Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(5):1884-1895. doi:10.7150/ijms.129084 This issue Cite
Research Paper
Effects of Initial Lactate Levels and 24-h Lactate Clearance on Mortality in Post-Cardiac Arrest Patients: Insights from the Multicenter TIMECARD Registry
1. Department of Critical Care Medicine, Changhua Christian Hospital, Changhua, Taiwan.
2. Department of Emergency Medicine, College of Medicine National Taiwan University, Taipei, Taiwan.
3. Department of Emergency Medicine National Taiwan University Hospital, Taipei, Taiwan.
4. Department of Critical Care Medicine, MacKay Memorial Hospital, Taipei branch, Taiwan.
5. Medical Intensive Care Unit, Cathay General Hospital, Taipei City, Taiwan.
6. Division of Pulmonary and Critical Care Medicine, Kaohsiung Medical University Hospital, Kaohsiung, Taiwan.
7. Department of Internal Medicine, Kaohsiung Medical University Hospital, Kaohsiung, Taiwan.
8. School of Medicine, Graduate Institute of Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan.
9. Division of Cardiology, Department of Internal Medicine, Ditmanson Medical Foundation Chia-Yi Christian Hospital, Chia-Yi, Taiwan
10. Department of Emergency Medicine National Taiwan University Hospital, Taipei, Taiwan.
11. Department of Neurology, Shuang Ho Hospital, Taipei Medical University, New Taipei City, Taiwan.
12. Department of Neurology, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan.
13. Stroke Centre, Shuang Ho Hospital, Taipei Medical University, New Taipei City, Taiwan.
14. Department of Emergency and Critical Medicine, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan.
15. Department of Emergency Medicine, School of Medicine, College of Medicine, Taipei Medical University, Taiwan.
16. Big Data and Digital Promotion Center, Changhua Christian Hospital, Changhua, 500, Taiwan.
17. Graduate Institute of Statistics and Information Science, National Changhua University of Education, Changhua, 500, Taiwan.
18. Graduate Institute of Clinical Medicine, College of Medicine, National Chung Hsing University, Taichung, Taiwan.
Received 2025-11-27; Accepted 2026-2-5; Published 2026-4-8
Abstract
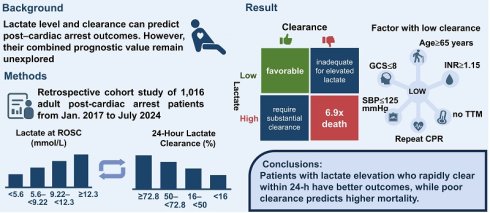
Background: Lactate concentration and clearance are critical indicators of systemic hypoperfusion and can predict post-cardiac arrest outcomes. However, their combined prognostic value and factors influencing clearance achievement remain unexplored.
Methods: This retrospective study analyzed 1,016 adults with cardiac arrest from the Taiwan TIMECARD registry (January 2017-July 2024) who achieved return of spontaneous circulation (ROSC). Patients were stratified into quartiles based on initial lactate levels and 24-hour lactate clearance, defined as the percentage reduction in serum lactate from ROSC to 24 hours. Cox proportional hazards models were used to assess associations with 3-day and 30-day all-cause mortality following ROSC.
Results: Higher initial lactate levels (≥12.3 mmol/L) were associated with increased risks of 3-day (hazard ratio [HR]: 3.42, 95% confidence interval [CI]: 1.88-6.22) and 30-day mortality (HR: 1.88, 95% CI: 1.44-2.46). Poor lactate clearance (<16%) was associated with increased 3-day (HR: 21.05, 95% CI: 9.19-48.22) and 30-day mortality (HR: 2.74, 95% CI: 2.13-3.53). Patients achieving rapid clearance had better mortality outcomes despite moderately elevated initial levels, whereas those with both high initial lactate and poor clearance had a 6.9-fold higher 30-day mortality risk. Factors associated with low clearance included age ≥65 years, INR ≥1.15, Glasgow Coma Scale score ≤8, systolic blood pressure ≤125 mmHg, and repeat cardiopulmonary resuscitation within 1 h; conversely, targeted temperature management was protective.
Conclusion: Routine measurement of lactate levels at ROSC, together with serial monitoring after cardiac arrest, provides clinically meaningful prognostic information. Although initial lactate levels reflect disease severity, their prognostic impact may be modifiable through adequate clearance, whereas poor clearance is associated with an increased risk of mortality. Early identification of patients with inadequate clearance may facilitate targeted interventions to improve survival.
Keywords: lactate levels, lactate clearance, cardiac arrest, return of spontaneous circulation, Taiwan TIMECARD registry, mortality
Introduction
Cardiac arrest (CA), defined as the abrupt cessation of cardiac mechanical activity, remains a leading cause of mortality and morbidity worldwide.1,2 Restoration of spontaneous circulation (ROSC) is a primary objective of cardiopulmonary resuscitation (CPR).3 However, the post-ROSC period is often characterized by complex and evolving systemic disturbances, including cardiovascular instability, tissue hypoperfusion, and metabolic acidosis. Despite advancements in emergency response systems and resuscitation protocols, overall outcomes remain poor.4
The Pan-Asian Resuscitation Outcomes Study clinical research network reported substantial regional differences in out-of-hospital cardiac arrest (OHCA) epidemiology across Asia, with annual emergency medical services-treated OHCA incidence per 100,000 persons of 87.1 in Japan and 47.4 in Singapore. Notably, Seoul demonstrated the highest survival to hospital discharge (9.9%) and favorable neurologic recovery (3.7%).5 In Taiwan, in-hospital cardiac arrest (IHCA) incidence has declined by 70% from 2003 to 2020.6 Conversely, the incidence of OHCA is approximately 51.1 per 100,000 persons, with mortality rates varying over time and worse outcomes observed among older adults, women, and those with more comorbidities.7 In rural regions, ROSC rates are 19.7% and survival to emergency department discharge only 16.4%.8 These findings underscore the critical need for timely and high-quality post-cardiac arrest care.
Serum lactate concentration is a widely used marker of systemic hypoperfusion and cellular oxygen debt.9,10 During cardiac arrest, inadequate oxygen delivery coupled with anaerobic metabolism leads to systemic lactate accumulation. Lactate levels are widely used to assess the severity of circulatory failure11,12 predict post-cardiac arrest clinical outcomes.13-16 However, single-timepoint lactate measurements provide only an initial snapshot and may not fully reflect the evolving physiological status during the early critical hours following cardiac arrest. Lactate kinetics offer additional insights into circulatory recovery and organ perfusion following cardiac arrest. Higher rates of lactate clearance have been associated with improved survival outcomes.17,18 Han et al. identified that lactate clearance exceeding 34% within 6-h serves as a prognostic indicator of favorable outcomes during early post-cardiac arrest management.19 However, most existing studies have evaluated either lactate concentration or lactate clearance, but not both, in relation to post-cardiac arrest prognosis. A physiological continuum-based approach that integrates both parameters may provide a more comprehensive assessment of the severity and trajectory of post-cardiac arrest metabolic derangement. Additionally, no studies have investigated factors influencing lactate clearance rates.
In this study, we investigated the prognostic value of lactate kinetics in patients with ROSC following cardiac arrest. Specifically, we evaluated (1) the associations between lactate-related variables and mortality outcomes; (2) the combined prognostic value of lactate concentration and lactate clearance in predicting mortality risk; and (3) the factors influencing lactate clearance. We hypothesized that the combined assessment of lactate concentration and lactate clearance can be used to enhance patient-specific risk stratification in post-resuscitation care, thereby guiding clinicians in tailoring interventions to improve tissue perfusion and optimize patient outcomes.
Materials and Methods
This retrospective cohort study utilized data from the Taiwan Management for Cardiac Arrest (TIMECARD) multicenter registry database, established by the Taiwan Society of Resuscitation and Critical Care (TSORCC) and described in detail previously.20-22 Briefly, 10 tertiary medical centers participated in the TSORCC collaborative network. The dataset included patients' baseline demographics, pre-existing comorbidities, location of cardiac arrest, resuscitation characteristics and medications, targeted temperature management (TTM) settings, extracorporeal membrane oxygenation (ECMO), documented DNR status, initial arrest rhythm, witnessed arrest, cause of arrest, laboratory data, vital signs, and Glasgow Coma Scale (GCS) scores. Survival status and date of death were also recorded.
