Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(6):1933-1951. doi:10.7150/ijms.130435 This issue Cite
Research Paper
Nutritional Supplementation Combined with Exercise for Musculoskeletal Health in Women: A Systematic Review and Meta-Analysis Evaluating Proteins, Amino Acids, and Creatine across Reproductive Stages
1. Post-Baccalaureate Program in Nursing, College of Nursing, Taipei Medical University, Taipei, Taiwan.
2. Cochrane Taiwan, Taipei Medical University, Taipei, Taiwan.
3. Department of Nursing, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan.
4. Evidence-based Knowledge Translation Center, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan.
5. School of Nursing, China Medical University, 100 Jingmao Rd., Sec. 1, Beitun Dist., Taichung 406040, Taiwan.
6. Department of Nursing, China Medical University Hospital, 2 Yude Rd., North Dist., Taichung 404327, Taiwan.
7. Department of Physical Education, Health, and Recreation, Teachers College, National Chiayi University, 85, Wen Long Vil., Minxiong Township, Chiayi County 621302, Taiwan.
8. School of Nursing, Center on Aging and the Life Course, Purdue University, 502 North University Street, West Lafayette, IN 47907, USA.
9. School of Gerontology and Long-Term Care, College of Nursing, Taipei Medical University, 250 Wuxing St, Xinyi District, Taipei 11031, Taiwan.
10. School of Nursing, College of Nursing, Taipei Medical University, 250 Wuxing St, Xinyi District, Taipei 11031, Taiwan.
Received 2025-12-23; Accepted 2026-4-9; Published 2026-4-16
Abstract
Background: Women experience progressive musculoskeletal deterioration across reproductive stages, with accelerated changes during the menopausal transition. Exercise provides established benefits, but the additive effects of nutritional supplementation strategies, including proteins, amino acids, and related compounds such as creatine, remain uncertain. Previous systematic reviews focused on older adults aged 65 years and above, leaving a knowledge gap regarding women facing unique metabolic challenges related to reproductive hormones. Therefore, this systematic review and meta-analysis was conducted to evaluate whether nutritional supplementation combined with exercise enhances muscle mass, strength, and bone health outcomes in women across reproductive stages, with particular attention to the menopausal transition.
Methods: Eight databases were searched from inception to July 2025: CINAHL, ClinicalTrials.gov, Cochrane Central Register of Controlled Trials, Embase, PsycINFO, MEDLINE/PubMed, Scopus, and Web of Science. We included randomized controlled trials examining nutritional supplementation combined with exercise in women across reproductive stages. Interventions comprised whole proteins, amino acids, amino acid derivatives, or protein-derived peptides administered with structured exercise programs. Comparators included exercise alone or a placebo plus exercise. Primary outcomes were muscle mass measures. Secondary outcomes included muscle strength, bone health parameters, body composition, and adverse events. Random-effects meta-analyses calculated Hedges' g with 95% confidence intervals (CIs).
Results: Fourteen trials including 763 women across reproductive stages met the inclusion criteria, which evaluated whole-protein supplements (five studies), amino acids and derivatives (six studies), and creatine monohydrate (three studies). The combined intervention showed no significant effects on muscle mass measures: skeletal muscle mass (g=0.065, 95% CI: -0.353 to 0.482, p=0.762), appendicular lean mass (g=0.197, 95% CI: -0.177 to 0.571, p=0.302), or fat-free mass (g=0.069, 95% CI: -0.110 to 0.249, p=0.447). Significant improvements occurred in bench press (g=0.279, 95% CI: 0.008 to 0.550, p=0.043) and handgrip strength (g=0.412, 95% CI: 0.039 to 0.786, p=0.031). No significant effects emerged for bone mineral content (g=0.195, 95% CI: -0.281 to 0.671, p=0.421) or bone mineral density (g=0.087, 95% CI: -0.129 to 0.303, p=0.430). No increase in adverse events was observed.
Conclusions: Current evidence does not support robust additive effects of nutritional supplementation on muscle mass or bone health when combined with exercise in women across reproductive stages. Selective upper-body strength improvements were observed, particularly in studies using creatine supplementation. Exercise alone provides reliable musculoskeletal health benefits. Given the heterogeneity of supplement types examined, future research should employ longer intervention durations (≥12 months) with site-specific bone measurements and conduct head-to-head comparisons of specific supplement types.
Keywords: exercise, menopause, muscle strength, osteosarcopenia, nutritional supplementation, sarcopenia
Introduction
Musculoskeletal health represents a critical concern across women's reproductive lifespan, with the menopausal transition marking a period of accelerated deterioration. While premenopausal women demonstrate relatively stable muscle mass and bone density, the perimenopausal period initiates progressive changes that accelerate during the first postmenopausal decade [1,2]. Women across the menopausal transition experience muscle mass declines of 0.5%-1.0% annually, substantially exceeding the 0.3%-0.8% observed in age-matched men [3,4]. Concurrently, bone loss accelerates to 1%-3% per year during the first 5 postmenopausal years, compared to 0.5%-1% annually in premenopausal women [5-8]. However, musculoskeletal health optimization begins before menopause, as preventive interventions implemented during the premenopausal years may prove more effective than interventions after significant deterioration occurs [9]. With over 1.2 billion women expected to be menopausal or postmenopausal by 2030 globally, developing effective interventions that span reproductive stages has become a public health priority [10].
This dual deterioration translates into meaningful functional declines including decreased walking speed, impaired balance, reduced capacity for activities of daily living, and increased fall risks [11]. The consequences extend beyond physical function, with over 25% of women reporting substantial limitations during the menopausal transition that affect independence and quality of life [12,13].
The relationship between muscle and bone health is particularly relevant for women during the menopausal transition. Sarcopenia increases fall risks through compromised balance and slower protective reflexes, while also reducing the mechanical loading (muscle forces applied to the bone during contraction) necessary for maintaining bone health. Both muscle strength--particularly lower-extremity strength for mobility and balance, and upper-extremity strength for daily activities--and muscle mass contribute to this mechanical loading. Emerging evidence has demonstrated that sarcopenia and osteoporosis frequently co-occur as osteosarcopenia, with shared pathophysiological mechanisms including an estrogen deficiency, increased inflammatory markers, and altered calcium-protein metabolism [14]. This interconnected deterioration suggests that interventions targeting multiple aspects of musculoskeletal health--muscle mass, strength, and bone density--may provide benefits for fracture prevention in women across the menopausal transition [15].
Exercise, particularly progressive resistance training, has demonstrated clear benefits for muscle mass, strength, and overall musculoskeletal health in women during the menopausal transition [16,17]. For bone health, high-intensity progressive resistance training combined with impact exercises has shown efficacy in maintaining or modestly improving bone mineral density (BMD) at clinically relevant sites in postmenopausal women with low bone mass [18,19]. These exercise-induced benefits occur through multiple mechanisms including enhanced muscle protein synthesis, improved neuromuscular coordination, increased growth factor production, and mechanical loading that stimulates bone remodeling [20,21].
Despite the established benefits of exercise alone, there is a compelling mechanistic rationale for adding nutritional supplementation. An estrogen deficiency impairs muscle protein synthesis through reduced mechanistic target of rapamycin complex 1 signaling and increased myostatin expression, while simultaneously increasing bone resorption through enhanced osteoclast activity and reduced osteoblast function [22,23]. Concurrently, aging-related anabolic resistance reduces muscle responsiveness to dietary protein, a phenomenon well-documented in older adults that may be exacerbated by hormonal changes [24]. Strategic protein supplementation, particularly when timed appropriately around exercise, can theoretically enhance post-exercise muscle protein synthesis rates and support recovery processes essential for both muscle adaptation and bone formation [25-27].
The potential additive effects of protein supplementation with exercise were demonstrated in older adult populations (≥65 years), with recent systematic reviews, including by Yoshimura et al. [28] who reported benefits for muscle mass and strength when protein supplementation was combined with resistance training. However, it remains unclear whether these benefits translate to women during the menopausal transition, who face unique metabolic challenges from an estrogen deficiency that differ from age-related changes alone.
To our knowledge, no systematic reviews have comprehensively evaluated nutritional supplementation strategies, including whole proteins, amino acids, and related compounds such as creatine, combined with exercise across women's reproductive stages. Previous systematic reviews predominantly focused on older adults aged 65 years and above [28-31], examining narrowly defined whole-protein supplements. This represents a significant knowledge gap for several reasons. First, women in their 40s and 50s experiencing the menopausal transition have different metabolic profiles, activity levels, and recovery capacities compared to adults over 65 years [32], yet musculoskeletal health concerns are not confined to the postmenopausal period. Second, the hormonal environment during perimenopause and early menopause creates unique challenges for protein metabolism that may require different supplementation strategies than those effective in older adults [22,24]. Third, diverse supplement types including creatine monohydrate—an amino acid derivative endogenously synthesized from glycine, arginine, and methionine [33,34]—and individual amino acids are frequently investigated for enhancing exercise adaptations [35-39], yet their comparative effectiveness in women remains unclear. Although creatine primarily functions through phosphocreatine-mediated energy metabolism rather than serving as a direct protein building block, we classify it as an amino acid derivative based on its biosynthetic origin and its frequent co-investigation alongside protein and amino acid supplements in exercise nutrition research [25,33-38,40]. We acknowledge that these compounds differ mechanistically—while whole proteins provide amino acid substrates for muscle protein synthesis, creatine primarily enhances phosphocreatine stores and ATP regeneration [33-36]. However, from a pragmatic clinical perspective, these supplements are frequently considered as complementary strategies within comprehensive nutritional approaches to support exercise adaptations [38]. Our inclusive approach aimed to capture the breadth of evidence regarding nutritional interventions studied in combination with exercise, recognizing that this heterogeneity limits mechanistic interpretability but enhances real-world applicability. Finally, understanding whether interventions should differ across reproductive stages versus employing consistent strategies throughout the lifespan has important implications for clinical practice and public health policies [41].
To address these knowledge gaps, we conducted this systematic review and meta-analysis examining nutritional supplementation combined with exercise for musculoskeletal health in women across reproductive stages, with particular attention to the menopausal transition as a critical period of accelerated change. Our primary objectives were to quantify treatment effect magnitudes on muscle mass measures, as these directly relate to sarcopenia prevention. Secondary objectives included evaluating effects on muscle strength parameters relevant to functional independence, bone health outcomes essential for fracture prevention, and body composition measures important for metabolic health. By employing an inclusive approach to both participant selection and supplement type, we aimed to capture the breadth of evidence regarding nutritional strategies to enhance exercise adaptations in women, thereby informing clinical practice and identifying priorities for future research. We recognize that pooling mechanistically distinct interventions limits causal inferences about specific supplement mechanisms, but provides valuable evidence regarding the overall state of research in this population.
