Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(5):1822-1844. doi:10.7150/ijms.126174 This issue Cite
Review
STATs Signaling Pathways in Acute Pancreatitis: Mechanisms and Regulation
1. School of Acupuncture and Tuina, Chengdu University of Traditional Chinese Medicine, Chengdu, China.
2. School of Clinical Medicine, Chengdu University of Traditional Chinese Medicine, Chengdu, China.
3. Institute of Hepatopancreatobiliary Surgery, Chongqing General Hospital, Chongqing University, Chongqing 401147, China.
4. International Collaborative Centre on Big Science Plan for Purinergic Signalling, Chengdu University of Traditional Chinese Medicine; School of Health and Rehabilitation, Chengdu University of Traditional Chinese Medicine, Chengdu, China.
5. Department of Emergency Medicine, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, China.
* Yang Peng, An-er Lin and Feng-ning Yang contributed equally to this work as co-first authors.
Received 2025-10-1; Accepted 2026-3-16; Published 2026-3-30
Abstract
Acute pancreatitis (AP) is a potentially life-threatening inflammatory disorder of the pancreas that can progress from local injury to systemic inflammatory response and multi-organ dysfunction. Despite improvements in supportive treatment, there is still no effective drug therapy for AP. This highlights the necessity of clarifying its molecular mechanisms and exploring novel therapeutic strategies. Among the diverse signaling pathways implicated in the pathogenesis of AP, the significance of the signal transducer and activator of transcription (STAT) family has become increasingly evident. Building on the increasing recognition of the STATs family in AP, this review provides a systematic synthesis in three domains. The first addresses mechanistic studies of STATs-related signaling, the second focuses on pharmacological regulation with an emphasis on natural products and chemical drugs, and the third explores clinical evidence connecting STATs activity to severity and outcomes of AP. By synthesizing mechanistic, pharmacological, and clinical evidence, this review highlights the central role of STATs family members in AP. It underscores the therapeutic potential of natural medicines in targeting STATs pathways and suggests directions for future translational studies to develop effective interventions for AP.
Keywords: Acute pancreatitis, The signal transducer and activator of transcription family, Traditional Chinese medicine, Pharmacological interventions
1. Introduction
Acute pancreatitis (AP) is a common gastrointestinal emergency characterized by abrupt pancreatic inflammation and a highly variable clinical course [1]. While most cases are mild and self-limiting, a considerable proportion progress rapidly to severe disease, accompanied by systemic inflammatory response syndrome (SIRS), multiple organ dysfunction syndrome (MODS), and even death. The global incidence of AP is rising, imposing a substantial clinical and socioeconomic burden [2]. In AP, pancreatic acinar cell damage and local inflammatory activation serve as the starting point for systemic amplification, ultimately causing oxidative stress, mitochondrial injury, and disruption of intestinal and pulmonary barriers [3,4]. Clarifying the mechanisms underlying this local-to-systemic progression is key to identifying effective therapeutic strategies.
Amidst the multiple signaling cascades involved in AP, accumulating evidence highlights the signal transducer and activator of transcription (STAT) family as a critical pathway driving disease progression. STATs proteins are cytoplasmic transcription factors that remain inactive under basal conditions and are rapidly activated in response to cytokines and growth factors [5,6]. STATs activation is most commonly mediated via janus kinases (JAKs) in response to cytokines. Additionally, STATs can be activated via interleukin (IL)-6 or suppressor of cytokine signaling (SOCS) pathways. Although nuclear factor kappa-B (NF-κB) does not directly activate STATs, it can promote STATs activation indirectly by inducing upstream cytokines like IL-6 that engage the JAK/STAT axis [7]. Once phosphorylated, STATs proteins form dimers, translocate into the nucleus, and control the expression of genes involved in immunity, inflammation, oxidative stress, and cell survival [8-10]. The mammalian STATs family consists of seven major members with distinct biological functions. STAT1 and STAT2 coordinate antiviral defense and interferon signaling [11,12]. STAT3 plays a key role in inflammation and tissue repair [13]. STAT4, STAT5a and STAT5b regulate T-cell differentiation and hematopoiesis [14,15], while STAT6 contributes to Th2 immune responses and tissue remodeling [16]. Aberrant activation of STATs signaling in AP acts as a central driver of disease progression by linking cytokine stimulation to pathological immune responses and systemic complications. However, the potential roles of other STATs members remain poorly characterized, as most existing studies have focused almost exclusively on STAT3.