We included 1,016 patients who experienced cardiac arrest with successful resuscitation and had at least two lactate measurements between January 2017 and July 2024. This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of Changhua Christian Hospital (No. 251202). Because the analyzed data were deidentified and encrypted, the requirement for informed consent was waived.
Lactate-related variables
The exposures of interest were serum lactate levels at ROSC and 24-hour lactate clearance. Initial lactate was defined as the first serum lactate measurement obtained at ROSC. Patients were stratified into quartiles based on their lactate levels at ROSC: Q1(< 5.6 mmol/L), Q2 (5.6-9.21 mmol/L), Q3 (9.22-12.2 mmol/L) and Q4 (≥ 12.3 mmol/L) (Figure 1).
Study flow-chart.
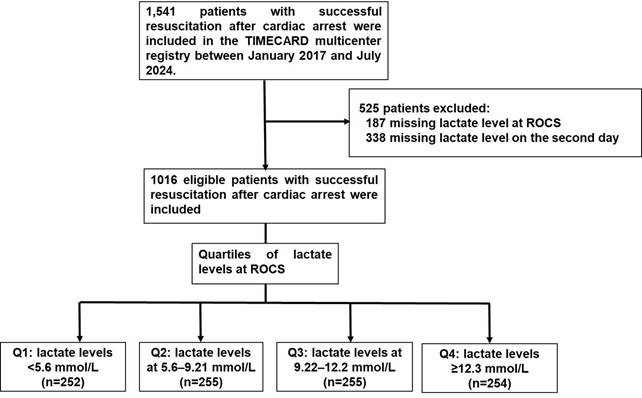
Follow-up lactate measurement was defined as the lactate value recorded on post-ROSC day 1, which represents the closest clinically available measurement approximately 24 hours after ROSC. The 24-hours lactate clearance was calculated as the percentage change from lactate at ROSC to lactate at 24 hours using the following formula:
[(Lactate level at ROSC - Lactate level on post-ROSC day 1)/Lactate level at ROSC] × 100%
Patients were stratified into quartiles of 24-hour lactate clearance, with Q1 representing the highest clearance (≥ 72.8%), indicative of rapid and favorable lactate clearance, followed by Q2 (50%-72.7%), Q3 (16.0%-49.9%), and Q4 (< 16.0%), reflecting progressively poorer lactate clearance.
A risk matrix combining lactate levels at ROSC and 24-hour lactate clearance was constructed to evaluate 30-day mortality risk.
Endpoints
The primary endpoints were 3-day and 30-day all-cause mortality. Patient death records were reviewed to ascertain mortality outcomes. Mortality follow-up was initiated at the time of return of spontaneous circulation (ROSC). Deaths following withdrawal of life-sustaining therapy were included as events. Patients were followed until death or the completion of the respective follow-up period (3-day or 30-day). Patients who were alive at the end of follow-up were censored at that time point.
Statistical analysis
Categorical variables are presented as counts and percentages and were compared using the chi-square test. Continuous variables are expressed as mean ± standard deviation and were compared using one-way analysis of variance. Three lactate-related variables were examined: lactate level at ROSC, second-day lactate level, and 24-hour lactate clearance. Each variable was analyzed as both a continuous measure and as a categorical variable based on quartiles.
Associations between lactate-related variables and mortality outcomes were evaluated using Cox proportional hazards regression models. Crude hazard ratios (cHR) with 95% confidence intervals (95% CI) were obtained from unadjusted models, followed by adjusted hazard ratios (aHR) from models controlling for potential confounders. Subgroup analyses stratified by OHCA and IHCA were performed to assess cardiac arrest location effect modification. Kaplan-Meier survival curves were generated to visualize survival probabilities across lactate categories, and differences were evaluated using the log-rank test.
Logistic regression analysis was performed to identify factors associated with inadequate lactate clearance rates. Restricted cubic spline (RCS) functions were used to explore the relationships between continuous lactate-related variables and 30-day mortality and to identify cutoff values associated with achievement of target lactate clearance rates. Three-dimensional histograms and heatmaps were constructed to illustrate the combined effects of lactate level at ROSC and 24-hour lactate clearance on 30-day mortality.
All statistical analyses were performed using SAS software (version 9.4), and the visualization plot was performed using the “ggplot” package for R software in version 4.5.1. A two-sided p < 0.05 was considered statistically significant.
Results
The mean age of the 1,016 included patients was 66.6±15.3 years, and their body mass index (BMI) was 24.4±5.4. Moreover, 352 (34.6%) and 664 (65.4%) experienced IHCA and OHCA, respectively. Overall, 158 (15.6%) patients died within 3 days and 530 (52.2%) died within 30 days (Table 1).
Patients demographic and clinical variables
| Lactate value at the time of ROSC | Total | P-value | ||||
|---|---|---|---|---|---|---|
| Q1: < 5.6 | Q2:5.6- < 9.22 | Q3:9.22- < 12.3 | Q4: ≥ 12.3 | |||
| Sample size | 252 | 255 | 255 | 254 | 1016 | |
| Age | 68.1±15.2 | 66.7±14.9 | 67.2±15 | 64.5±15.8 | 66.6±15.3 | 0.047 |
| Male | 171 (67.9%) | 185 (72.5%) | 152 (59.6%) | 173(68.1%) | 681 (67%) | 0.018 |
| BMI | 23.9±5.2 | 25.2±5.3 | 24.7±5.3 | 24.1±5.6 | 24.4±5.4 | 0.028 |
| Comorbidity disease | ||||||
| Diabetes mellitus | 117 (46.4%) | 109 (42.7%) | 111 (43.5%) | 85 (33.5%) | 422 (41.5%) | 0.020 |
| Hypertension | 170 (67.5%) | 158 (62%) | 166 (65.1%) | 134 (52.8%) | 628 (61.8%) | 0.004 |
| Coronary artery disease | 80 (31.7%) | 67 (26.3%) | 79 (31%) | 39 (15.4%) | 265 (26.1%) | < 0.001 |
| Heart failure | 49 (19.4%) | 41 (16.1%) | 34 (13.3%) | 26 (10.2%) | 150 (14.8%) | 0.026 |
| Valvular heart disease | 11 (4.4%) | 9 (3.5%) | 13 (5.1%) | 8 (3.1%) | 41 (4%) | 0.683 |
| Arrhythmia | 47 (18.7%) | 36 (14.1%) | 31 (12.2%) | 33 (13%) | 147 (14.5%) | 0.162 |
| COPD | 27 (10.7%) | 21 (8.2%) | 23 (9%) | 21 (8.3%) | 92 (9.1%) | 0.743 |
| Chronic kidney disease | 45 (17.9%) | 41 (16.1%) | 43 (16.9%) | 26 (10.2%) | 155 (15.3%) | 0.075 |
| ESRD | 41 (16.3%) | 34 (13.3%) | 36 (14.1%) | 17 (6.7%) | 128 (12.6%) | 0.008 |
| Liver cirrhosis | 11 (4.4%) | 6 (2.4%) | 4 (1.6%) | 17 (6.7%) | 38 (3.7%) | 0.011 |
| Cerebral vascular accident | 35 (13.9%) | 40 (15.7%) | 32 (12.5%) | 38 (15%) | 145 (14.3%) | 0.762 |
| Dementia | 25 (9.9%) | 10 (3.9%) | 19 (7.5%) | 17 (6.7%) | 71 (7%) | 0.068 |