Methods
Protocol and registration
This systematic review and meta-analysis were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines [42]. The protocol was registered in PROSPERO (registration no.: CRD420251110761). Two modifications were made to the original registered protocol during the conduct of this review. First, secondary outcomes including muscle strength, bone health parameters, body composition measures, and adverse events were added because the majority of included trials reported these outcomes alongside the primary muscle mass measures; their inclusion provides a more comprehensive and clinically relevant evaluation of musculoskeletal health. Second, certainty of the evidence was assessed using the GRADE approach, which was not prespecified but was incorporated to enhance the interpretability and clinical utility of our findings, consistent with current recommendations for systematic reviews [43]. These protocol deviations were transparently reported in accordance with PRISMA 2020 guidelines [42].
Eligibility criteria
Studies were included based on predefined PICOS (population, intervention, comparison, outcomes, and study) criteria. We included randomized controlled trials (RCTs) examining the effects of nutritional supplementation combined with exercise programs in women across reproductive stages. Eligible interventions comprised whole-protein supplements of any type or dose, individual amino acids including branched-chain amino acids and leucine, amino acid derivatives such as creatine and L-citrulline, or protein-derived peptides administered concurrently with structured exercise programs including resistance training, aerobic exercise, or combined modalities. This inclusive approach was adopted to comprehensively capture nutritional strategies investigated to enhance exercise adaptations in women. We acknowledge that these compounds are mechanistically distinct: whole proteins provide complete amino acid profiles for muscle protein synthesis, isolated amino acids (e.g., leucine) serve as specific anabolic signals, and creatine—although synthesized from amino acids (glycine, arginine, and methionine)—primarily functions through phosphocreatine-mediated energy metabolism rather than as a protein building block [33-36]. Despite these mechanistic differences, we included all of these interventions because: (1) they are frequently studied together in exercise nutrition research as complementary strategies [25,33,35-38,40]; (2) from a clinical practice perspective, women and healthcare providers often consider these as part of comprehensive nutritional approaches to musculoskeletal health [38]; and (3) excluding mechanistically distinct supplements would fragment the evidence base and obscure the overall state of research in this population. We addressed the implications of this heterogeneity in our limitations and interpreted pooled estimates with appropriate caution. This inclusive approach to supplementation use was adopted because these compounds share common metabolic pathways related to muscle protein synthesis and energy metabolism [40], and are frequently investigated as strategies to enhance exercise adaptations [25,33,35-38].
Study populations included women classified as premenopausal, perimenopausal, menopausal, or postmenopausal according to original study definitions. This broad inclusion criterion was chosen because musculoskeletal health optimization is relevant across all reproductive stages [44], with the menopausal transition representing a period of accelerated but not exclusive change [1,2,5-8]. Comparators included exercise alone or placebo plus exercise. Primary outcomes were quantitative measures of muscle mass including lean body mass, appendicular lean mass, skeletal muscle mass, and fat-free mass measured at the baseline and post-intervention. Secondary outcomes included muscle strength parameters, bone health measures including bone mineral density (BMD) and bone mineral content at any skeletal site, body composition indicators, and adverse events. Studies were excluded if they involved participants with hereditary muscle disorders or conditions associated with muscle wasting (cancer, HIV/AIDS, or chronic kidney disease), or had a non-randomized study design. Only English-language publications were considered for inclusion.
Search strategy
We conducted a comprehensive literature search across eight electronic databases from inception through July 2025: CINAHL, ClinicalTrials.gov, Cochrane Central Register of Controlled Trials, Embase, PsycINFO, MEDLINE/PubMed, Scopus, and Web of Science. The comprehensive search strategy combined controlled vocabulary (e.g., MeSH terms, Emtree terms, CINAHL headings) and free-text keywords using Boolean operators, tailored to each database's syntax. The complete search strategies for all eight databases are provided in Supplementary Table S1. An example search for MEDLINE included the following: (menopause OR postmenopaus OR perimenopaus*) AND (protein* OR amino acid* OR supplement*) AND (exercise OR resistance training OR physical activity) AND (muscle mass OR lean mass OR sarcopenia OR bone density). Reference lists of included studies and relevant reviews were manually searched to identify additional eligible studies.
Study selection and data extraction
Following duplicate removal, two independent reviewers (KHC and IHC) screened titles and abstracts against eligibility criteria. Full-text articles were retrieved for potentially eligible studies and independently assessed for final inclusion. Disagreements were resolved through discussion. Data were independently extracted by two reviewers (KHC and IHC) using a standardized, pilot-tested form. Extracted variables included study characteristics (author, year, and country); participant characteristics (sample size, age, menopausal status, and health condition); intervention parameters (protein type, dose, timing, exercise modality, duration, and adherence); comparison group details (type of control intervention, dose, exercise modality, duration, and adherence); outcome measures (muscle mass assessment method, and specific measures and units); and results (pre-intervention and post-intervention values, within-group changes, between-group mean differences, and statistical significance). When reported data were incomplete or required clarification, study authors were contacted via email with a maximum of two contact attempts over a 4-week period.
Risk of bias assessment
Risk of bias was assessed by two independent reviewers (KHC and IHC) using the revised Cochrane Risk of Bias tool (RoB 2) [45] across five domains: randomization process, deviations from intended interventions, missing outcome data, outcome measurement, and selective reporting. Each domain was rated as 'low risk,' 'some concerns,' or 'high risk.' The overall study quality was determined by the highest risk rating across all domains. Disagreements were resolved through discussion.
Data synthesis and analysis
Random-effects meta-analyses were conducted using Comprehensive Meta-Analysis software (vers. 3.0, Biostat, Englewood, NJ, USA). Effect sizes were computed from change scores (mean±standard deviation (SD)) as Hedges' g (standardized mean difference) with 95% confidence intervals (CIs) for continuous outcomes. For dichotomous outcomes, we computed risk ratios with 95% CIs. Statistical significance was evaluated using Z-statistics associated with pooled effect estimates, with a two-tailed p value of <0.05 considered significant. Between-study heterogeneity was assessed using I² statistics, with values of 25%, 50%, and 75% respectively considered low, moderate, and high heterogeneity [46]. Publication bias was evaluated using funnel plots and Egger's regression test when at least 10 studies were available for analysis [47].
For bone health outcomes, we acknowledge a priori that most included trials employed relatively short intervention durations and assessed total body BMD rather than site-specific measurements at clinically relevant locations such as the lumbar spine or hip [48]. Nevertheless, we included these outcomes because they provide preliminary insights into the potential skeletal effects of combined interventions and can inform the design of future longer-term studies with site-specific assessments [18,19,49,50].
Pre-planned subgroup analyses were designed to examine: (1) protein type and dosage (≤20 vs. >20 g/day); (2) exercise modality (resistance alone vs. combined training); (3) intervention duration (≤12 vs. >12 weeks); (4) menopausal status (perimenopausal vs. postmenopausal); and (5) the baseline muscle mass status. However, these analyses were not conducted due to the limited number of included studies, as subgroup analyses require at least 10 studies per subgroup to provide meaningful results according to Cochrane guidelines [51]. Sensitivity analyses by excluding studies with a high risk of bias were not performed as no studies were classified as having a high risk of bias in any domain. We assessed the quality of evidence for all meta-analyzed outcomes using the GRADE approach [43].
Results
Study selection and characteristics
Our systematic search yielded 2117 records across eight databases (Supplementary Table S1). After removing 663 duplicates, 1454 records were screened by title and abstract, of which 1309 were excluded. Of the 145 reports sought for retrieval, five could not be obtained. The remaining 140 full-text articles were assessed for eligibility, and 126 were excluded for the following reasons: wrong patient group (n=18), animal study (n=1), wrong intervention (n=79), not English (n=2), no outcome of interest (n=4), conference paper (n=4), and wrong study design (n=18). Ultimately, 14 studies met all inclusion criteria (Fig. 1). Included studies were conducted in five countries: the US (five studies [52-56]), Brazil (four studies [57-60]), Canada (two studies [61,62]), Germany (two studies [63,64]), and New Zealand (one study [65]).
PRISMA flow chart.
According to Table 1, these studies (published between 2007 and 2025) included 763 participants (386 in the intervention groups and 377 in the comparison groups), with mean ages ranging 32.8-67.1 years, reflecting diverse reproductive stages from premenopausal through postmenopausal years. Two studies exclusively enrolled premenopausal women with mean ages below 42 years [63,65], two studies included women during the perimenopausal transition [52,53], and the remaining studies focused on postmenopausal women with various years since menopause onset [54-62,64]. This heterogeneity in reproductive stage was considered acceptable given our research question regarding nutritional supplementation effects across the female lifespan, with particular attention to the menopausal transition as a critical period of accelerated musculoskeletal change [1,2,5-8]. Most participants were sedentary or inactive at the baseline (reported in eight of 14 studies [54,55,57,58,61-63,65]) and were living in the community (all studies). The body-mass index (BMI) at the baseline ranged 24.0±3.9-35.0±3.4 kg/m² (reported in 13 of 14 studies [52,53,55-65]). Among studies reporting years since menopause, the duration ranged 7.6±4.7-9.1±5.2 years [58,59]. Use of hormone replacement therapy was explicitly prohibited in most studies. Dropout rates considerably varied from 0% [54,61] to 50% [57].
