Rev Dis Primers. 2022;8:11
20. Varki A. Trousseau's syndrome: multiple definitions and multiple mechanisms. Blood. 2007;110:1723-9
21. Gervaso L, Dave H, Khorana AA. Venous and Arterial Thromboembolism in Patients with Cancer: JACC: CardioOncology State-of-the-Art Review. JACC CardioOncol. 2021;3:173-90
22. Yen CC, Wang CK, Chen SY, Gao SY, Lo HY, Ng CJ. et al. Risk assessment and prognostic analysis of patients with splenic infarction in emergency department: a multicenter retrospective study. Sci Rep. 2021;11:21423
23. Bewersdorf JP, Parmar N, Israel GM, Gettinger SN, Lee AI. Clinical characteristics and outcomes of splenic infarction in cancer patients: a retrospective, single center report of 206 cases. J Thromb Thrombolysis. 2021;52:854-62
24. Nores M, Phillips EH, Morgenstern L, Hiatt JR. The clinical spectrum of splenic infarction. Am Surg. 1998;64:182-8
25. Hunt BJ. Bleeding and coagulopathies in critical care. N Engl J Med. 2014;370:847-59
26. Iba T, Levy JH, Maier CL, Helms J, Umemura Y, Moore H. et al. Updated definition and scoring of disseminated intravascular coagulation in 2025: communication from the ISTH SSC Subcommittee on Disseminated Intravascular Coagulation. J Thromb Haemost. 2025;23:2356-62
27. Popescu NI, Lupu C, Lupu F. Disseminated intravascular coagulation and its immune mechanisms. Blood. 2022;139:1973-86
28. Haines RW, Prowle JR, Day A, Bear DE, Heyland DK, Puthucheary Z. Association between urea trajectory and protein dose in critically ill adults: a secondary exploratory analysis of the effort protein trial (RE-EFFORT). Crit Care. 2024;28:24
29. Paulus MC, Melchers M, van Es A, Kouw IWK, van Zanten ARH. The urea-to-creatinine ratio as an emerging biomarker in critical care: a scoping review and meta-analysis. Crit Care. 2025;29:175
30. Martinez LC, Khan SF, Bowman BT. Approach to Electrolyte Abnormalities, Prerenal Azotemia, and Fluid Balance. Prim Care. 2020;47:555-69
31. Liu P, Qu F, Ma C, Ren Y. Association of longitudinal blood urea nitrogen trajectory with in-hospital and 28-day mortality in acute heart failure with cardiorenal syndrome. Eur J Med Res. 2025;30:936
32. Cornman-Homonoff J, Fortune BE, Kolli KP, Kothary N, Nadolski G, Thornburg BG. et al. Management of Ascites: AJR Expert Panel Narrative Review. AJR Am J Roentgenol. 2025
33. Im JH, Chung MH, Lee HJ, Kwon HY, Baek JH, Jang JH. et al. Splenic infarction and infectious diseases in Korea. BMC Infect Dis. 2020;20:915
Author contact
Corresponding author: Hsien-Hao Huang, MD, PhD. Department of Emergency Medicine, Taipei Veterans General Hospital. No. 201, Sec. 2, Shipai Rd., Beitou Dist., Taipei City 112201, Taiwan. Telephone: +886-2-2875-7628. Fax: +886-2-2873-8013. E-mail: hhhuanggov.tw.

 Global reach, higher impact
Global reach, higher impact