| Bedridden | 27 (10.7%) | 18 (7.1%) | 17 (6.7%) | 21 (8.3%) | 83 (8.2%) | 0.339 |
| Post tracheostomy | 7 (2.8%) | 3 (1.2%) | 2 (0.8%) | 2 (0.8%) | 14 (1.4%) | 0.170 |
| Hyperlipidemia | 43 (17.1%) | 48 (18.8%) | 30 (11.8%) | 44 (17.3%) | 165 (16.2%) | 0.148 |
| Malignancy | 42 (16.7%) | 45 (17.6%) | 45 (17.6%) | 51 (20.1%) | 183 (18%) | 0.780 |
| CA location | ||||||
| INCA | 131 (52%) | 82 (32.2%) | 74 (29%) | 65 (25.6%) | 352 (34.6%) | < 0.001 |
| OHCA | 121 (48%) | 173 (67.8%) | 181 (71%) | 189 (74.4%) | 664 (65.4%) | |
| CPR medication | ||||||
| Epinephrine | 204 (81%) | 210 (82.4%) | 228 (89.4%) | 237 (93.3%) | 879 (86.5%) | < 0.001 |
| Amiodarone | 45 (17.9%) | 55 (21.6%) | 57 (22.4%) | 52 (20.5%) | 209 (20.6%) | 0.617 |
| Lidocaine | 16 (6.3%) | 19 (7.5%) | 32 (12.5%) | 17 (6.7%) | 84 (8.3%) | 0.038 |
| Calcium gluconate | 26 (10.3%) | 18 (7.1%) | 34 (13.3%) | 33 (13%) | 111 (10.9%) | 0.084 |
| Calcium chloride | 16 (6.3%) | 34 (13.3%) | 31 (12.2%) | 28 (11%) | 109 (10.7%) | 0.060 |
| Bicarbonate | 83(32.9%) | 98 (38.4%) | 107 (42%) | 149 (58.7%) | 437 (43%) | < 0.001 |
| Initial electrocardiogram rhythm | ||||||
| Asystole | 37 (14.68%) | 52 (20.39%) | 69 (27.06%) | 70 (27.56%) | 228 (22.44%) | 0.011 |
| Pulseless Electrical Activity | 108 (42.86%) | 85 (33.33%) | 79 (30.98%) | 90 (35.43%) | 362 (35.63%) | |
| Ventricular Tachycardia or Fibrillation | 80 (31.75%) | 87 (34.12%) | 76 (29.8%) | 67 (26.38%) | 310 (30.51%) | |
| Other | 27 (10.71%) | 31 (12.16%) | 31 (12.16%) | 27 (10.63%) | 116 (11.42%) | |
| Shockable rhythm | 99 (39.29%) | 110 (43.14%) | 112 (43.92%) | 105 (41.34%) | 426 (41.93%) | 0.723 |
| Cause of cardia arrest | ||||||
| non-cardiac | 119 (47.22%) | 117 (45.88%) | 121 (47.45%) | 131 (51.57%) | 488 (48.03%) | 0.604 |
| cardiac | 133 (52.78%) | 138 (54.12%) | 134 (52.55%) | 123 (48.43%) | 528 (51.97%) | |
| Witnessed arrest | 203 (80.56%) | 184 (72.16%) | 173 (67.84%) | 186 (73.23%) | 746 (73.43%) | 0.013 |
| TTM use | 103 (40.9%) | 139 (54.5%) | 141 (55.3%) | 112 (44.1%) | 495 (48.7%) | 0.001 |
| ECMO | 22 (8.73%) | 42 (16.47%) | 57 (22.35%) | 64 (25.2%) | 185 (18.21%) | < 0.001 |
| DNR status | 116 (46.03%) | 130 (50.98%) | 148 (58.04%) | 144 (56.69%) | 538 (52.95%) | 0.025 |
| CPR duration | 13.5±12.2 | 21±17.8 | 22.9±17.7 | 27.7±20.9 | 21.3±18.1 | < 0.001 |
| Vital sign at ROSC | ||||||
| Heart rate | 103.1±31.6 | 104.4±31 | 103.6±29.8 | 108.2±29.3 | 104.8±30.5 | 0.218 |
| SBP | 135.5±40.2 | 132.6±42.3 | 127.7±44.6 | 119.4±40.5 | 128.8±42.3 | < 0.001 |
| DBP | 77.1±24 | 78±27.1 | 73.9±26.7 | 70.3±27 | 74.8±26.4 | 0.004 |
| Body temperature | 36.2±1.3 | 36±1.3 | 35.8±1.2 | 35.5±1.4 | 35.9±1.3 | < 0.001 |
| Lab data at ROSC | ||||||
| PCO2 | 57.5±29.4 | 59±26.6 | 68.7±30.9 | 75.7±35.8 | 65.2±31.7 | < 0.001 |
| PO2 | 112.9±122.2 | 107.4±117.2 | 103.5±114.5 | 99.6±109.7 | 105.9±115.9 | 0.610 |
| HCO3 | 21.3±6.5 | 18.4±5.1 | 16.9±4.7 | 14.4±6 | 17.7±6.2 | < 0.001 |
| WBC count | 12980±7229 | 14209±7165 | 13120±6074 | 14952±9089 | 13816±7500 | 0.008 |
| Hemoglobin | 11.7±3 | 11.9±3.1 | 11.9±3.1 | 11.7±3.5 | 11.8±3.2 | 0.772 |
| Platelet count | 226.2±107 | 1153±14727 | 210±102 | 210±104 | 451±7379 | 0.379 |
| INR | 1.2±0.3 | 1.2±0.6 | 1.3±0.5 | 1.5±1.4 | 1.3±0.8 | < 0.001 |
| Creatinine | 2.7±2.8 | 2.4±2.3 | 2.5±2.6 | 2±1.5 | 2.4±2.4 | 0.002 |
| Sodium | 136.9±7.1 | 136±6.8 | 137±7 | 138.7±7.5 | 137.2±7.1 | < 0.001 |
| Potassium | 4.4±1.3 | 4.4±1.3 | 4.5±1.3 | 4.9±1.4 | 4.5±1.3 | < 0.001 |
| pH | 7.2±0.2 | 7.1±0.2 | 7±0.2 | 7±0.2 | 7.1±0.2 | < 0.001 |
| Sustained ROSC | 211 (83.7%) | 205(80.4%) | 214 (83.9%) | 211 (83.1%) | 841 (82.8%) | 0.699 |
| GCS at ROSC | 7.3±4 | 7.2±3.7 | 6.9±3.3 | 6.2±3.0 | 6.9±3.6 | 0.002 |
| Lactate value at ROSC | 3.6±1.3 | 7.4±1.0 | 10.6±0.8 | 16.1±3.7 | 9.5±5.0 | < 0.001 |
| Lactate value on day 1 | 3.1±3.3 | 4.0±3.2 | 6.2±4.8 | 9.9±8.3 | 5.8±5.9 | < 0.001 |
| 24-h lactate clearance | 25.2±47.9 | 48.2±37.1 | 41.8±44.2 | 41.2±43.1 | 39.3±43.9 | < 0.001 |
| Mortality outcome | ||||||
| 3-day mortality | 17 (6.7%) | 30 (11.8%) | 42 (16.5%) | 69 (27.2%) | 158 (15.6%) | < 0.001 |
| 30-day mortality | 95 (37.7%) | 119 (46.7%) | 145 (56.9%) | 171 (67.3%) | 530 (52.2%) | < 0.001 |
Abbreviations: COPD, chronic obstructive pulmonary disease; ESRD, end-stage renal disease; CA, cardiac arrest; IHCA, in-of-hospital cardiac arrest; OHCA, out-of-hospital cardiac arrest; CPR, cardiopulmonary resuscitation; ROSC, return of spontaneous circulation; TTM, targeted temperature management; ECMO, extracorporeal membrane oxygenation; DNR, do-not-resuscitate; SBP, systolic blood pressure; DBP, diastolic blood pressure; WBC, white blood cell; INR, International Normalized Ratio; GCS, Glasgow Coma Scale scores
Table 1 lists the baseline characteristics of the patients with ROSC stratified by quartiles of lactate levels at ROSC: < 5.6 mmol/L (n = 252), 5.6-9.21 mmol/L (n = 255), 9.22-12.2 mmol/L (n = 255), and ≥ 12.3 mmol/L (n = 254) (Figure 1). Significant between-group differences were observed in baseline demographics and comorbidities. Compared with the other lactate quartiles, patients in the highest lactate quartile (≥ 12.3 mmol/L) were younger, had lower BMI, and had a lower prevalence of diabetes mellitus, hypertension, coronary artery disease, heart failure and end-stage renal disease on dialysis but had a higher prevalence of liver cirrhosis (all p < 0.05). IHCA predominated in the lowest lactate quartile (Q1: 52%), whereas OHCA was more frequent in highest quartile (Q4: 74.4%, p < 0.001). Overall, a cardiac etiology was identified in 51.97% of cardiac arrest cases, and a shockable rhythm was present in 41.93% of patients; however, neither differed significantly across lactate quartiles (p = 0.604 and 0.723, respectively). Ventricular tachycardia or ventricular fibrillation was observed in 30.51% of patients. Witnessed arrests were more common in Q1, accounting for 73.43% of cases.