Study characteristics of the included articles
| First author (year) country | Health status | Intervention group | Comparison group | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N/n a | Age, years (mean±SD) | BMI. kg/m2 (SD) | Years since menopause (mean±SD) | Hormone replacement therapy use (%) | Dropout rates (%) | N/n a | Age, years (mean±SD) | BMI, kg/m2 (SD) | Years since menopause (mean±SD) | Hormone replacement therapy use (%) | Dropout rates (%) | ||
| Bagheri (2021) [54] USA | Postmenopausal (≥1 year without menstruation), sedentary, healthy | 10/10 | NR* | NR | NR | No | 0 | 10/10 | NR* | NR | NR | No | 0 |
| Chapman-Lopez (2024) [52] USA | Perimenopausal/postmenopausal (self-identification) | 23/17 | 55.76±7.43 | 27.7 (4.7) | NR | NR | 26 | 23/18 | 53.50±6.20 | 28.6 (5.6) | NR | NR | 22 |
| Chilibeck (2015) [62] Canada | Postmenopausal (a questionnaire regarding menstrual period + FSH/ luteinizing hormone if <2 years) | 23/15 | 57.0±4.0‡ | 26.6 (4.2) | NR | No | 43 | 24/18 | 57.0±7.0‡ | 27.5 (6.0) | NR | No | 26 |
| Chilibeck (2023) [61] Canada | Postmenopausal (a questionnaire regarding menstrual period + FSH/ luteinizing hormone if <2 years), low & moderate risk of osteoporosis | 120/120 | 59.0±5.6 | 27.3 (5.9) | NR | No | 0 | 117/117 | 59.0±5.7 | 27.0 (5.7) | NR | No | 2.5 |
| Figueroa (2015) [55] USA | Postmenopausal (≥1 year without menstruation), BMI ≥ 30 kg/m2, prehypertension or stage-1 HTN, nonsmokers, sedentary | 13/13 | 58.0±3.6 | 33.8 (4.0) | NR | No | NR | 14/14 | 58.0±3.7 | 35.0 (3.4) | NR | No | NR |
| Funderburk (2020) [53] USA | Perimenopausal/postmenopausal (self-identification), healthy | 23/18 | 53.5±6.2 | NR | NR⁋ | NR | 22 | 23/18 | 55.6±7.2 | NR | NR⁋ | NR | 22 |
| Gualano (2014) [60] Brazil | Postmenopausal, osteopenia or osteoporosis | 19/15 | 67.1±5.6 | 28.0 (2.1) | NR | NR | 21 | 19/15 | 63.6±3.6 | 28.2 (3.6) | NR | NR | 21 |
| Ioannidou (2024) [64] Germany | Postmenopausal (last period >2 years ago + low E2 and P levels confirmed by saliva test), healthy | NR/15 | 57.0±6.1 | 25.8 (3.8) | NR | 0 | NR‡ | NR/12 | 57.6±5.1 | 24.5 (3.4) | NR | 0 | NR‡ |
| Jendricke (2019) [63] Germany | Premenopausal (a comprehensive anamnesis) | 45/40 | 38.3±8.7 | 26.4 (3.8) | NA | NA | 11 | 45/37 | 41.6±6.9 | 26.5 (3.4) | NR | NA | 18 |
| Kang (2023) [56] USA | Postmenopausal (≥1 year without menstruation), sedentary, HTN | 14/13 | 62.0±2.0 | 29.6 (4.0) | NR | 29 | 7 | 14/11 | 63.0±1.0 | 29.2 (5.6) | NR | 21 | 21 |
| Maesta (2007) [59] Brazil | Postmenopausal (>1 year amenorrhea+FSH >40 mIU/mL) | 14/14 | 57.6±6.7 | 27.8 (4.0) | 8.1±5.3 | No | NR† | 11/11 | 60.7±7.1 | 27.7 (4.4) | 7.6±4.7 | No | NR† |
| Murray (2025) [65] New Zealand | Premenopausal, healthy, nonsmokers, BMI >18.5 <30 kg/m2 | 16/15 | 34.2±9.1 | 24.0 (3.9) | NA | NA | 6 | 15/12 | 32.8±9.7 | 27.1 (3.1) | NA | NA | 20 |
| Orsatti (2018) [58] Brazil | Postmenopausal (>1 year amenorrhea+FSH >40 mIU/mL), healthy, sedentary | 21/16 | 56.8±6.6 | 27.5 (4.0) | 7.6±5.2 | No | 24 | 20/16 | 58.8±8.9 | 27.3 (4.1) | 9.1±5.2 | No | 20 |
| Trevisan (2010) [57] Brazil | Postmenopausal (>1 year amenorrhea+FSH >40 mIU/mL), sedentary | 30/15 | 58.0±7.0 | 28.0 (4.0) | NR | No | 50 | 30/15 | 57.0±9.0 | 28.0 (5.0) | NR | No | 50 |
a Sample sizes given as N (recruited)/n (included in analysis).‡ Sample age at recruitment.
BMI, body-mass index; E2, estradiol; P, progesterone; NR, not reported; HTN, hypertension; FSH, follicle-stimulating hormone; NA, not available. NR* Not reported stratified for intervention and control groups, total sample age 56±3.7 years. NR⁋ Only reported postmenopausal women were with a mean duration of 7 years since the onset of menopause. NR† Not reported stratified for intervention and control groups, total dropout rate 23%. NR‡ Not reported stratified for intervention and control groups, total dropout rate 13%. Note: Studies included women across diverse reproductive stages from premenopausal through postmenopausal years. Menopausal status definitions were as reported in original studies.
According to Table 2, studies investigated diverse nutritional supplements rather than exclusively whole-protein supplements. Three studies examined creatine supplementation [60-62], a nitrogen-containing compound synthesized from amino acids that plays a critical role in cellular energy metabolism and has been extensively studied for its effects on muscle mass and strength [33,35,36]. Creatine dosages ranged from 0.1 g/kg body weight (BW) to 20 g/day. Other supplements included 9 g of branched-chain amino acids daily [54], 5 g of leucine daily [52,53], 6-10 g of L-citrulline daily [55,56], 15 g of collagen peptides daily [63], 24 g of whey protein daily [65], 25 g of soy protein daily [57,58], combined soy plus milk protein of 37.2 g/day [59], and 2.5 g/kg fat-free mass of a high-protein diet daily [64]. This heterogeneity reflects the breadth of nutritional strategies investigated to enhance exercise adaptations in women. Baseline dietary protein intake was reported in eight of 14 studies, and ranged 55.5-83.4 g/day (or 0.8-1.0 g/kg BW/day where reported) [52-54,57-59,61,65]. With supplementation, total protein intake would have ranged from approximately 60 to 120 g/day, although actual total intake could only be calculated for studies reporting baseline values.
Intervention characteristics of the included articles (nutritional supplements and exercise protocols)
| First author | Intervention group | Comparison group | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Intervention | Exercise | Dose per serving (g) | Total dose (g/day) | Frequency | Timing | Duration (weeks) | Adherence (%) | Baseline protein intake (g/day) | Intervention | Baseline protein intake (g/day) | |
| Bagheri [54] | BCAA+supervised resistance exercise | Progressed from 3×12 @60%-65% 1RM to 4×10 @70%-75%. | 9 (4.5 g leucine+2.25 g isoleucine+2.25 g valine) | 9 | 3 times/week | Half 30 min before & half within 30 min following exercise | 8 | 100 (questionnaire+container return) | 55.5±9.0 | PL+supervised resistance exercise Both: 100 | 57.2±11 |
| Chapman-Lopez [52] | Leucine+supervised resistance exercise | Weeks 1-3: 1-2 sets of upper- & lower-body machines. Weeks 4-10: 3 sets × 10 reps. Progressive overload. Equipment: machines. | 5 | 5 | 3 times/week | Immediately after exercise | 10 | Leucine: 99.1±1.4 Exercise: 97.6±3.8 (attendance+pill count) | 83.4±29.3 | PL(apple pectin 5 g/day)+supervised resistance exercise PL: 96.3±3.2 Exercise: 95.8±3.7 | 68.3±40.9 |
| Chilibeck [62] | Creatine+supervised resistance exercise | 3 sets × 10 reps to fatigue. Intensity ≈80% 1RM for hack squat and bench press; ≈10 RM for other exercises. Progressive overload applied individually. ~15 exercises/session (e.g., hack squat, multihip machine, curls, presses, lat pulldown, dumbbells). Equipment: free weights, machines. | NA | 0.1 g/kg | Creatine: 7 days/week Exercise: 3 times/week | Immediately before (0.05 g/kg) & after (0.05 g/kg) exercise With two meals (0.05 g/kg) on non-training days | 48 | Creatine: 79 Exercise: 75 (117/156 sessions) (attendance logs, pill count, diaries) | NR | PL (maltodextrin)+supervised resistance exercise PL: 78 Exercise: 77 (120/156 sessions) | NR |
| Chilibeck [61] | Creatine+supervised resistance/walking exercise | Resistance: 2 sets × 8 reps to muscular fatigue at ≥80% 1RM or 8 RM. Progressive overload by 2-5 kg when able to complete 2 × 8. ~12 exercises/session (hack squat, hip machines, bench press, etc.). Equipment: plate-loaded and weight-stack machines. Walking: 20-30 min/session at 70% HR max. | 0.14 g/kg creatine+0.14 g/kg maltodextrin | 0.14 g/kg | Creatine: 7 days/week Exercise: 3 times/week resistance; 6 times/week walking (3 supervised+3 unsupervised) | Half 5 min post resistance & half with a meal 1/3 with each meal on non-training days | 96 | Creatine: 56 Resistance: 61 Walking: 65 (pill count, logs) | 67.0±22.0 | PL (maltodextrin 0.28 g/kg/day)+supervised resistance/walking exercise PL: 60 Resistance: 61 Walking: 60 | 64.0±23.0 |
| Figueroa [55] | L-citrulline+WBVT | 4 static + 4 dynamic leg exercises Equipment: vibration platform (pro5 AIRdaptive). Intensity: vibration 25-40 Hz, amplitude 1-2 mm. Sets × Reps: 1-5 sets, 30-60 s per set; dynamic exercises at 3 s eccentric / 2 s concentric pace. Progressive overload: increase vibration intensity, duration, # sets, total session time (11-60 min); decrease rest (60→30 s). | 0.75 | 6 | L-citrulline: 7 days/week WBVT: 3 times/week | Half before breakfast & half before sleep | 8 | L-citrulline: 96.4±3.6 WBVT: 99.7±1.1 (capsule return count, logs) | NR | PL (maltodextrin)+WBVT PL: 95.8±3.0 WBVT: 99.7±1.1 | NR |
| Funderburk [53] | Leucine+supervised resistance exercise | Weeks 1-2: 2×10 reps at 80% 1RM; Weeks 3-10: 3×10 reps. Load adjusted to reach volitional exhaustion in final set. 6 resistance machine exercises: leg press, leg extension, seated leg curl, shoulder press, bench press, seated row. 60 s rest between sets. | 0.5 | 5 | Leucine: 7 days/week Exercise: 3 times/week | Immediately after exercise With first meal on non-training days | 10 | Leucine: 99.1±1.4 Exercise: 97.6±3.8 (capsule count, verbal check every 2 weeks, attendance logs) | 83.4±29.3 | PL (apple pectin 5 g/day)+supervised resistance exercises PL: 96.3±3.1 Exercise: 95.8±3.7 | 68.2±39.6 |