Several reviews have addressed JAK/STAT signaling in the context of AP and gastrointestinal diseases [17,18]. Research to date has largely centered on STAT3 as the pivotal mediator of pathological signaling in AP, whereas STAT1 and other STATs family members have been only sporadically addressed, leaving their roles insufficiently defined. To address this gap, the present review provides the first comprehensive synthesis of the STATs family in AP. By providing a comprehensive and forward-looking analysis, this review not only highlights the mechanistic and therapeutic significance of the STATs family in AP but also offers a conceptual framework for the rational development of natural medicine-based and integrative therapeutic approaches (Figure 1).
Mechanism by which natural products alleviate acute pancreatitis.
2. Mechanistic Investigations
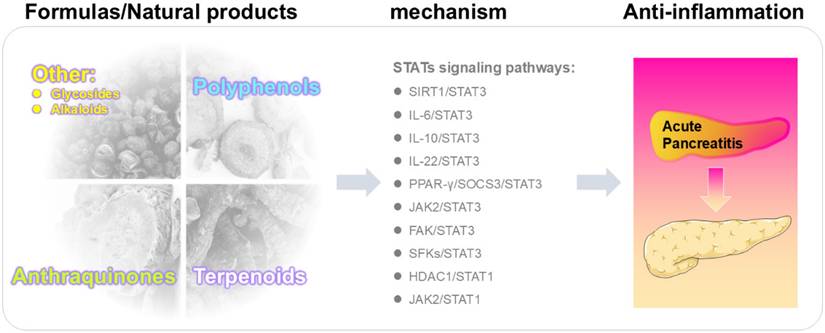
Mechanistic studies highlight the central role of STATs signaling in AP, integrating upstream inflammatory stimuli, downstream effector responses, and multiple pathological processes. A comprehensive overview of these mechanisms is summarized in Figure 2 and Tables 1-2, providing the basis for subsequent discussion.
STATs signaling governing injury-repair balance in AP. In STAT3 axis (left), IL-22, IL-10, and IL-6 positively regulate JAK1 or JAK2 and thereby upregulate STAT3 activity. PPAR-γ functions as a connecting positive modulator across the parallel STAT3 branches. Intracellularly, p-STAT3 (Ser727) and p-STAT3 (Tyr705) were increased. Functionally, STAT3 activation enhances epithelial protection and tissue repair; in parallel, it upregulates chemokine production and inflammatory amplification, which increases macrophage and neutrophil recruitment and may progress to ALI and MODS. In STAT1 axis (right), IFN-γ activates JAK1/STAT1 pathway while SOCS1 negatively regulates this pathway. A second route involves PIAS1, which negatively regulates STAT1; miR-92a-3p downregulates PIAS1, then net-upregulating STAT1. Intracellular readouts show increased acetylation and phosphorylation of STAT1. Downstream, STAT1 activation upregulates NF-κB and COX-2 signaling and induces IL-17 as well as the pro-apoptotic mediators FasL, TRAIL, and Bax, culminating in SIRS and apoptosis. Standard arrows denote positive regulation (upregulation/activation) and T-shaped lines denote negative regulation (downregulation/inhibition). Abbreviations: Ac, acetylation; ALI, acute lung injury; AP, acute pancreatitis; Bax, Bcl-2-associated X protein; COX-2, cyclooxygenase-2; FasL, Fas ligand; IFN-γ, interferon-gamma; IL, Interleukin; JAK, janus kinase; miR, microRNA; MODS, multiple-organ dysfunction syndrome; NF-κB, nuclear factor kappa-B; P, phosphorylation; PIAS1, protein inhibitor of activated STAT1; PPAR-γ, peroxisome proliferator-activated receptor gamma; SIRS, systemic inflammatory response syndrome; SOCS1, suppressor of cytokine signaling 1; STAT, signal transducer and activator of transcription; TRAIL, tumor necrosis factor-related apoptosis-inducing ligand.
Agonist modulating the STATs pathway for AP therapy.