Regarding resuscitation medications, the use of epinephrine and bicarbonate increased progressively with higher lactate levels (both p < 0.001). Lidocaine use was more common in Q3 (p = 0.038). CPR duration increased stepwise from 13.5 ± 12.2 min in the lowest lactate quartile to 27.7 ± 20.9 min in the highest (p < 0.001). Targeted temperature management (TTM) was most frequently applied in Q3 (55.3%), whereas extracorporeal membrane oxygenation (ECMO) was most commonly used in Q4 (25.2%).
Patients in higher lactate quartiles at ROSC exhibited lower systolic and diastolic blood pressures, body temperature, pH, and bicarbonate (HCO₃) as well as higher partial pressure of carbon dioxide (PCO₂), serum sodium, and serum potassium (all p < 0.01); these changes are consistent with more severe metabolic acidosis and hemodynamic instability.
Mortality rates rose progressively with lactate level: 3-day mortality increased from 6.7% in Q1 to 27.2% in Q4, and 30-day mortality increased from 37.7% to 67.3%, respectively (both p < 0.001).
Association of lactate-related variables with mortality
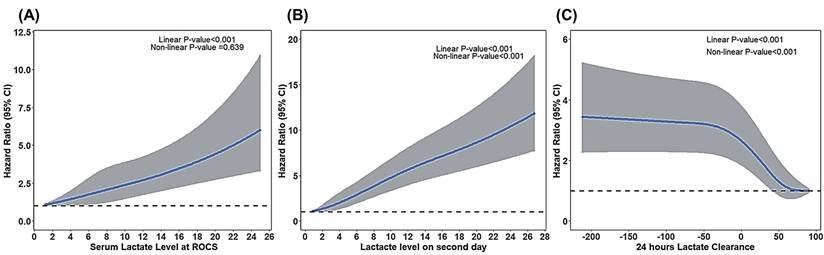
In Figure 2, the RCS curve exhibits a continuous positive correlation between lactate-related variables and 30-day mortality. Higher lactate levels and lower lactate clearance were associated with an increased mortality risk. Notably, the 24-hour lactate clearance (Figure 2c) exhibited a nonlinear relationship: less or negative lactate clearance—indicating slower clearance or rising lactate levels—was linked to substantially higher mortality.
Restricted cubic spline showing the effects of lactate-related value on 30-day mortality.
In Figure 3, Kaplan-Meier survival analysis revealed a clear separation of survival curves across lactate quartiles, demonstrating a significant association between higher lactate levels and reduced 30-day survival following cardiac arrest. Patients with the highest lactate at ROSC exhibited substantially lower survival throughout the 30-day follow-up period than the other groups. Similar patterns were observed for second-day lactate levels. Lactate clearance demonstrated a significant inverse association with survival; patients with inadequate lactate clearance (<50%) had markedly poorer survival outcomes than those achieving rapid clearance. The log-rank test confirmed significant differences in survival across lactate categories for all comparisons (p < 0.001).
Kaplan-Meier curve for 30-day mortality based on quartiles of lactate-related categorical variables.
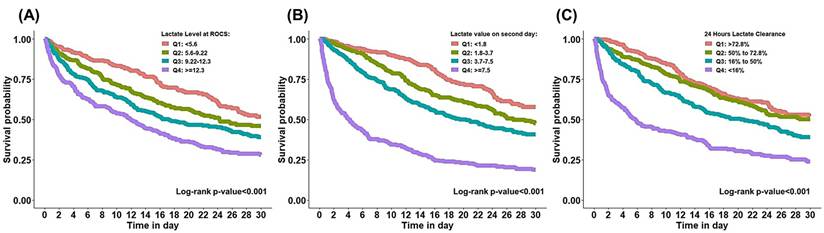
Table 2 and Table 3 summarizes the results of multivariable Cox regression analysis of categorical lactate-related variables: lactate at ROSC, second-day lactate levels, and lactate clearance were independently associated with both 3-day and 30-day mortality (all p < 0.001).
The hazard ratio of lactate-related variables as a categorical variable based on quartiles on 3-day mortality
| Lactate-related variables | Cardio-arrest patients (n=1016) | OHCA | IHCA | Pinteraction | |||
|---|---|---|---|---|---|---|---|
| cHR (95% CI) | P-value | aHR (95% CI) | P-value | aHR (95% CI) | aHR (95% CI) | ||
| lactate value at the time of ROSC | |||||||
| Q1:< 5.6 | 1 | 1 | 1 | 1 | |||
| Q2:5.6 - < 9.22 | 1.78 (0.98,3.22) | 0.058 | 1.65(0.89,3.04) | 0.1093 | 1.94 (0.81,4.62) | 1.52 (0.65,3.56) | 0.700 |
| Q3:9.22 - < 12.3 | 2.55 (1.45,4.48) | 0.001 | 1.98(1.09,3.57) | 0.0241 | 1.73 (0.71,4.24) | 2.37 (1.07,5.25) | 0.604 |
| Q4:≥ 12.3 | 4.59 (2.70,7.81) | < 0.001 | 3.42(1.88,6.22) | <. 0001 | 2.88 (1.23,6.77) | 3.67 (1.69,8.01) | 0.680 |
| Lactate value on day 1 | |||||||
| Q1:< 1.8 | 1 | 1 | 1 | 1 | |||
| Q2: 1.8 - < 3.7 | 1.23 (0.51,2.97) | 0.646 | 1.30(0.54,3.16) | 0.560 | 3.20 (0.48,21.22) | 0.93 (0.34,2.52) | 0.257 |
| Q3:3.7 - < 7.5 | 2.74 (1.27,5.90) | 0.010 | 2.97(1.37,6.43) | 0.006 | 4.56 (0.69,30.32) | 2.26 (1.00,5.09) | 0.503 |
| Q4: ≥ 7.5 | 17.11 (8.67,33.74) | < 0.001 | 15.76(7.87,31.55) | < 0.001 | 42.98 (8.04,229.59) | 9.36 (4.49,19.51) | 0.101 |
| Lactate clearance | |||||||
| Q1≥ 72.8% | 1 | 1 | 1 | 1 | |||
| Q2:50% - < 72.8% | 3.43 (1.38,8.55) | 0.008 | 3.44(1.38,8.57) | 0.008 | 6.18 (0.30,128.5) | 3.03 (1.22,7.54) | 0.659 |
| Q3:16.0% - < 50% | 5.56 (2.33,13.3) | < 0.001 | 5.65(2.35,13.55) | < 0.001 | 14.88 (0.78,283.03) | 4.24 (1.78,10.13) | 0.424 |
| Q4:< 16.0% | 21.78 (9.55,49.65) | < 0.001 | 21.05(9.19,48.22) | < 0.001 | 78.00 (4.47,1360.75) | 12.64 (5.58,28.64) | 0.230 |
Abbreviations: IHCA, in-of-hospital cardiac arrest; OHCA, out-of-hospital cardiac arrest; cHR, crude hazard ratio; aHR, adjusted hazard ratio; CI, confidence interval;
aHRs were estimated using multivariable Cox proportional hazards regression models with a backward elimination procedure. In step 1, the models were adjusted for potential confounders, including age, sex, BMI, comorbid conditions, OHCA/INCA, CPR medications, vital signs at ROSC, laboratory data at ROSC, resuscitation characteristics (sustained ROSC, CPR duration, cause of cardiac arrest, initial electrocardiogram rhythm, shockable rhythm, and witnessed arrest), GCS score, post-resuscitation interventions (TTM and ECMO), and do-not-resuscitate (DNR) status
The hazard ratio of lactate-related variables as a categorical variable based on quartiles on 30-day mortality