| Gualano [60] | Creatine+supervised resistance exercise | Week 1: 2 sets of 15-20 RM; Weeks 2-24: 3 sets of 8-12 RM. Load progressed when >12 reps were achieved in a set. Exercises: leg press, leg extension, squat, seated row, bench press, lat pulldown, sit-ups. | 5 | 20 | Creatine: 7 days/week Exercise: 2 times/week | Breakfast, lunch, dinner & before bed time on training days Lunch on non-training days | 24 | Creatine: NR Exercise: 84.4±8.0 | 56.0±13.0 | PL (dextrose 20 g/day)+supervised resistance exercise PL: NR Exercise: 83.9±6.1 | 58.0±18.0 |
| Ioannidou [64] | Protein+supervised resistance exercise | Each session: 6 sets, reps from 10→3, RIR from 8→1. Rest: 60-180 s based on RIR. Includes peak (1RM test) and deload weeks (↓30% load). Tempo: 1 s concentric / 1 s hold / 1 s eccentric. Exercises: box back squat, deadlift, lat pulldown, lateral dumbbell raises, single arm lateral walking carries. | Daily: Whey & protein-rich cheese 2.5 g/kg FFM/day Post-training: 30 g whey+40 g maltodextrin | Daily: 2.5 g/kg FFM/day Post-training: 30 | Protein: 7 days/week Post-training: 3 days/week Exercise: 3 times/week | Daily: NR Post-training | 12 | NR | NR | Supervised resistance exercise Exercise: NR | NR |
| Jendricke [63] | Collagen peptides+ Supervised resistance exercise | Exercises: leg press, back trainer, lat pull-down, sit-ups, chest press. Sets: 3 per exercise. Reps progressed: Weeks 1&2: 15 reps; Weeks 3&4: 12 reps; Weeks 5-8: 10 reps; Weeks 9-12: 8 reps. Load adjusted to maintain rep targets with proper form. | NA | 15 | Collagen: 7 days/week Exercise: 3 times/week | 60 min after exercise Same time on non-training days | 12 | NR (missing >6 sessions were excluded) | NR | PL (silicon dioxide)+ supervised resistance exercise | NR |
| Kang [56] | L-citrulline+ SVLIRT | Exercises: leg press, leg extension, leg curl, calf raise. Intensity: Weeks 1&2 at 40% 1RM; Weeks 3&4 at 50% 1RM. Tempo: 3 s concentric + 3 s eccentric using metronome. Volume: 3 sets × 15 reps; rest: 1-3 min. | 0.77 | 10 | L-citrulline: 7 days/week SVLIRT: 3 times/week | 4.62 g in morning & 5.39 g at night | L-citrulline: 8 SVLIRT: 4 | L-citrulline: 96±6† SVLIRT: 95±7† (capsule return, biweekly phone check, Attendance logs) | NR | PL (maltodextrin 10 g/day)+ SVLIRT PL: 96±6† SVLIRT: 95±7† | NR |
| Maesta [59] | Soy+milk protein+supervised resistance exercise | Week 1: 1 set of 15 reps at 40%-50% 1RM; progressed to 3 sets of 8-12 reps at 60%-80% 1RM. Monthly load adjustments. Full-body workout (40-50 min): 2 exercises per major muscle group (chest, back, thighs), 1 per minor group (biceps, triceps). Exercises included: leg press, leg extension, peck deck, bench press, seated row, lat pulldown, triceps pulley, biceps curl, plus abs (3×30) and calves (3×20 BW). | 37.2 | Soy: 25 Milk: 12.2 | Soy+milk: NR Exercise: 3 times/week | NR | 16 | NR | NR | PL (maltodextrine 25 g/day) +supervised resistance exercise | NR |
| Murray [65] | Whey protein+supervised resistance exercise/interval training | Resistance: upper body dumbbell training. Exercises: chest press, single-arm row, shoulder press; other upper-body & abs rotated (not recorded). Load progressed via “rule of two”; adjusted per 3RM. Interval: cycling intervals. Sprint = RPE 10, rest = RPE 2. Intervals progressed from 6 to 9. | 24 | 24 | Whey protein 5 days/week Resistance: 2 times/week (48 h apart) Interval: 3 times/week | After exercise | Whey protein 12 Resistance 9 Interval 12 | Whey protein NR Resistance 89±7 Interval 82±13 (attendance logs) | 70±20 g/day | PL (Milo powder)+supervised resistance exercise/interval training PL: NR Resistance: 91±7 Interval: 89±8 | 79±12 g/day |
| Orsatti [58] | Soy protein+ supervised resistance exercise | Week 1 - 1×15 reps at 40%-50% 1RM; Week 4 - 3×8-12 reps at 60%-80% 1RM. Monthly load adjustments (0%-5%) to maintain 8-12 RM range. Exercises: leg press, leg extension, leg curl, fly, bench press, seated row, lat pulldown, triceps pulley, biceps curl, abs (3×30), calves (3×20 BW). | 25 | 25 | Soy protein: 7 days/week Exercise: 3 times/week | After exercise Same time on non-training days | 16 | Soy: NR Exercise: 100 | 1.0±0.3 g/kg | PL (maltodextrin 25 g/day)+ supervised resistance exercise PL: NR Exercise: 100 | 0.8±0.3 g/kg |
| Trevisan [57] | Soy protein+ supervised resistance exercise | First 4 weeks aimed at physical aptitude equalization. Warm-up: 10 unloaded reps before each exercise; post-session: 15 s static stretch per muscle group. 10 exercises: leg press, leg extension, leg curl, bench press, pec-deck fly, seated row, lat pulldown, triceps pulley, barbell curls, abdominal crunches (3×30 reps). Progressed to 3 sets of 8-12 reps at 60%-80% 1RM with periodic adjustments. 1-2 min rest between sets; 1-2 s pause between reps. All sessions used appropriate machines | 25 | 25 | Soy protein: 7 days/week Exercise: 3 times/week | NR | 16 | NR | 60.0±21.0 | PL (maltodextrin)+supervised resistance exercise | 59.0±39.0 |
BCAA, branched-chain amino acid; FFM, fat-free mass; RM, repetition maximum; 1RM, one repetition maximum; PL, placebo; NA, not applicable; NR, not reported; RIR, repetitions in reserve. RPE, rating of perceived exertion; WBVT, whole-body vibration exercise training; SVLIRT, slow-velocity low-intensity resistance training.
† Mean of all participants.
Exercise protocols predominantly consisted of supervised resistance training performed two or three times per week. One study incorporated both resistance training (three times/week) and walking (six times/week) [61]. Two studies used alternative resistance training modalities: whole-body vibration training [55] and slow-velocity, low-intensity resistance training [56]. One study combined resistance exercise (twice/week) with interval training (three times/week) [65]. Training intensity progressed from 40%-50% to 60%-80% of one-repetition maximum (1RM) in most studies. Intervention durations ranged from 8 [54-56] to 96 weeks [61], with a median duration of 16 weeks. Adherence data showed protein supplementation compliance ranging from 56% [61] to 100% [54,58]. Exercise adherence ranged from 61% [61] to 100% [58].
Risk of bias assessment
As shown in Fig. S1, nine of 14 studies (64%) demonstrated a low risk of bias across all domains [52,54,57,58,60-63,65]. The remaining five studies (36%) showed some concerns regarding the randomization process, primarily due to insufficient reporting of allocation concealment methods [53,55,56,59,64]. All studies demonstrated a low risk of bias for deviations from the intended intervention, missing outcome data, measurement of outcomes, and selection of reported results. The overall methodological quality of the included studies was high, with no study classified as having a high risk of bias in any domain. The concerns in five studies arose from insufficient reporting detail rather than clear evidence of bias; given the low risk of bias across all other domains, we retained all studies in the primary analyses.
Effects on muscle mass outcomes
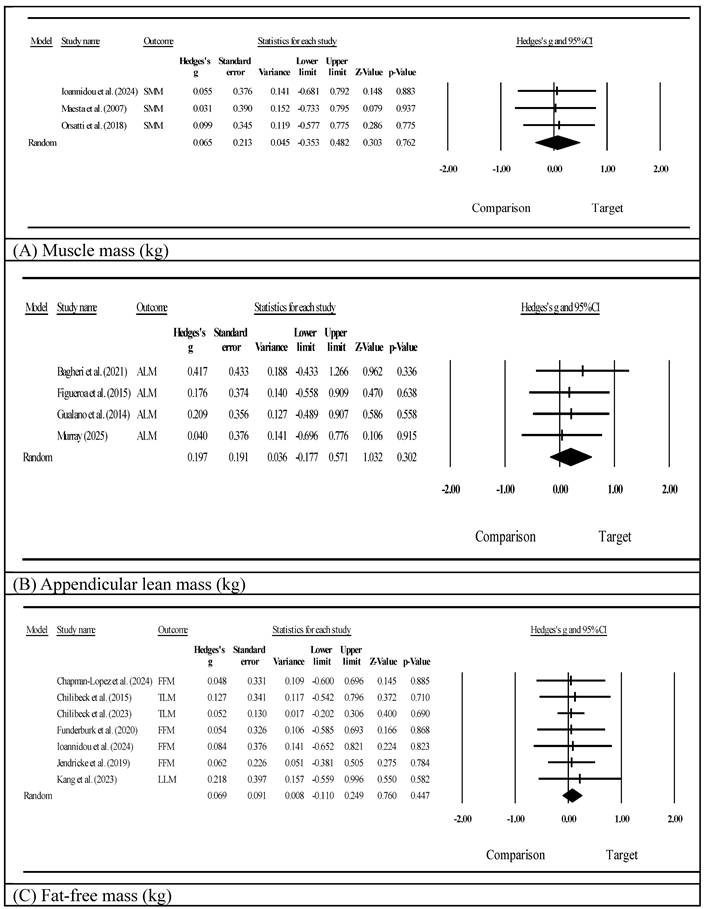
Muscle mass was assessed using either a bioelectrical impedance analysis (BIA; n=6) [54,55,57-59,64] or dual-energy x-ray absorptiometry (DXA; n=8) [52,53,56,60-63,65] (Supplementary Table S2). Specific muscle mass parameters considerably varied, including appendicular lean mass, fat-free mass, total lean mass, skeletal muscle mass, and the appendicular lean mass index. Because the small number of studies per supplement type precluded formal subgroup meta-analyses [51], we present a narrative summary by supplement category. Among whole-protein studies (k=5) [57-59,64,65], individual studies showed within-group improvements in both the intervention and control groups, but none demonstrated significant between-group differences. Amino acid and derivative studies (k=6) [52-56,63] showed mixed results: Kang et al. [56] (L-citrulline) reported a significant between-group improvement in leg lean mass (p<0.05), and Jendricke et al. [63] (collagen peptides) reported improved fat-free mass (d=0.55, p<0.05), while the remaining four studies did not reach significance. Creatine studies (k=3) [60-62] also showed mixed findings: Gualano et al. [60] reported a significant between-group improvement in appendicular lean mass (p=0.002), whereas Chilibeck et al. [61,62] reported no significance for total lean mass. Overall, no supplement category showed consistently superior effects on muscle mass outcomes. A meta-analysis of muscle mass (skeletal muscle mass, Fig. 2A) showed a statistically non-significant effect favoring protein supplementation combined with exercise (Hedges' g=0.065, 95% CI: -0.353 to 0.482, p=0.762, I²=0.000). Appendicular lean mass (Fig. 2B) measured in four studies similarly showed a non-significant positive effect (Hedges' g=0.197, 95% CI: -0.177 to 0.571, p=0.302, I²=0.000). Fat-free mass (Fig. 2C) as assessed in seven studies showed a non-significant trend (Hedges' g=0.069, 95% CI: -0.110 to 0.249, p=0.447, I²=0.000). All three pooled effect sizes were below the threshold for a small effect (g<0.2), suggesting trivial between-group differences. For the clinical context, individual studies reporting raw values showed within-group changes of approximately 1.2-1.4 kg for muscle mass measures [64], although between-group differences were not statistically significant in any study.
Effects on muscle mass outcomes.