| Compounds | Mechanism | Concentration | Cell/Animal Study | Function | Ref |
|---|---|---|---|---|---|
| CpG 1826 | ↑TLR9, ↑IL-6, ↑p-STAT3 | 4 mg/kg i.p. | CER-AP mice | Regulation of the TLR9/IL-6/STAT3 pathway to modulate immune responses, and alleviate pancreatic and intestinal tissue injury | 59 |
| Garcinone D | ↑p-JAK2, ↑p-STAT3 (Tyr705) | 10 μM | CER-AP AR42J cells | Activating the JAK2/STAT3 pathway to exacerbate pancreatitis | 65 |
| Recombinant IFN-γ | ↑Ac-STAT1 | 10⁴ U per mouse, s.c. | CER-AP mice | Targeting the Ac-STAT1 signaling to alleviate pancreatic injury and modulate immune responses | 21 |
| Recombinant IL-10 | ↑p-STAT3 (Tyr705) | 10 μg/kg i.v. | NaT-SAP rats | Modulation of the IL-10/STAT3 pathway to alleviate pancreatic injury | 80 |
| Recombinant IL-22 | ↑p-STAT3 (Tyr705) | 200 ng per mouse, i.p. or s.c. in vivo 20 ng/mL in vitro | CDE-AP mice NaT-SAP mice ARG-SAP mice CER-AP PACs | Modulating the IL-22/STAT3/Reg3 pathway to regulate immune responses and attenuate pancreatic, pulmonary, and intestinal injury | 37 38 45 78 |
| Recombinant MFG-E8 | ↑p-STAT3 (Ser727), ↑p-STAT3 (Tyr705) | 5, 10, 20 μg/kg i.p. in vivo 20, 100 ng/mL in vitro | ARG-SAP mice, CER-AP mice, CER+LPS-AP mice CER+LPS-AP AR42J cells | Targeting the FAK/STAT3 pathway to alleviate pancreatic injury | 43 |
Abbreviations: Ac, Acetylated; AP, acute pancreatitis; ARG, arginine; CDE, DL-ethionine; CER, caerulein; FAK, focal adhesion kinase; IL, interleukin; i.p., intraperitoneal; i.v., intravenous; LPS, lipopolysaccharide; MFG-E8, milk fat globule epidermal growth factor 8; NaT, sodium taurocholate; PACs, pancreatic acinar cells; p-STAT, phosphorylated signal transduction and transcriptional activation factor; Reg3,Regenerating islet-derived protein 3; SAP, severe acute pancreatitis; s.c., subcutaneously; Ser, serine; STAT, signal transduction and transcriptional activation factor; TLR9, toll-like receptor 9; Tyr, tyrosine.
Antagonist targeting the STATs pathway for AP treatment.
| Compounds | Mechanism | Concentration | Cell/Animal Study | Function | Ref |
|---|---|---|---|---|---|
| AG490 | ↓p-JAK2 (Tyr1007/1008), ↓p-STAT3 (Ser727), ↓p-STAT3 (Tyr705) | 8 mg/kg i.p. or i.v. in vivo 0.02 g/kg i.p. in vivo 50 μM in vitro | NaT-SAP rats CER-AP mice CER-AP AR42J cells | Inhibiting the JAK2/STAT3 and IL-6/STAT3 pathways to modulate immune responses and attenuate pancreatic and hepatic injury | 23 46 20 34 25 |
| A17pro | ↓p-STAT3 (Tyr705) | 1 mg/kg i.p. | CER-AP mice NNK-AP mice | Suppression of the IL-6/STAT3 pathway to alleviate pancreatic injury and modulate immune responses | 53 |
| Anti-viral cocktail | ↓TLR9, ↓IL-6, ↓p-STAT3 | 30 mg/kg ribavirin, 10 mg/kg lamivudine, 20 mg/kg acyclovir i.g. | CER-AP mice | Targeting the TLR9/IL-6/STAT3 pathway to modulate immune responses, and alleviate pancreatic and intestinal tissue injury | 59 |
| APTSTAT3-9R | ↑p-FAK, ↓p-STAT3 (Ser727) | 30 μM | CER+LPS-AP AR42J cells | Modulation of the FAK/STAT3 pathway to alleviate pancreatic injury | 43 |
| Cilengitide | ↓p-FAK, ↓p-STAT3 (Ser727), ↓p-STAT3 (Tyr705) | 20 mg/kg i.p. | ARG-SAP mice | Targeting the FAK/STAT3 pathway to alleviate pancreatic injury | 43 |
| DADS | ↑PPAR-γ, ↑SOCS3, ↓p-STAT3(Tyr705) | 200 μg/kg i.p. | CER-AP mice | Inhibition of the PPAR-γ/SOCS3/STAT3 pathway to attenuate pancreatic and pulmonary tissue injury | 55 |
| Dexamethasone | ↓STAT3 | 10-⁷ M | PACs from NaT-SAP rats | Suppression of the STAT3 signaling to alleviate pancreatic injury | 60 |
| Diphenyleneiodonium | ↓p-JAK2 (Tyr1007/1008), ↓p-STAT3 (Tyr705) | 10 μM | CER-AP AR42J cells | Targeting the JAK2/STAT3 pathway to alleviate pancreatic injury | 25 |