| Lactate-related variables | Cardio-arrest patients (n=1016) | OHCA | IHCA | Pinteraction | ||||
|---|---|---|---|---|---|---|---|---|
| cHR (95% CI) | P-value | aHR (95% CI) | P-value | aHR (95% CI) | aHR (95% CI) | |||
| Lactate value at the time of ROSC | ||||||||
| Q1:< 5.6 | 1 | 1 | 1 | 1 | ||||
| Q2:5.6 - < 9.22 | 1.36 (1.03,1.77) | 0.027 | 1.41 (1.07,1.85) | 0.015 | 1.72 (1.12,2.64) | 1.18 (0.82,1.70) | 0.191 | |
| Q3:9.22 - < 12.3 | 1.70 (1.32,2.21) | < 0.001 | 1.59 (1.22,2.07) | < 0.001 | 1.49 (0.97,2.28) | 1.53 (1.08,2.17) | 0.919 | |
| Q4:≥12.3 | 2.42 (1.88,3.11) | < 0.001 | 1.88 (1.44,2.46) | < 0.001 | 2.23 (1.45,3.42) | 1.62 (1.14,2.3) | 0.252 | |
| Lactate value on day 1 | ||||||||
| Q1:< 1.8 | 1 | 1 | 1 | 1 | ||||
| Q2: 1.8 - < 3.7 | 1.51 (1.13,2.03) | 0.006 | 1.41 (1.04,1.89) | 0.025 | 1.88 (1.14,3.12) | 1.21 (0.84,1.76) | 0.167 | |
| Q3:3.7 - < 7.5 | 2.02 (1.53,2.68) | < 0.001 | 1.66 (1.24,2.21) | < 0.001 | 1.85 (1.09,3.12) | 1.54 (1.09,2.17) | 0.572 | |
| Q4: ≥7.5 | 4.92 (3.77,6.42) | < 0.001 | 3.83 (2.88,5.07) | < 0.001 | 6.15 (3.76,10.08) | 2.88 (2.06,4.02) | 0.011 | |
| Lactate clearance | ||||||||
| Q1≥72.8% | 1 | 1 | 1 | 1 | ||||
| Q2:50% - < 72.8% | 1.13 (0.86,1.49) | 0.379 | 1.14 (0.86,1.51) | 0.358 | 1.33 (0.79,2.26) | 1.08 (0.77,1.50) | 0.495 | |
| Q3:16.0% - < 50% | 1.59 (1.22,2.06) | 0.001 | 1.49 (1.15,1.93) | 0.003 | 1.68 (1.00,2.82) | 1.42 (1.05,1.92) | 0.582 | |
| Q4:< 16.0% | 3.05 (2.38,3.91) | < 0.001 | 2.74 (2.13,3.53) | < 0.001 | 3.15 (1.96,5.07) | 2.66 (1.97,3.59) | 0.548 | |
Abbreviations: IHCA, in-of-hospital cardiac arrest; OHCA, out-of-hospital cardiac arrest; cHR, crude hazard ratio; aHR, adjusted hazard ratio; CI, confidence interval;
aHRs were estimated using multivariable Cox proportional hazards regression models with a backward elimination procedure. In step 1, the models were adjusted for potential confounders, including age, sex, BMI, comorbid conditions, OHCA/INCA, CPR medications, vital signs at ROSC, laboratory data at ROSC, resuscitation characteristics (sustained ROSC, CPR duration, cause of cardiac arrest, initial electrocardiogram rhythm, shockable rhythm, and witnessed arrest), GCS score, post-resuscitation interventions (TTM and ECMO), and do-not-resuscitate (DNR) status.
Table 2 presents aHRs with 95% CI for 3-day mortality. Patients with lactate at ROSC ≥ 12.3 mmol/L had an approximately 3.4-fold higher adjusted hazard of death compared with the lowest category (< 5.6 mmol/L) (aHR: 3.42, 95% CI: 1.88-6.22, p < 0.001). Intermediate categories also displayed an elevated risk, with aHR of 1.65 (95% CI: 0.89-3.04) for 5.6-9.1 mmol/L and 1.98 (95% CI: 1.09-3.57) for 9.22-12.2 mmol/L. Second-day lactate ≥ 7.5 mmol/L was associated with markedly higher mortality risk (aHR: 15.76, 95% CI: 7.87-31.55, p < 0.001), whereas intermediate second-day lactate levels (3.7-7.4 mmol/L) also conferred increased risk (aHR: 2.97, 95% CI: 1.37-6.43, p = 0.006). Lactate clearance < 16% was associated with substantially elevated short-term mortality risk (aHR: 21.05, 95% CI: 9.19-48.22, p < 0.001). Intermediate clearance categories also exhibited an elevated risk, with aHRs of 5.65 (95% CI: 2.35-13.55) and 3.44 (95% CI: 1.38-8.57) for clearance of 16.0%-49.9% and 50%-72.7%, respectively, compared with optimal clearance rate at ≥ 72.8%.
For 30-day mortality in Table 3, lactate at ROSC ≥ 12.3 mmol/L was associated with an aHR of 1.88 (95% CI: 1.44-2.46, p < 0.001). Second-day lactate ≥ 7.5 mmol/L conferred an aHR of 3.83 (95% CI: 2.88-5.07, p < 0.001). Lactate clearance < 16% remained associated with increased mortality risk (aHR: 2.74, 95% CI: 2.13-3.53, p < 0.001).
Across both outcomes, the results supported the prognostic importance of high initial lactate, elevated second-day lactate, and unfavorable lactate kinetics. Greater lactate clearance was linked to improved survival. No significant interactions were observed between OHCA and IHCA subgroups, except that patient with OHCA with second-day lactate ≥ 7.5 mmol/L demonstrated greater 30-day mortality risk than patients with IHCA.
Combined effects of lactate-related variables and 30-day mortality
Mortality risk varied across different combinations of initial lactate levels at ROSC and 24-h lactate clearance, with a significant interaction between the two variables (P for interaction < 0.001). Using the lowest quartile (Q1) of both lactate at ROSC and lactate clearance as the reference category, distinct patterns emerged based on clearance rates.
In Figure 4, patients with lactate clearance > 72.8% demonstrated favorable mortality outcomes across all baseline lactate levels at ROSC (HR: 0.836-1.006), indicating that robust early lactate clearance is protective regardless of initial lactate burden.
Three-dimensional histogram and heatmap showing the hazard ratio of lactate value at ROCS and 24-h lactate clearance on 30-day mortality.
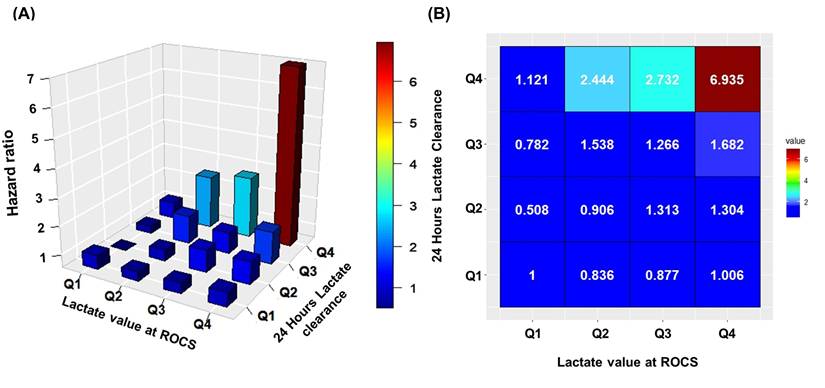
For patients with suboptimal clearance (50%-72.7%), favorable outcomes were limited to those with low baseline lactate levels, as evidenced by an HR of 0.508 for Q1 (< 5.6 mmol/L) and 0.906 for Q2 (5.6-9.21 mmol/L). Conversely, patients with higher baseline lactate levels (Q3 and Q4) experienced substantially increased mortality risk (HR: 1.313-1.304).