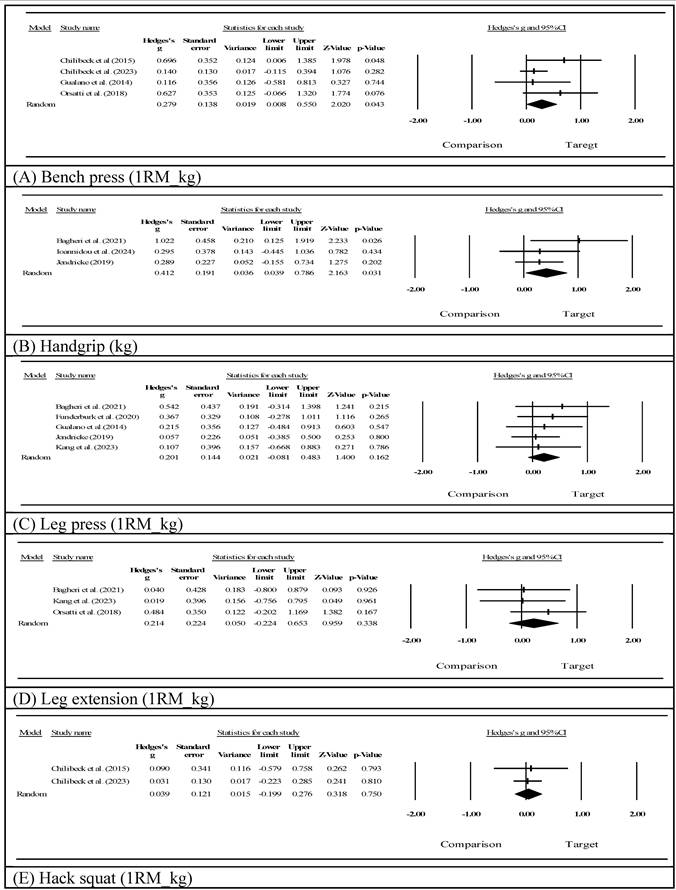
Muscle strength outcomes
Upper-body strength assessed via bench pressing (Fig. 3A) showed a significant improvement (Hedges' g=0.279, 95% CI: 0.008 to 0.550, p=0.043, I²=16.613). Notably, three of the four studies contributing to this outcome involved creatine supplementation [60-62], suggesting that this finding may be largely attributable to creatine's established ergogenic effects on high-intensity resistance exercise [33,35]. Handgrip strength (Fig. 3B) demonstrated a statistically significant improvement following the intervention (Hedges' g=0.412, 95% CI: 0.039 to 0.786, p=0.031, I²=7.416). In contrast, lower-extremity strength showed favorable but non-significant trends, including leg press 1RM (Fig. 3C, Hedges' g=0.201, 95% CI: -0.081 to 0.483, p=0.162, I²=0.000), leg extension strength (Fig. 3D, Hedges' g=0.214, 95% CI: -0.224 to 0.653, p=0.338, I²=0.000), and hack squat performance (Fig. 3E, Hedges' g=0.039, 95% CI: -0.199 to 0.276, p=0.750, I²=0.000; k=2, very limited evidence).
Effects on muscle strength outcomes.
Bone health outcomes
Bone health was assessed in five studies [52,60-62,65], with all studies employing DXA for bone mineral measurements. Bone mineral content (Supplementary Fig. S2A) showed a non-significant positive trend favoring the combined intervention (Hedges' g=0.195, 95% CI: -0.281 to 0.671, p=0.421, I²=0.000; k=2, very limited evidence). BMD (Fig. S2B) similarly failed to reach statistical significance (Hedges' g=0.087, 95% CI: -0.129 to 0.303, p=0.430, I²=0.000). Notably, all six studies assessed total body BMD rather than site-specific measurements (e.g., lumbar spine or femoral neck), and intervention durations ranged 8-96 (median 16) weeks, which may have been insufficient to detect clinically meaningful skeletal changes that typically require 12 months or longer [48,50].
Body composition measures
There were no significant pooled effects of the intervention on body composition indicators. BW (Fig. S3A) remained stable (Hedges' g=-0.016, 95% CI: -0.272 to 0.239, p=0.901, I²=0.000), and no meaningful change in the BMI was observed (Fig. S3B, Hedges' g=-0.033, 95% CI: -0.279 to 0.212, p=0.790, I²=0.000). The body fat percentage (Supplementary Fig. S3C) demonstrated a non-significant reduction (Hedges' g=-0.081, 95% CI: -0.303 to 0.141, p=0.473, I²=0.000), while visceral adipose tissues (Supplementary Fig. S3D, Hedges' g=-0.111, 95% CI: -0.527 to 0.304, p=0.599, I²=0.000), and waist circumference (Supplementary Fig. S3E, Hedges' g=-0.056, 95% CI: -0.601 to 0.488, p=0.839, I²=0.000; k=2, very limited evidence) were also unaffected.
Adverse events
Limited information was provided regarding adverse events across the included studies. No statistically significant differences were observed in adverse event risks between the intervention and control groups (Supplementary Fig. S4, risk ratio (RR)=1.177, 95% CI: 0.510 to 2.717, p=0.703; k=2, very limited evidence). Most reported events were mild in nature, including muscle soreness and gastrointestinal discomfort. No serious adverse events were reported in any of the included trials. However, systematic reporting of adverse events was lacking in many of the included trials, and the available data are insufficient to draw definitive conclusions regarding supplement safety.
GRADE evidence summary
The quality of evidence was assessed using the GRADE approach [43] for all meta-analyzed outcomes and is summarized in Supplementary Table S3. For muscle mass measures, the quality of evidence was rated as low, being downgraded due to imprecision and indirectness. Upper-body strength (bench press and handgrip), BW, BMI, and body fat percentage were rated as low-quality evidence, being downgraded for indirectness and imprecision. Bone health outcomes were rated as very low-quality evidence due to very serious indirectness and serious to very serious imprecision. Overall, the evidence suggested that nutritional supplementation combined with exercise probably makes little or no difference to muscle mass outcomes in women across the menopausal transition.
Discussion
Principal findings in context
In this systematic review and meta-analysis of 14 RCTs including 763 women across diverse reproductive stages from premenopausal through postmenopausal years, we evaluated the effects of nutritional supplementation combined with exercise compared to exercise alone or a placebo plus exercise. Our inclusive approach to both participant selection and supplementation type was designed to capture the breadth of evidence regarding nutritional strategies to enhance exercise adaptations in women, with particular attention to the menopausal transition as a period of accelerated musculoskeletal change [1,2,5-8]. While pooling mechanistically distinct supplements (whole proteins, amino acids, or creatine) limited causal inferences about specific mechanisms, our approach characterized the current research landscape and provided practical insights into the diverse nutritional strategies studied in this population. Our findings revealed limited additional benefits of nutritional supplementation beyond exercise alone for muscle mass outcomes, with significant improvements observed only for upper-body strength measures. Critically, bone health outcomes assessed in short-duration studies using total body measurements showed non-significant trends (BMD: g=0.087, p=0.430; bone mineral content: g=0.195, p=0.421) that neither support nor refute potential skeletal benefits; these findings should be considered inconclusive rather than evidence of no effect. Heterogeneity in the reproductive stage, supplementation type, and exercise protocols reflects real-world clinical complexity but also limits our ability to identify optimal intervention strategies for specific populations and precludes definitive mechanistic conclusions about individual supplement types.
The significant improvements in the bench press (g=0.279, p=0.043) and handgrip strength (g=0.412, p=0.031) without corresponding lower-extremity strength improvements have important functional implications. Lower-extremity strength more directly relates to mobility, balance, and fall prevention--critical factors for fracture risk reduction [11,13,66,67]. This selective improvement pattern may reflect distinct fiber type distributions and hormonal sensitivity between the upper- and lower-body musculature in women [68].
Mechanistic considerations and dose-response relationships
The selective improvement in the bench press may partly be attributable to creatine, as three of four contributing studies included creatine supplementation [60-62]. Creatine enhances strength performance through increased phosphocreatine stores and accelerated ATP regeneration [33,35], with effects potentially more pronounced in women whose baseline muscle creatine stores are typically 10%-15% lower than men's [38]. The phosphocreatine system provides rapid ATP replenishment during short-duration, high-intensity contractions typical of resistance training, potentially explaining strength gains without corresponding muscle mass increases [36]. Dosing protocols varied across the three creatine studies: Chilibeck et al. [61,62] used 0.1-0.14 g/kg BW/day, and Gualano et al. [60] used 20 g/day initially then 5 g maintenance; none followed a loading protocol of 20 g/day for 5-7 days, which expedites muscle creatine saturation, although maintenance-only dosing at 3-5 g/day can achieve comparable saturation over approximately 3-4 weeks [35,37].
The limited efficacy observed for muscle mass outcomes raises important mechanistic questions. While some studies in our review used protein doses (20-37 g/day) similar to those used in older adult studies [29,30], and others used lower doses (5-15 g/day), neither approach showed consistent benefits. Among the eight studies reporting the baseline dietary protein intake, participants consumed 55-83 g/day (0.8 to 1.0 g/kg BW/day) [52-54,57-59,61,65], suggesting that even with supplementation, the total protein intake may have remained below the 1.2-1.6 g/kg BW/day threshold shown to be effective in older adults [30,31,69-70]. The unique metabolic environment of an estrogen deficiency may create different protein requirements. An estrogen deficiency induces profound anabolic resistance through multiple pathways: increased inflammatory cytokines (tumor necrosis factor-α and interleukin-6), altered insulin-like growth factor-1 signaling and mitochondrial dysfunction, and increased protein breakdown via the ubiquitin-proteasome pathway [22,23,71]. This suggests that women during the menopausal transition may require different supplementation strategies--potentially higher total protein intake levels with leucine enrichment as suggested by Sims et al. [31]. Moreover, muscle-bone unit crosstalk through shared regulatory pathways including insulin-like growth factor-1 and myokines [72,73] suggests that standard supplementation doses may be insufficient to enhance exercise-induced anabolic pathways in an estrogen-deficient state [20,21].
Bone health considerations and measurement limitations
The reliance on total body BMD rather than site-specific measurements at clinically relevant locations (hip, spine, and distal radius) represents a significant limitation of the current evidence [74,75]. Total body measurements have lower sensitivity for detecting regional adaptations, particularly at trabecular bone-predominant sites most responsive to interventions [76]. Future trials should prioritize site-specific measurements at the lumbar spine and proximal femur, consistent with International Society for Clinical Densitometry recommendations [77].
Clinical implications and future research directions
Our findings suggest that exercise alone remains the cornerstone intervention for musculoskeletal health in women across reproductive stages [16-19]. Current evidence does not support routine nutritional supplementation at the doses studied for enhancing exercise benefits in this population. However, creatine monohydrate may offer targeted benefits for upper-body strength when combined with progressive resistance training, following evidence-based protocols (e.g., a loading phase of 20 g/day for 5-7 days followed by 3-5 g/day maintenance, or 3-5 g/day continuously for 3-4 weeks [35,37]). Adequate baseline protein intake also remains important to support exercise adaptations; among the eight studies reporting baseline dietary protein intake, participants consumed below recommended thresholds for older adults (1.2-1.6 g/kg BW/day) [30,69-70], although whether women during the menopausal transition require similar intake levels warrants further investigation.