| EX527 | ↓SIRT1, ↑Ac-STAT3 | 20 mg/kg i.p. | CER-AP mice | Modulation of the IL-6/STAT3 pathway and SIRT1/STAT3 pathway to alleviate pancreatic injury and modulate immune responses | 56 |
| Fedratinib | ↓p-JAK2, ↓p-STAT3 (Tyr705) | 3 nM | CER-AP AR42J cells | Inhibiting the JAK2/STAT3 pathway to mitigate pancreatitis | 65 |
| HJC0152 | ↓STAT1, ↓p-STAT1 (Tyr701), ↓p-STAT3 (Tyr705) | 10 μM | CVB3-AP 266-6 cells | Suppression of STAT3 signaling to enhance STAT1 activation, mitigating pancreatic and cardiac damage | 44 |
| N-acetylcysteine | ↓STAT3 | 50 mg/kg i.p. | BPDO-AP rats NaT-SAP rats | Targeting the STAT3 signaling to alleviate pancreatic injury | 57 |
| ODN2088 | ↓TLR9, ↓p-STAT3 | 4 mg/kg i.p. | CER-AP mice | Inhibition of the TLR9/IL-6/STAT3 pathway to modulate immune responses, and alleviate pancreatic and intestinal tissue injury | 59 |
| PF-00562271 | ↓p-FAK, ↓p-STAT3 (Ser727) | 1 μM | CER+LPS-AP AR42J cells | Suppression of the FAK/STAT3 pathway to alleviate pancreatic injury | 43 |
| PP2 | ↓p-Src, ↓p-ERK, ↓p-JNK, ↓p-STAT3 (Tyr705) | 0.5, 1, 1.5 mg/kg i.p. in vivo 1, 10 μM in vitro | substance P-AP PACs CER- AP mice | Targeting the SFKs/STAT3 pathway to alleviate pancreatic injury | 29 |
| Peptide YY | ↓STAT1, ↓STAT3 | 500 pM | TNF-α-AP AR42J cells | Modulation of the STAT1 and STAT3 signaling to alleviate pancreatic injury | 39 |
| (R)-TML104 | ↑SIRT1, ↓Ac-STAT3, ↓p-STAT3 (Tyr705), ↓IL-6 | 5, 10, 20 mg/kg i.p. | CER-AP mice | Targeting the IL-6/STAT3 pathway and SIRT1/STAT3 pathway to alleviate pancreatic injury and modulate immune responses | 56 |
| Ruxolitinib | ↓p-JAK2, ↓p-STAT3 | 180 mg/kg i.g. | NaT-SAP rats | Targeting the JAK2/STAT3 pathway to alleviate pancreatic and intestinal tissue injury | 82 |
| S3I-201 | ↓p-STAT3 (Tyr705) | 5 mg/kg i.p. | NaT-SAP rats | Targeting the IL-10/STAT3 pathway to alleviate pancreatic injury | 80 |
| SEW2871 | 1. ↓p-STAT3 | 20 mg/kg i.g. | CER-AP mice | Inhibition of the STAT3 signaling to alleviate pancreatic injury and modulate immune responses | 61 |
| sgp130Fc | ↓p-STAT3 (Tyr705), ↓p-STAT3 (Ser727) | 0.3 mg/mouse i.p. | CER-AP mice | Suppression of the IL-6/STAT3 pathway to attenuate pancreatic and pulmonary tissue injury | 31 |
| Stattic | ↓p-STAT1 (Tyr701), ↓p-STAT3 (Tyr705), ↓p-JAK2 | 3.75 mg/kg i.p. in vivo 2.5, 5 μM in vitro | NaT-SAP rats CVB3-AP 266-6 cells CER-AP AR42J cells | Inhibiting the JAK2/STAT3 pathway to alleviate pancreatic, intestinal and myocardial injury | 44 82 65 |
Abbreviations: Ac, acetylated; AP, acute pancreatitis; A17pro, ADAM17 Prodomain Inhibitor; ARG, arginine; BPDO, bile-pancreatic duct obstruction; CER, caerulein; CP96345, (2S,3S)-cis-2-(diphenylmethyl)-N-((2-methoxyphenyl)-methyl)-1-azabicyclo(2.2.2.)-octan-3-amine; FAK, focal adhesion kinase; IL, interleukin; i.g., intragastric; i.p., intraperitoneal; i.v., intravenous; JAK, janus kinase; LPS, lipopolysaccharide; MFG-E8, milk fat globule epidermal growth factor 8; NaT, sodium taurocholate; NNK, nicotine-derived nitrosamine ketone; PACs, pancreatic acinar cells; PP2, 4-amino-5-(4-chlorophenyl)-7-(t-butyl) pyrazolo [3,4-D] pyrimidine; PPAR-γ, peroxisome proliferator-activated receptor gamma; p-STAT, phosphorylated signal transduction and transcriptional activation factor; (R)-TML104, (R)-4,6-dimethoxy-3-(4-methoxy phenyl)-2,3-dihydro-1H-indanone; SAP, severe acute pancreatitis; Ser, serine; SFK, Src family kinase; SIRT1, sirtuin 1; SOCS3, suppressor of cytokine signaling 3; STAT, signal transduction and transcriptional activation factor; TLR9, toll-like receptor 9; TNF-α, tumor necrosis factor alpha; Tyr, tyrosine.