At moderate clearance rate (Q3: 16.0%-49.9%), mortality outcomes again depended on baseline lactate levels, as evidenced by a low HR of 0.782 for Q1. However, those with higher baseline lactate (Q2-Q4) experienced progressively increased mortality hazard (HR: 1.226-1.682). These results indicated that moderate clearance is inadequate for managing elevated initial lactate.
Poor or negative lactate clearance combined with high baseline lactate was associated with the greatest mortality risk. Patients with high lactate at ROSC (≥ 12 mmol/L) and poor clearance or ongoing lactate accumulation demonstrated a 6.9-fold increased hazard of 30-day mortality (HR: 6.938).
These findings indicate that patients with higher lactate levels at ROSC who achieved substantial clearance (> 73%) within 24 hours demonstrated lower mortality risk, whereas those with low lactate levels at ROSC showed favorable outcomes even with lower clearance rates. Inadequate clearance relative to initial lactate burden was associated with progressively increased 30-day mortality risk.
Factors contributing to inadequate and poor lactate clearance
Multivariate logistic regression analysis revealed four variables significantly associated with inadequate lactate clearance (< 50%): age ≥ 65 years (adjusted odds ratio [aOR]: 1.357; 95% CI: 1.043-1.764; p = 0.023), INR ≥ 1.15 (aOR: 1.690; 95% CI: 1.305-2.188; p < 0.001), GCS score ≤ 8 (aOR: 2.470; 95% CI: 1.759-3.468; p < 0.001), and systolic blood pressure (SBP) ≤ 125 mmHg (aOR: 1.771; 95% CI: 1.369-2.290; p < 0.001). Bicarbonate use was not statistically significant (aOR: 1.188; 95% CI: 0.916-1.541; p = 0.194) (Figure 5A).
Risk factor of inadequate clearance and poorly lactate clearance.
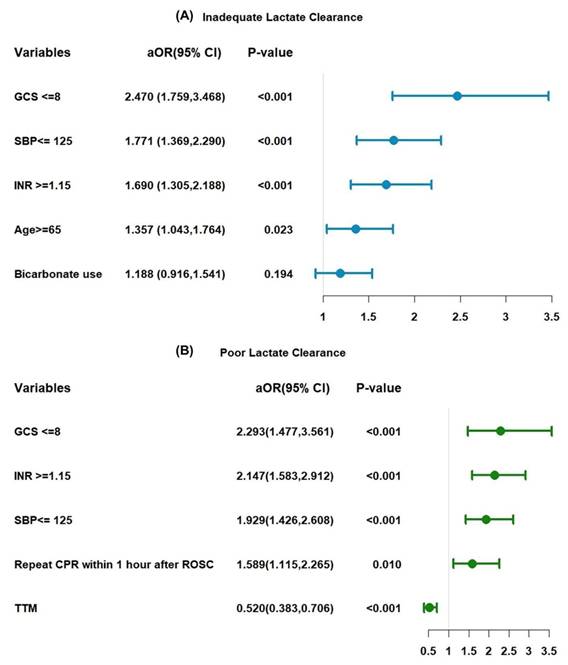
Figure 5B presents the results for poor lactate clearance, defined as a clearance rate of < 16%. Significant predictors included GCS score ≤ 8 (aOR: 2.293; 95% CI: 1.477-3.561; p < 0.001), INR ≥ 1.15 (aOR: 2.147; 95% CI: 1.583-2.912; p < 0.001), and SBP ≤ 125 mmHg (aOR: 1.929; 95% CI: 1.426-2.608; p < 0.001). Additionally, repeat CPR within 1-h after ROSC increased the likelihood of poor lactate clearance (aOR: 1.589; 95% CI: 1.115-2.265; p = 0.010), whereas TTM was a protective factor (aOR: 0.520; 95% CI: 0.383-0.706; p < 0.001). The cutoff points for each factor were determined using RCS analysis (Figure S1).
Discussion
Our analysis of a large, multicenter registry of patients after cardiac arrest revealed that absolute lactate levels at ROSC, second-day lactate levels, and 24-h lactate clearance were independently associated with both 3-day and 30-day mortality. The highest mortality rates were observed in patients with lactate at ROSC ≥ 12.3 mmol/L or second-day lactate ≥ 7.5 mmol/L. Furthermore, unfavorable lactate kinetics—reflected by inadequate or absent lactate decline within 24 h—substantially amplified mortality risk. Combined analysis revealed that patients with higher initial lactate levels who achieved outcomes comparable to those with lower initial lactate demonstrated progressively greater clearance rates. Among clinicodemographic variables, older age, higher INR, lower GCS score, lower SBP, repeated CPR within 1 h after ROSC, and bicarbonate use were significantly associated with poor lactate clearance, whereas TTM was associated with improved lactate clearance.
The strong association between elevated lactate levels and mortality likely reflects the combined effects of systemic hypoperfusion, impaired oxygen utilization, and metabolic derangements during and after resuscitation23-25. High lactate at ROSC indicates prolonged low-flow states, delayed clearance, or ongoing tissue hypoxia, all of which are markers of severe post-cardiac arrest syndrome26-28. Serum lactate > 4 mmol/L is generally considered a threshold for increased mortality26,29-32. However, no consensus exists regarding the optimal lactate range for patients after cardiac arrest, as it varies by the timing of measurement and patient population. In IHCA, mortality risk increases with lactate levels: 44% for < 5 mmol/L, 58% for 5-10 mmol/L, and 73% for > 10 mmol/L33. In nontraumatic OHCA, lactate cutoffs of 9.1 and 9.4 mmol/L predict 24-h and 48-h mortality, respectively34. In another multicenter OHCA cohort, survival dropped sharply when lactate exceeded 10.6 mmol/L35. Consistently, our findings demonstrate a linear positive relationship between lactate at ROSC and 30-day mortality, with initial lactate of < 5.6 mmol/L and second-day lactate of < 3.7 mmol/L associated with better survival.
Rapid lactate clearance reflects effective restoration of tissue oxygenation and resolution of anaerobic metabolism, whereas persistently high or increasing lactate levels suggest ongoing hypoperfusion, mitochondrial dysfunction, or continued metabolic stress. Inadequate clearance strongly predicts poor outcomes18,28,36. Specifically, failure to achieve significant reduction within 24-hour, such as not reaching thresholds of 1.55-3.1 mmol/L, and 34% or <65% clearance at 6 h has been linked to poor survival18,36-38. Our results are consistent, showing that clearance greater than 72.8% within 24-hour is associated with improved survival. While early lactate clearance at 6 hours primarily reflects acute resuscitation adequacy and guides immediate interventions, 24-hour clearance may better capture sustained metabolic recovery and identify patients at risk for delayed deterioration despite initial hemodynamic stabilization.
Our findings demonstrate that baseline lactate and clearance are not independent prognostic markers but a significant interplay component of a physiologic continuum, thus providing a more comprehensive risk assessment than isolated measurements. The association between lactate clearance and mortality was modified by initial lactate levels. Specifically, patients with low baseline lactate (< 5.6 mmol/L) showed favorable outcomes across a range of clearance rates, whereas those with high baseline lactate (≥ 12.3 mmol/L) with clearance > 72.8% within 24-hour associated with mortality rates comparable to lower-risk patients. These observations may inform risk stratification and clinical decision-making. Patients with high initial lactate and inadequate clearance represent a high-risk subset who may warrant closer monitoring and thorough assessment of resuscitation adequacy. However, given the observational nature of this study, we cannot establish causality between lactate clearance and improved survival. We do not claim that interventions specifically targeting a clearance threshold of ≥ 72.8% will directly improve clinical outcomes. Instead, lactate clearance likely acts as a surrogate marker of global physiological recovery in response to appropriate resuscitation, rather than serving as an independent therapeutic target. Future prospective trials are needed to evaluate whether lactate clearance-guided protocols improve clinical outcomes compared to standard care. The limited effect of bicarbonate administration implies that buffering alone is insufficient39; instead, treatment should focus on underlying causes of hyperlactatemia, optimizing cardiac output, improving perfusion, enhancing oxygen delivery, and correcting metabolic derangements.