Exercise programs should emphasize lower-body and multi-joint exercises with progressive overload to maximize functional benefits and fall prevention [12,61,78], given the absence of lower-extremity strength improvements in our analysis. The metabolic context of an estrogen deficiency during and after the menopausal transition may create distinct challenges for supplementation effectiveness [22,24,31], but interventions implemented before menopause may prevent rather than merely treat deterioration [79]. The absence of reproductive stage-specific subgroup analyses in our review, due to insufficient study numbers, represents an important knowledge gap.
The lack of detectable bone adaptations in short-duration studies using total body measurements suggests that adequate baseline nutrition, optimal exercise programming, and a sufficient intervention duration all require consideration for bone health optimization during this critical transition [80]. Several priority areas for future investigation emerged from our findings: (1) adequately powered trials with intervention durations of at least 12 months and site-specific bone density assessments, incorporating bone turnover markers to evaluate early biochemical responses [18,19,48,49,81]; (2) dose-response studies examining whether higher protein intakes or specific amino acid compositions can overcome estrogen deficiency-related anabolic resistance [22,24,31,82]; (3) comparative effectiveness trials directly comparing supplement types, as our findings suggested that creatine may offer particular advantages for upper-body strength [33,35-38,60-62]; (4) functional outcomes including gait speed, balance, fall incidence, and fracture occurrence [11,13,66,83]; and (5) standardized adverse event monitoring and reporting protocols, as the current evidence base lacks systematic safety data to inform clinical recommendations.
Strengths and limitations
This systematic review represents a comprehensive synthesis of evidence specifically examining combined nutritional and exercise interventions in women across the menopausal transition, addressing a critical knowledge gap [84]. Our pragmatic, inclusive approach to supplement selection provides a broad overview of nutritional strategies studied in this population. The inclusion of diverse protein types and control conditions enhanced the generalizability of the findings. The generally high methodological quality of the included studies, with 64% demonstrating a low risk of bias across all domains, strengthens confidence in our results.
Several important methodological limitations warrant acknowledgment. First, our inclusive approach to participant selection, encompassing women from premenopausal through postmenopausal stages, created heterogeneity that may have obscured stage-specific effects. While this breadth provided insights across the female lifespan [85], it limited our ability to draw definitive conclusions specifically about the menopausal transition. The two studies including exclusively premenopausal women [63,65] represent a distinct metabolic context compared to studies in women experiencing an estrogen deficiency [22,24], yet our decision to retain those studies reflects the clinical reality that musculoskeletal health optimization is relevant across all reproductive stages [86], and preventive interventions before menopause may prove more effective than interventions after deterioration is established [87]. Second, our pooling of mechanistically distinct nutritional supplements—whole proteins, isolated amino acids, and creatine—represents a key methodological consideration. The mechanisms of actions of these compounds substantially differ: whole proteins provide complete amino acid profiles for muscle protein synthesis, isolated amino acids function as specific anabolic signals, and creatine primarily affects cellular energy metabolism through phosphocreatine stores rather than serving as a protein building block [33,35-36]. Pooling these interventions limited the mechanistic interpretability of our effect estimates and prevented isolation of supplement-specific effects. We adopted this approach because: (a) these supplements are frequently studied together in exercise nutrition research as complementary strategies [25,33,35-38,40]; (b) the small number of studies per supplement type (three for creatine, and two or three for other types) precluded adequately powered separate analyses according to Cochrane guidelines requiring ≥10 studies per subgroup [51]; and (c) our objective was to characterize the breadth of nutritional approaches studied in this population rather than to establish causal mechanisms for individual supplements. This heterogeneity is reflected in our GRADE ratings, which were downgraded for indirectness. Readers should interpret our pooled estimates as descriptive summaries of the current literature rather than as precise estimates of the effects of specific supplements. Future research should conduct adequately powered head-to-head comparisons of specific supplement types to provide clearer mechanistic and clinical guidance. Third, bone health outcomes were predominantly assessed using total body measurements over short intervention durations, limiting our ability to evaluate the fracture prevention potential. The median study duration of 16 weeks was insufficient for detecting meaningful changes in BMD, which typically requires 12 months or longer in postmenopausal women [18,19,64,88]. Furthermore, total body measurements might not reflect regional adaptations at weight-bearing sites most relevant to osteoporotic fracture risk [89]. The absence of site-specific measurements of the lumbar spine, femoral neck, or radius represents a significant limitation [77]. Additionally, no included studies reported bone turnover markers, which could have provided insights into early skeletal responses over shorter time frames [90]. The small number of included studies (n=14) precluded planned subgroup analyses that could identify responder characteristics [51]. Only eight of 14 studies reported the baseline dietary protein intake [52-54,57-59,61,65], limiting our ability to determine whether the total protein intake reached recommended thresholds. Substantial heterogeneity in intervention protocols and the relatively short median duration of 16 weeks may have been insufficient for detecting changes in bone tissues, which typically requires 6-12 months for measurable adaptations [91]. The limited assessment of functional outcomes directly related to fall risks [12,92] and the absence of fracture data limited the clinical applicability of findings for osteoporosis prevention. The exclusion of gray literature may have introduced publication bias.
Conclusions
Results of this systematic review demonstrate that nutritional supplementation does not provide consistent additive benefits for muscle mass when combined with exercise in women across reproductive stages. While selective improvements in upper-body strength were observed, particularly in studies incorporating creatine supplementation, lower-extremity strength and muscle mass outcomes showed no significant enhancements. Bone health outcomes assessed in short-duration studies using total body measurements showed non-significant trends, with study limitations precluding definitive conclusions regarding the fracture prevention potential. For clinical practice, exercise alone currently provides the most reliable benefits for musculoskeletal health across women's reproductive stages. Current evidence does not support routine nutritional supplementation at the doses studied for enhancing muscle mass or bone health benefits of exercise. Adequate baseline protein intake remains important to support exercise adaptations, although optimal intake levels for women during the menopausal transition remain to be determined.
Abbreviations
ALM: appendicular lean mass; BIA: bioelectrical impedance analysis; BMC: bone mineral content; BMD: bone mineral density; BMI: body mass index; BW: body weight; CI: confidence interval; DXA: dual-energy x-ray absorptiometry; FFM: fat-free mass; GRADE: Grading of Recommendations Assessment, Development and Evaluation; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; PROSPERO: International Prospective Register of Systematic Reviews; RCT: randomized controlled trial; RoB: risk of bias; RR: risk ratio; SD: standard deviation; SMD: standardized mean difference.
Supplementary Material
Supplementary figures and tables.
Acknowledgements
Funding
This study was supported by a research grant from Wan Fang Hospital, Taipei Medical University (grant no. 111-wf-f-11).
Data availability
All data supporting the findings of this study are included within the study. Additional information can be obtained from the corresponding authors upon reasonable request.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Greendale GA, Sternfeld B, Huang M, Han W, Karvonen-Gutierrez C, Ruppert K. Changes in body composition and weight during the menopause transition. JCI Insight. 2019;4(8):e124865 doi: 10.1172/jci.insight.124865
2. Kim SW, Kim R. The association between hormone therapy and sarcopenia in postmenopausal women: The Korea National Health and Nutrition Examination Survey, 2008-2011. Menopause. 2020;27(5):506-511 doi: 10.1097/GME.0000000000001509
3. Mitchell WK, Williams J, Atherton P, Larvin M, Lund J, Narici M. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength: a quantitative review. Front Physiol. 2012;3:260 doi: 10.3389/fphys.2012.00260
4. Zhang C, Feng X, Zhang X, Chen Y, Kong J, Lou Y. Research progress on the correlation between estrogen and estrogen receptor on postmenopausal sarcopenia. Front Endocrinol (Lausanne). 2024;15:1494972 doi: 10.3389/fendo.2024.1494972
5. Ahlborg HG, Johnell O, Nilsson B, Kröger H, Lorentzon M. Bone loss in relation to menopause: a prospective study during 16 years. J Bone Miner Res. 2001;16(9):1759-1764 doi: 10.1002/jbmr.565
6. Bainbridge KE, Barrett-Connor E, Laughlin GA, von Mühlen D. Natural history of bone loss over 6 years among premenopausal and early postmenopausal women. Am J Epidemiol. 2002;156(5):410-422 doi: 10.1093/aje/kwf043
7. Finkelstein JS, Brockwell SE, Mehta V. et al. Bone mineral density changes during the menopause transition in a multiethnic cohort of women. J Clin Endocrinol Metab. 2008;93(3):861-868 doi: 10.1210/jc.2007-1876