We identified several factors associated with low lactate clearance (age ≥ 65 years, INR ≥ 1.15, GCS score ≤ 8, SBP ≤ 125 mmHg, and repeat CPR within 1 hour after ROSC), which may help clinicians recognize patients at risk of impaired metabolic recovery and guide targeted interventions. Gissel et al. demonstrated that coagulation dynamics are significantly impaired when pH falls to levels typical of metabolic acidosis in severe trauma. Inadequate tissue perfusion leads to hypoxia, anaerobic metabolism, and lactic acid accumulation, and the resulting acidosis inhibits key coagulation enzymes, exacerbating coagulopathy40. In our study, the mean pH was 7.1 ± 0.2, indicating clinically significant acidosis, which likely contributed to both impaired coagulation and reduced lactate metabolism. Impaired perfusion not only promotes lactate buildup but also hinders its clearance, as demonstrated by He et al. in patients with sepsis41. In our cohort, elevated INR, which is a marker of coagulopathy, may similarly reflect impaired tissue perfusion and oxygen delivery, linking coagulation dysfunction to reduced lactate clearance. High INR may therefore serve as a biomarker for identifying patients at risk of poor lactate clearance; further research is warranted to comprehensively investigate this relationship.
Although TTM does not directly reduce lactate levels, it stabilizes metabolic processes disrupted by ischemia, improves glucose metabolism, and reduces lactate production. TTM may also enhance organ perfusion by stabilizing the blood-brain barrier and mitigating endothelial dysfunction42, thereby creating favorable conditions for recovery and promoting lactate clearance. In our cohort, TTM was significantly associated with improvement in mild lactate clearance (< 16%) but had no effect in cases with moderate reductions (< 50%), suggesting that TTM should be considered a supportive strategy rather than a primary intervention for lactate elimination.
The strengths of this study include the use of a large, multicenter registry database; integrated assessment of multiple lactate-related variables; adjustment for key confounders; and identification of factors associated with poor clearance. The study also had some limitations. First, this was a retrospective observational study, which precludes the establishment of causal relationships. Second, the TIMECARD registry does not capture data on arrest downtime, CPR quality, or the timing of DNR documentation relative to cardiac arrest, all of which may reflect underlying illness severity and influence goals of care decisions. Third, unmeasured post-ROSC interventions—including vasopressor strategies, fluid management, hemodynamic optimization protocols, and institutional practice variations—represent potential sources of residual confounding that may influence the observed associations. Finally, the identified lactate thresholds should be regarded as hypothesis-generating, intended to inform risk stratification rather than define fixed treatment targets. Future studies should prospectively validate both the conceptual framework and specific numerical thresholds across diverse healthcare settings with varying prehospital response capabilities, post-cardiac arrest care protocols, and TTM practices before these values are adopted into clinical practice.
Conclusion
Our results support the use of serial lactate measurement from ROSC through the early post-resuscitation period to provide meaningful prognostic information after cardiac arrest. Importantly, the prognostic impact of 24-hour lactate clearance dependent on the initial lactate level at ROSC. Patients with moderately to highly elevated lactate levels at ROSC demonstrated a strong association between higher 24-hour lactate clearance and more favorable outcomes, whereas poor clearance was associated with markedly increased risk of mortality. These findings establish lactate clearance as a baseline-adjusted prognostic biomarker that provides dynamic information regarding physiological recovery during post-cardiac arrest care, particularly in patients with marked metabolic derangement. Lactate clearance reflects the adequacy of resuscitative efforts aimed at restoring tissue perfusion and cellular oxygen delivery, rather than serving as a direct therapeutic target. Early identification of patients with high lactate levels and insufficient clearance may assist in risk stratification, inform decisions regarding the intensity of ongoing supportive care, and help assess whether resuscitative interventions are achieving their intended physiological effect during the post-cardiac arrest care continuum.
Abbreviations
TSORCC : Taiwan society of resuscitation and critical care; TIMECARD: Taiwan management for cardiac arrest multicenter registry; COPD: chronic obstructive pulmonary disease; ESRD: end-stage renal disease; CA: cardiac arrest; IHCA: in-of-hospital cardiac arrest; OHCA: out-of-hospital cardiac arrest; CPR: cardiopulmonary resuscitation; ROSC: return of spontaneous circulation; TTM: targeted temperature management; SBP: systolic blood pressure; DBP: diastolic blood pressure; WBC: white blood cell; INR: international normalized ratio; GCS: Glasgow coma scale scores; cHR: crude hazard ratio; aHR: adjusted hazard ratio; OR: odds ratio; 95% CI: 95% confidence interval; RCS: restricted cubic spline.
Supplementary Material
Supplementary figure.
Acknowledgements
The authors would like to acknowledge the Taiwan Society of Resuscitation and Critical Care (TSORCC) for providing access to the Taiwan Management for Cardiac Arrest (TIMECARD) multicenter registry database.
Funding
This study was supported by grants from the Changhua Christian Hospital, grant number 114-CCH-IRP-054. The study sponsors were not involved in the study design, in the collection of data, or in the forthcoming analyses and interpretation of data, writings of manuscript of manuscripts or in the decisions to submit manuscripts for publication.
Data availability statement
The data underlying this article are available from the TIMECARD database but cannot be publicly released due to ethical and privacy restrictions. Additional information regarding data access may be obtained upon reasonable request and with permission from the TSORCC (https://www.tsorcc.org.tw/timecard-formosa-registry).
Competing Interests
The authors have declared that no competing interest exists.
References
1. Berdowski J, Berg RA, Tijssen JG, Koster RW. Global incidences of out-of-hospital cardiac arrest and survival rates: systematic review of 67 prospective studies. Resuscitation. 2010;81:1479-87
2. Gräsner JT, Bottiger BW, Bossaert L. European Registry of Cardiac Arrest (EuRe-Ca) ONE Steering Committee; EuReCa ONE Study Management Team. EuReCa ONE-ONE month-ONE Europe-ONE goal. Resuscitation. 2014;85:1307-8
3. McCarthy JJ, Carr B, Sasson C, Bobrow BJ. et al. Out-of-hospital cardiac arrest resuscitation systems of care: a scientific statement from the American Heart Association. Circulation. 2018;137(21):e645-e660
4. Važanić D, Prkačin I, Nesek-Adam V, Kurtović B, Rotim C. Out-of-hospital cardiac arrest outcomes - bystander cardiopulmonary resuscitation rate improvement. Acta Clin Croat. 2022Aug;61(2):265-272
5. Oh YS, Ahn KO, Shin SD, Kagino K. et al. Variability in the effects of prehospital advanced airway management on outcomes of patients with out-of-hospital cardiac arrest. Clin Exp Emerg Med. 2020Jun;7(2):95-106
6. Chang FC, Hsieh MJ, Yeh JK, Wu VC. et al. Longitudinal analysis of in-hospital cardiac arrest: trends in the incidence, mortality, and long-term survival of a nationwide cohort. Crit Care. 2025Jan23;29(1):41
7. Wang CY, Wang JY, Teng NC, Chao TT. et al. The secular trends in the incidence rate and outcomes of out-of-hospital cardiac arrest in Taiwan-a nationwide population-based study. PLoS One. 2015Apr15;10(4):e0122675
8. Hung SC, Mou CY, Hung HC, Lai SW. et al. Non-traumatic out-of-hospital cardiac arrest in rural Taiwan: A retrospective study. Aust J Rural Health. 2017Dec;25(6):354-361 [Epub ahead of print]
9. Deulkar P, Singam A, Mudiganti VNKS, Jain A. Lactate Monitoring in Intensive Care: A Comprehensive Review of Its Utility and Interpretation. Cureus. 2024Aug7;16(8):e66356 doi: 10.7759/cureus.66356
10. Zhang T, Chen L, Kueth G, Shao E. et al. Lactate's impact on immune cells in sepsis: unraveling the complex interplay. Front Immunol. 2024Sep20;15:1483400
11. Topjian AA, Clark AE, Casper TC, Berger JT. et al. Pediatric Emergency Care Applied Research Network. Early lactate elevations following resuscitation from pediatric cardiac arrest are associated with increased mortality. Pediatr Crit Care Med. 2013Oct;14(8):e380-7
12. Yuzefpolskaya M, Schwartz S, Ladanyi A, Abraham J. et al. The Role of Lactate Metabolism in Heart Failure and Cardiogenic Shock: Clinical Insights and Therapeutic Implications. J Card Fail. 2025 Jan 29; S1071-9164(25):00041-7
13. Lazzeri C, Valente S, Chiostri M, Gensini GF. Clinical significance of lactate in acute cardiac patients. World J Cardiol. 2015Aug26;7(8):483-9
14. Wang CH, Huang CH, Chang WT, Tsai MS. et al. Monitoring of serum lactate level during cardiopulmonary resuscitation in adult in-hospital cardiac arrest. Crit Care. 2015Sep21;19(1):344
15. Hsu SH, Kao PH, Lu TC, Wang CH. et al. Serum Lactate for Predicting Cardiac Arrest in the Emergency Department. Journal of Clinical Medicine. 2022;11(2):403
16. Dell'Anna AM, Sandroni C, Lamanna I. et al. Prognostic implications of blood lactate concentrations after cardiac arrest: a retrospective study. Ann. Intensive Care. 2017;7:101
17. Attanà P, Lazzeri C, Picariello C, Dini CS. et al. Lactate and lactate clearance in acute cardiac care patients. Eur Heart J Acute Cardiovasc Care. 2012Jun;1(2):115-21
18. Donnino MW, Miller J, Goyal N, Loomba M, Sankey SS, Dolcourt B. et al. Effective lactate clearance is associated with improved outcome in post-cardiac arrest patients. Resuscitation. 2007;75:229-34
19. Han KS, Kim SJ, Lee EJ, Park KY. et al. Impact of rapid lactate clearance as an indicator of hemodynamic optimization on outcome in out-of-hospital cardiac arrest: A retrospective analysis. PloS one. 2019;14(4):e0214547
20. Chang HCH, Tsai MS, Kuo LK. et al. Factors affecting outcomes in patients with cardiac arrest who receive target temperature management: The multi-center TIMECARD registry. J Formos Med Assoc. 2022;121(1):294-303
21. Wang MT, Tsai MS, Huang CH. et al. Comparison of outcomes between cardiogenic and non-cardiogenic cardiac arrest patients receiving targeted temperature management: The nationwide TIMECARD multicenter registry. J Formos Med Assoc. 2023;122(8):675-689
22. Chi CY, Tsai MS, Kuo LK. et al. Post-resuscitation diastolic blood pressure is a prognostic factor for outcomes of cardiac arrest patients: a multicenter retrospective registry-based analysis. J Intensive Care. 2022;10(1):39
23. Liu J, Chen Y, Yang B, Zhao J. et al. Association between alactic base excess on mortality in sepsis patients: a retrospective observational study. J Intensive Care. 2025Apr11;13(1):20
24. Casserly B, Phillips GS, Schorr C, Dellinger RP. et al. Lactate measurements in sepsis-induced tissue hypoperfusion: results from the Surviving Sepsis Campaign database. Crit Care Med. 2015;43(3):567-73
25. Mikkelsen ME, Miltiades AN, Gaieski DF, Goyal M. et al. Serum lactate is associated with mortality in severe sepsis independent of organ failure and shock. Crit Care Med. 2009;37(5):1670-7
26. Andersen LW, Mackenhauer J, Roberts JC, Berg KM. et al. Etiology and therapeutic approach to elevated lactate levels. Mayo Clin Proc. 2013Oct;88(10):1127-40
27. Shinozaki K, Oda S, Sadahiro T. et al. Blood ammonia and lactate levels on hospital arrival as a predictive biomarker in patients with out-of-hospital cardiac arrest. Resuscitation. 2011;82:404-9
28. Donnino MW, Andersen LW, Giberson T. et al. Initial lactate and lactate change in post-cardiac arrest: a multicenter validation study. Crit Care Med. 2014Aug;42(8):1804-11
29. Howell MD, Donnino M, Clardy P, Talmor D, Shapiro NI. Occult hypoperfusion and mortality in patients with suspected infection. Intensive Care Med. 2007Nov;33(11):1892-1899
30. Cox K, Cocchi MN, Salciccioli JD, Carney E, Howell M, Donnino MW. Prevalence and significance of lactic acidosis in diabetic ketoacidosis. J Crit Care. 2012Apr;27(2):132-7
31. Shapiro NI, Howell MD, Talmor D. et al. Serum lactate as a predictor of mortality in emergency department patients with infection. Annals of emergency medicine. 2005May;45(5):524-528
32. Callaway DW, Shapiro NI, Donnino MW, Baker C, Rosen CL. Serum lactate and base deficit as predictors of mortality in normotensive elderly blunt trauma patients. The Journal of trauma. 2009Apr;66(4):1040-1044
33. Issa MS, Grossestreuer AV, Patel H, Ntshinga L. et al. Lactate and hypotension as predictors of mortality after in-hospital cardiac arrest. Resuscitation. 2021;158:208-214
34. Dadeh AA, Nuanjaroan B. Using initial serum lactate level in the emergency department to predict the sustained return of spontaneous circulation in nontraumatic out-of-hospital cardiac arrest patients. Open Access Emerg Med. 2018Sep26;10:105-111
35. Nishioka N, Kobayashi D, Izawa et al. Association between serum lactate level during cardiopulmonary resuscitation and survival in adult out-of-hospital cardiac arrest: a multicenter cohort study. Scientific Reports. 2021 11
36. Lindholm MG, Hongisto M, Lassus J, Spinar J. et al. Serum Lactate and A Relative Change in Lactate as Predictors of Mortality in Patients With Cardiogenic Shock - Results from the Cardshock Study. Shock. 2020Jan;53(1):43-49
37. Mizutani T, Umemoto N, Taniguchi T, Ishii H. et al. The lactate clearance calculated using serum lactate level 6 h after is an important prognostic predictor after extracorporeal cardiopulmonary resuscitation: a single-center retrospective observational study. J Intensive Care. 2018 Jun 1;6:33. doi: 10.1186/s40560-018-0302-z. Erratum in: J Intensive Care. 2019Mar21;7:18
38. Scolari FL, Schneider D, Fogazzi DV, Gus M. et al. Association between serum lactate levels and mortality in patients with cardiogenic shock receiving mechanical circulatory support: a multicenter retrospective cohort study. BMC Cardiovasc Disord. 2020Nov24;20(1):496
39. Velissaris D, Karamouzos V, Pierrakos C, Koniari I. et al. Use of Sodium Bicarbonate in Cardiac Arrest: Current Guidelines and Literature Review. J Clin Med Res. 2016Apr;8(4):277-83
40. Gissel M, Brummel-Ziedins KE, Butenas S, Pusateri AE, Mann KG, Orfeo T. Effects of an acidic environment on coagulation dynamics. J Thromb Haemost. 2016;14(10):2001-2010
41. He HW, Liu DW, Long Y, & Wang XT. High central venous-to-arterial CO2 difference/arterial-central venous O2 difference ratio is associated with poor lactate clearance in septic patients after resuscitation. Journal of Critical Care. 2016;31(1):76-81
42. Szarpak L, Smereka J, & Ruetzler K. Targeted temperature management: State of the Art. Disaster and Emergency Medicine Journal. 2019;4(2):68-73
Author contact
Corresponding author: Prof. Kor Chew-Teng, Changhua Christian Hospital, No. 135, Nan-Siau Street, Changhua, Taiwan 500; Tel: +886-4-7238595 ext. 7069; Fax: +886-4-7228289; E-mail: 179297org.tw.

 Global reach, higher impact
Global reach, higher impact