8. Guthrie JR, Nowson CA, Henry MJ. A prospective study of bone loss in menopausal Australian-born women. Osteoporos Int. 1998;8(2):107-112 doi: 10.1007/s001980050129
9. Hansen M, Gliemann L, Hellsten Y. Optimizing female health: the crucial role of exercise initiation before and during the menopausal transition. Exerc Sport Sci Rev. 2025;53(4):195-204 doi: 10.1249/JES.0000000000000371
10. Delanerolle G, Phiri P, Elneil S. et al. Menopause: a global health and wellbeing issue that needs urgent attention. Lancet Glob Health. 2025;13:e196-e198 doi: 10.1016/S2214-109X(24)00528-X
11. Perez-Sousa MA, Venegas-Sanabria LC, Chavarro-Carvajal DA, Cano-Gutierrez CA, Izquierdo M, Correa-Bautista JE, Ramirez-Velez R. Gait speed as a mediator of the effect of sarcopenia on dependency in activities of daily living. J Cachexia Sarcopenia Muscle. 2019;10(5):1009-1015 doi: 10.1002/jcsm.12444
12. El Khoudary SR, Greendale G, Crawford SL, Avis NE, Brooks MM, Thurston RC, Karvonen-Gutierrez C, Waetjen LE, Matthews K. The menopause transition and women's health at midlife: a progress report from the Study of Women's Health Across the Nation (SWAN). Menopause. 2019;26(11):1213-1227 doi: 10.1097/GME.0000000000001424
13. Zhang X, Huang P, Dou Q, Wang C, Zhang W, Yang Y, Wang J. Falls among older adults with sarcopenia dwelling in nursing home or community: a meta-analysis. Clin Nutr. 2020;39(1):33-39 doi: 10.1016/j.clnu.2019.01.002
14. Mandelli A, Tacconi E, Levinger I, Duque G, Hayes A. The role of estrogens in osteosarcopenia: from biology to potential dual therapeutic effects. Climacteric. 2022;25(1):81-87 doi: 10.1080/13697137.2021.1965118
15. Lin YH, Shih YT, Teng MMH. The impact of the “osteo” component of osteosarcopenia on fragility fractures in post-menopausal women. Int J Mol Sci. 2021;22(10):5256 doi: 10.3390/ijms22105256
16. Khalafi M, Habibi Maleki A, Sakhaei MH, Rosenkranz SK, Pourvaghar MJ, Ehsanifar M, Hooshmand Moghadam B. The effects of exercise training on body composition in postmenopausal women: a systematic review and meta-analysis. Front Endocrinol (Lausanne). 2023;14:1183765 doi: 10.3389/fendo.2023.1183765
17. Tan TW, Tan HL, Hsu MF, Huang HL, Chung YC. Effect of non-pharmacological interventions on the prevention of sarcopenia in menopausal women: a systematic review and meta-analysis of randomized controlled trials. BMC Womens Health. 2023;23:606 doi: 10.1186/s12905-023-02749-7
18. Kemmler W, Kohl M, Fröhlich M, Jakob F, Engelke K, von Stengel S, Sieber CC. Effects of high-intensity resistance training on osteopenia and sarcopenia parameters in older men with osteosarcopenia—one-year results of the randomized controlled Franconian Osteopenia and Sarcopenia Trial (FrOST). J Bone Miner Res. 2020;35(9):1634-1644 doi: 10.1002/jbmr.4084
19. Watson SL, Weeks BK, Weis LJ, Harding AT, Horan SA, Beck BR. High-intensity resistance and impact training improves bone mineral density and physical function in postmenopausal women with osteopenia and osteoporosis: the LIFTMOR randomized controlled trial. J Bone Miner Res. 2018;33(2):211-220 doi: 10.1002/jbmr.3284
20. Lim C, Nunes EA, Currier BS, McLeod JC, Thomas ACQ, Phillips SM. An evidence-based narrative review of mechanisms of resistance exercise-induced human skeletal muscle hypertrophy. Med Sci Sports Exerc. 2022;54(9):1546-1559 doi: 10.1249/MSS.0000000000002929
21. Snijders T, Holwerda AM, van Loon LJC, Verdijk LB. Myonuclear content and domain size in small versus larger muscle fibres in response to 12 weeks of resistance exercise training in older adults. Acta Physiol (Oxf). 2021;231(4):e13599 doi: 10.1111/apha.13599
22. Maltais ML, Desroches J, Dionne IJ. Changes in muscle mass and strength after menopause. J Musculoskelet Neuronal Interact. 2009;9(4):186-197 PMID: 19949277
23. Phillips SM. A brief review of critical processes in exercise-induced muscular hypertrophy. Sports Med. 2014;44(Suppl 2):S71-S77 doi: 10.1007/s40279-014-0152-3
24. Moore DR, Churchward-Venne TA, Witard O, Breen L, Burd NA, Tipton KD, Phillips SM. Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J Gerontol A Biol Sci Med Sci. 2015;70(1):57-62 doi: 10.1093/gerona/glu103
25. Jäger R, Kerksick CM, Campbell BI, Cribb PJ, Wells SD, Skwiat TM, Purpura M, Ziegenfuss TN, Ferrando AA, Arent SM, Smith-Ryan AE, Stout JR, Arciero PJ, Ormsbee MJ, Taylor LW, Wilborn CD, Kalman DS, Kreider RB, Willoughby DS, Hoffman JR, Krzykowski JL, Antonio J. International Society of Sports Nutrition Position Stand: protein and exercise. J Int Soc Sports Nutr. 2017;14:20 doi: 10.1186/s12970-017-0177-8
26. Kuo YY, Chang HY, Huang YC, Liu CW. Effect of whey protein supplementation in postmenopausal women: a systematic review and meta-analysis. Nutrients. 2022;14(19):4210 doi: 10.3390/nu14194210
27. Witard OC, Jackman SR, Breen L, Smith K, Selby A, Tipton KD. Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am J Clin Nutr. 2014;99(1):86-95 doi: 10.3945/ajcn.112.055517
28. Yoshimura Y, Matsumoto A, Inoue T, Okamura M, Kuzuya M. Protein supplementation alone or combined with exercise for sarcopenia and physical frailty: a systematic review and meta-analysis of randomized controlled trials. Arch Gerontol Geriatr. 2025;131:105783 doi: 10.1016/j.archger.2025.105783
29. Mertz KH, Reitelseder S, Bechshoeft R, Bulow J, Højfeldt G, Jensen M, Jespersen AP, Holm L. The effect of daily protein supplementation, with or without resistance training for 1 year, on muscle size, strength, and function in healthy older adults: a randomized controlled trial. Am J Clin Nutr. 2021;113(4):790-800 doi: 10.1093/ajcn/nqaa372
30. Zhang L, Liu G, Huang X, He F. Effects of protein supplementation on muscle mass, muscle strength, and physical performance in older adults with physical inactivity: a systematic review and meta-analysis. BMC Geriatr. 2025;25:228 doi: 10.1186/s12877-025-05885-x
31. Sims ST, Kerksick CM, Smith-Ryan AE, Janse de Jonge XAK, Hirsch KR, Arent SM, Antonio J, Arciero PJ, Ormsbee MJ, Desbrow B, Willoughby DS, Kreider RB. International Society of Sports Nutrition position stand: nutritional concerns of the female athlete. J Int Soc Sports Nutr. 2023;20:2204066 doi: 10.1080/15502783.2023.2204066
32. Figueira B, Bravo J, Raimundo A, Lopes V, Marques E, Mota J, Sardinha LB. Effects of a 26-week multicomponent exercise program on cardiovascular and lipid profiles in premenopausal and postmenopausal women. Sci Rep. 2025;15:22485 doi: 10.1038/s41598-025-05378-2
33. Antonio J, Candow DG, Forbes SC, Gualano B, Jagim AR, Kreider RB, Willoughby DS, Stout JR, Smith-Ryan AE, VanDusseldorp TA, Roberts MD, Rawson ES. Common questions and misconceptions about creatine supplementation: what does the scientific evidence really show? J Int Soc Sports Nutr. 2021;18:13 doi: 10.1186/s12970-021-00412-w
34. Brosnan JT, da Silva RP, Brosnan ME. The metabolic burden of creatine synthesis. Amino Acids. 2011;40(5):1325-1331 doi: 10.1007/s00726-011-0853-y
35. Kreider RB, Kalman DS, Antonio J, Ziegenfuss TN, Wildman R, Collins R, Cooke M, Davis JM, Stout JR, Greenwood M, Spano M, Willoughby DS, Campbell B, Zawadzki KM, Bemben MG, Al-Dujaili EAS, Schilling BK, Engelhardt M, Ferreira M, Hiruntrakul A, Kleiner SM, Leutholtz B, Lopez HL, Lowery LM, Mendel R, Smith A, Somma CT, Tresca DM. International Society of Sports Nutrition position stand: safety and efficacy of creatine supplementation in exercise, sport, and medicine. J Int Soc Sports Nutr. 2017;14:18 doi: 10.1186/s12970-017-0173-z
36. Wax B, Kerksick CM, Jagim AR, Mayo JJ, Lyons BC, Kreider RB. Creatine for exercise and sports performance, with recovery considerations for healthy populations. Nutrients. 2021;13(6):1915 doi: 10.3390/nu13061915
37. Hultman E, Söderlund K, Timmons JA, Cederblad G, Greenhaff PL. Muscle creatine loading in men. J Appl Physiol. 1996;81(1):232-237 doi: 10.1152/jappl.1996.81.1.232
38. Smith-Ryan AE, Cabre HE, Eckerson JM, Candow DG. Creatine supplementation in women's health: a lifespan perspective. Nutrients. 2021;13(3):877 doi: 10.3390/nu13030877
39. Li G, Li Z, Liu J. Amino acids regulating skeletal muscle metabolism: mechanisms of action, physical training dosage recommendations and adverse effects. Nutr Metab (Lond). 2024;21:41 doi: 10.1186/s12986-024-00820-0
40. Morton RW, Murphy KT, McKellar SR, Schoenfeld BJ, Henselmans M, Helms E, Aragon AA, Devries MC, Banfield L, Krieger JW, Phillips SM. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br J Sports Med. 2018;52(6):376-384 doi: 10.1136/bjsports-2017-097608
41. Hall J, Chawla M, Watson D. et al. Addressing reproductive health needs across the life course: an integrated, community-based model combining contraception and preconception care. Lancet Public Health. 2023;8(1):e76-e84 doi: 10.1016/S2468-2667(22)00254-7
42. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff J, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71 doi: 10.1136/bmj.n71
43. GRADE Working Group. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. https://gdt.gradepro.org/app/handbook/handbook.html
44. World Health Organization. Musculoskeletal conditions. WHO Fact sheet. 14 July 2022. https://www.who.int/news-room/fact-sheets/detail/musculoskeletal-conditions
45. Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng H-Y, Corbett MS, Eldridge SM, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898 doi: 10.1136/bmj.l4898
46. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-560 doi: 10.1136/bmj.327.7414.557
47. Sterne JA, Sutton AJ, Ioannidis JP, Terrin N, Jones DR, Lau J, Carpenter J, Rücker G, Harbord RM, Schmid CH, Tetzlaff J, Deeks JJ, Peters J, Macaskill P, Schwarzer G, Duval S, Altman DG, Moher D, Higgins JPT. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ. 2011;343:d4002 doi: 10.1136/bmj.d4002
48. Zhao R, Zhao M, Xu Z. The effects of differing resistance training modes on the preservation of bone mineral density in postmenopausal women: a meta-analysis. Osteoporos Int. 2015;26(5):1605-1618 doi: 10.1007/s00198-015-3034-0
49. Kitagawa T, Hiraya K, Denda T, Yamamoto S. A comparison of different exercise intensities for improving bone mineral density in postmenopausal women with osteoporosis: a systematic review and meta-analysis. Bone Rep. 2022;17:101631 doi: 10.1016/j.bonr.2022.101631
50. Jensen SBK, Sørensen V, Sandsdal RM. et al. Bone health after exercise alone, GLP-1 receptor agonist treatment, or combination treatment: a secondary analysis of a randomized clinical trial. JAMA Netw Open. 2024;7(6):e2416775 doi: 10.1001/jamanetworkopen.2024.16775
51. Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, Sterne JAC, Tugwell P, Cochrane Collaboration. Cochrane handbook for systematic reviews of interventions. Version 6.5, Chapter 10, Section 10.11.5.1. London: Cochrane; 2022. https://www.cochrane.org/authors/handbooks-and-manuals/handbook
52. Chapman-Lopez TJ, Funderburk LK, Heileson JL, Wilburn DT, Koutakis P, Gallucci AR. et al. Effects of L-leucine supplementation and resistance training on adipokine markers in untrained perimenopausal and postmenopausal women. J Strength Cond Res. 2024;38(2):526-532 doi: 10.1519/jsc.0000000000004661
53. Funderburk LK, Beretich KN, Chen MD, Willoughby DS. Efficacy of L-leucine supplementation coupled with resistance training in untrained midlife women. J Am Coll Nutr. 2020;39(3):316-324 doi: 10.1080/07315724.2019.1650675
54. Bagheri R, Forbes SC, Candow DG, Wong A. Effects of branched-chain amino acid supplementation and resistance training in postmenopausal women. Exp Gerontol. 2021;144:111185 doi: 10.1016/j.exger.2020.111185
55. Figueroa A, Alvarez-Alvarado S, Ormsbee MJ, Madzima TA, Campbell JC, Wong A. Impact of L-citrulline supplementation and whole-body vibration training on arterial stiffness and leg muscle function in obese postmenopausal women with high blood pressure. Exp Gerontol. 2015;63:35-40 doi: 10.1016/j.exger.2015.01.046
56. Kang Y, Dillon KN, Martinez MA, Maharaj A, Fischer SM, Figueroa A. Combined L-citrulline supplementation and slow velocity low-intensity resistance training improves leg endothelial function, lean mass, and strength in hypertensive postmenopausal women. Nutrients. 2023;15(1):74 doi: 10.3390/nu15010074
57. Trevisan MC, de Souza JMP, Marucci MFN. Influence of soy protein and exercises with weights on the resting energy expenditure of women in post-menopause. Rev Assoc Med Bras (1992). 2010;56(5):572-578 doi: 10.1590/S0104-42302010000500016
58. Orsatti FL, Maestá N, de Oliveira EP, Nahas Neto J, Burini RCP, Nunes PRP. et al. Adding soy protein to milk enhances the effect of resistance training on muscle strength in postmenopausal women. J Diet Suppl. 2018;15(2):140-152 doi: 10.1080/19390211.2017.1330794
59. Maestá N, Nahas EA, Nahas-Neto J, Orsatti FL, Fernandes CE, Traiman P. et al. Effects of soy protein and resistance exercise on body composition and blood lipids in postmenopausal women. Maturitas. 2007;56(4):350-358 doi: 10.1016/j.maturitas.2006.10.001
60. Gualano B, Macedo AR, Alves CRR, Roschel H, Benatti FB, Takayama L. et al. Creatine supplementation and resistance training in vulnerable older women: a randomized double-blind placebo-controlled clinical trial. Exp Gerontol. 2014;53:7-15 doi: 10.1016/j.exger.2014.02.003
61. Chilibeck PD, Candow DG, Gordon JJ, Duff WRD, Mason R, Shaw K. et al. A 2-year randomized controlled trial on creatine supplementation during exercise for postmenopausal bone health. Med Sci Sports Exerc. 2023;55(10):1750-1760 doi: 10.1249/MSS.0000000000003202
62. Chilibeck PD, Candow DG, Landeryou T, Kaviani M, Paus-Jenssen L. Effects of creatine and resistance training on bone health in postmenopausal women. Med Sci Sports Exerc. 2015;47(8):1587-1595 doi: 10.1249/mss.0000000000000571
63. Jendricke P, Centner C, Zdzieblik D, Gollhofer A, König D. Specific collagen peptides in combination with resistance training improve body composition and regional muscle strength in premenopausal women: a randomized controlled trial. Nutrients. 2019;11(4):892 doi: 10.3390/nu11040892
64. Ioannidou P, Dóró Z, Schalla J, Wätjen W, Diel P, Isenmann E. Analysis of combinatory effects of systematic free weight resistance training and a high-protein diet on body composition and strength capacity in postmenopausal women: a 12-week randomized controlled trial. J Nutr Health Aging. 2024;28:100349 doi: 10.1016/j.jnha.2024.100349
65. Murray M, Vlietstra L, Best AMD, Sims ST, Loehr JA, Rehrer NJ. Post-exercise whey protein supplementation: effects on IGF-1, strength, and body composition in pre-menopausal women, a randomised controlled trial. Nutrients. 2025;17(12):2033 doi: 10.3390/nu17122033
66. Cannataro R, Cione E, Bonilla DA, Cerullo G, Angelini F, D'Antona G. Strength training in elderly: an useful tool against sarcopenia. Front Sports Act Living. 2023;5:950949 doi: 10.3389/fspor.2023.950949
67. Wang Q, Jiang X, Shen Y. et al. Effectiveness of exercise intervention on fall-related fractures in older adults: a systematic review and meta-analysis of randomized controlled trials. BMC Geriatr. 2020;20:322 doi: 10.1186/s12877-020-01721-6
68. Kodete CS, Thuraka B, Pasupuleti V, Malisetty S. Hormonal influences on skeletal muscle function in women across life stages: a systematic review. Muscles. 2024;3(3):271-286 doi: 10.3390/muscles3030024
69. Bauer J, Biolo G, Cederholm T, Cesari M, Cruz-Jentoft AJ, Morley JE. et al. Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE Study Group. J Am Med Dir Assoc. 2013;14(8):542-559 doi: 10.1016/j.jamda.2013.05.021
70. Deutz NE, Bauer JM, Barazzoni R, Biolo G, Boirie Y, Bosy-Westphal A. et al. Protein intake and exercise for optimal muscle function with aging: recommendations from the ESPEN Expert Group. Clin Nutr. 2014;33(6):929-936 doi: 10.1016/j.clnu.2014.04.007
71. Bilodeau PA, Coyne ES, Wing SS. The ubiquitin proteasome system in atrophying skeletal muscle: roles and regulation. Am J Physiol Cell Physiol. 2016;311(3):C392-C403 doi: 10.1152/ajpcell.00125.2016
72. Chen Y, Qian M, Gao F, Li G, Peng K, Sun Q. et al. Potential effect of irisin on sarcopenia: a systematic review. BMC Musculoskelet Disord. 2025;26:520 doi: 10.1186/s12891-025-08767-w
73. Norton A, Thieu K, Baumann CW, Lowe DA, Mansky KC. Estrogen regulation of myokines that enhance osteoclast differentiation and activity. Sci Rep. 2022;12:15900 doi: 10.1038/s41598-022-19438-4
74. Kanis JA, Cooper C, Rizzoli R, Reginster JY; Scientific Advisory Board of the European Society for Clinical and Economic Aspects of Osteoporosis (ESCEO) and the Committees of Scientific Advisors and National Societies of the International Osteoporosis Foundation (IOF). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int. 2019;30(1):3-44 doi: 10.1007/s00198-018-4704-5
75. Williams KM, Darukhanavala A, Hicks R, Kelly A. An update on methods for assessing bone quality and health in cystic fibrosis. J Clin Transl Endocrinol. 2021;27:100281 doi: 10.1016/j.jcte.2021.100281
76. Hart NH, Newton RU, Tan J. et al. Biological basis of bone strength: anatomy, physiology and measurement. J Musculoskelet Neuronal Interact. 2020;20(3):347-371 PMID: 32877972
77. International Society for Clinical Densitometry. 2023 ISCD Official Positions—Adult. Published August 24, 2023. Accessed October 17, 2025. https://iscd.org/learn/official-positions/adult-positions/
78. Tøien T, Berg OK, Modena R, Brobakken MF, Wang E. Heavy strength training in older adults: implications for health, disease and physical performance. J Cachexia Sarcopenia Muscle. 2025;16(2):e13804 doi: 10.1002/jcsm.13804
79. Abraham A, Cohen A, Shane E. Premenopausal bone health: osteoporosis in premenopausal women. Clin Obstet Gynecol. 2013;56(4):722-729 doi: 10.1097/GRF.0b013e3182a8ae55
80. Kędzia G, Woźniak M, Samborski W, Grygiel-Górniak B. Impact of dietary protein on osteoporosis development. Nutrients. 2023;15(21):4581 doi: 10.3390/nu15214581
81. Eastell R, Pigott T, Gossiel F, Naylor KE, Walsh JS, Peel NFA. Diagnosis of endocrine disease: bone turnover markers: are they clinically useful? Eur J Endocrinol. 2018;178(1):R19-R31 doi: 10.1530/EJE-17-0585
82. Smith-Ryan AE, Cabre HE, Moore SR. Active women across the lifespan: nutritional ingredients to support health and wellness. Sports Med. 2022;52(Suppl 1):101-117 doi: 10.1007/s40279-022-01755-3
83. Cruz-Jentoft AJ, Sayer AA. Sarcopenia. Lancet. 2019;393(10191):2636-2646 doi: 10.1016/S0140-6736(19)31138-9
84. Sánchez-García JC, López Hernández D, Piqueras-Sola B. et al. Physical exercise and dietary supplementation in middle-aged and older women: a systematic review. J Clin Med. 2023;12(23):7271 doi: 10.3390/jcm12237271
85. Khalaf A, Mathew R, Nayak SG. Exploring symptom clusters across the menopausal stages—systematic review and meta-analysis. Sex Reprod Healthc. 2025;33:100758 doi: 10.1016/j.srhc.2025.100758
86. Shah N, Ariel D. The role of menopausal hormone therapy in the prevention and treatment of low bone density in perimenopausal and postmenopausal women. Curr Opin Obstet Gynecol. 2023;35(2):141-149 doi: 10.1097/GCO.0000000000000858
87. Mohebbi R, Shojaa M, Kohl M. et al. Exercise training and bone mineral density in postmenopausal women: an updated systematic review and meta-analysis of intervention studies with emphasis on potential moderators. Osteoporos Int. 2023;34(7):1145-1178 doi: 10.1007/s00198-023-06682-1
88. Blay R, Flores LE, Kupzyk K. et al. Twelve-month resistance and impact exercise program or risedronate provides a relative benefit to hip bone structure in postmenopausal women: results from a randomized controlled trial. Osteoporos Int. 2024;35(5):877-891 doi: 10.1007/s00198-023-07008-x
89. Warden SJ, Mantila Roosa SM, Kersh ME, Hurd AL, Fuchs RK, Hernandez CJ. Heterogeneous spatial and strength adaptation of the proximal femur to physical activity: a within-subject controlled cross-sectional study. J Bone Miner Res. 2020;35(10):1926-1936 doi: 10.1002/jbmr.4078
90. Schini M, Vilaca T, Gossiel F, Salam S, Eastell R. Bone turnover markers: basic biology to clinical applications. Endocr Rev. 2023;44(3):417-473 doi: 10.1210/endrev/bnac031
91. Wherry SJ, Miller RM, Jeong SH, Beavers KM. The ability of exercise to mitigate caloric restriction-induced bone loss in older adults: a structured review of RCTs and narrative review of exercise-induced changes in bone biomarkers. Nutrients. 2021;13(4):1250 doi: 10.3390/nu13041250
92. Pinheiro MB, Oliveira J, Bauman A. et al. Evidence on physical activity and osteoporosis prevention for people aged 65+ years: a systematic review to inform the WHO guidelines on physical activity and sedentary behaviour. Int J Behav Nutr Phys Act. 2020;17:150 doi: 10.1186/s12966-020-01040-4
Author contact
Corresponding author: I-Hui Chen, School of Nursing, College of Nursing, Taipei Medical University, 250 Wuxing Street, Taipei 11031, Taiwan; E-mail: ichen4edu.tw, Tel: +886-2-27361661 ext. 3619.

 Global reach, higher impact
Global reach, higher impact