2.1 Activation of the STATs pathway: upstream signals and functional duality
2.1.1. STAT1
Studies indicate that JAK1 acts as a critical upstream regulator of STAT1 signaling during the progression of AP. In cerulein (CER)-stimulated AR42J cells, the total and phosphorylated levels of JAK1, STAT1, and STAT3 increase synchronously with TNF-α, IL-1β, and IL-6 [19]. AG490 (tyrphostin AG490; tyrphostin B42) is a tyrosine kinase inhibitor that targets EGFR, STAT3, and JAK2/3. Treatment with AG490 significantly suppresses these inflammatory responses, suggesting therapeutic potential during the hyperacute phase [19]. Furthermore, the JAK2/STAT1 pathway contributes to disease progression, as demonstrated by the effective inhibition of its activation through AG490 or FKN-siRNA [20]. Notably, despite being a canonical STAT1 activator, interferon gamma (IFN-γ) may exert context-dependent protective effects in CER-induced AP by promoting STAT1 acetylation, thereby inhibiting NF-κB nuclear translocation and downstream pro-inflammatory signaling [21]. Additionally, the miR-155/SOCS1/STAT1 axis is involved in immune dysregulation, where miR-155 inhibition restores SOCS1-mediated negative feedback on STAT1 signaling and reduces IL-17-driven injury [22].
2.1.2. STAT3
JAK2-mediated STAT3 phosphorylation is essential for local and systemic inflammation. The activation of the JAK2/STAT3 pathway promotes macrophage polarization, whereas pharmacological inhibition using AG490 effectively reduces the levels of p-JAK2 and p-STAT3 at the Ser727 residue [23]. FXYD domain-containing protein 5 (FXYD5) and NADPH oxidase act as crucial upstream triggers, linking transmembrane signaling and oxidative stress to the JAK2/STAT3 pathway [24,25]. Beyond these molecular triggers, activation often varies by etiology. Bile acids in biliary AP models induce rapid JAK2/STAT3 activation linked to severe oxidative stress [26], whereas in alcoholic AP, ethanol metabolites and reactive oxygen species may activate STAT3 as a protective response to resolve cellular redox imbalance [27]. Beyond JAK2-dependent mechanisms, peroxisome proliferator-activated receptor gamma (PPAR-γ) inhibits STAT3 through direct interaction [28], while the neurokinin 1 receptor and Src family kinases upregulate its expression [29]. Interestingly, in biliary models, oxidized phospholipids can exert dual effects by triggering STAT3-mediated inflammation at low concentrations or recruiting PPAR-γ to inhibit such signaling at higher levels [30].
IL-6 remains a primary upstream activator that recruits JAK2 via the gp130 receptor complex to upregulate p-STAT3 at the Tyr705 site [31]. The intensity of this signal is significantly altered by metabolic factors such as obesity, which leads to prolonged elevations of IL-6 and sustained STAT3 phosphorylation that differs from the transient activation seen in lean models [32]. Within the first three hours after AP induction, IL-6 expression is typically upregulated, and its neutralization has been shown to reduce p-STAT3 levels while alleviating associated lung injury [33]. The STAT3 signalosome is further balanced by non-coding RNAs and counter-regulatory cytokines. While miR-148a and miR-551b-5p provide opposing regulatory frameworks [34,35], IL-10 and IL-22 further define the functional duality of STAT3. Specifically, IL-10-driven STAT3 activation can, under certain viral contexts, paradoxically amplify inflammation [36], whereas IL-22-mediated signaling contributes to barrier repair and cellular protection [37]. Notably, the protective IL-22/STAT3 response in the colon tends to decline more rapidly than pro-inflammatory markers, which potentially increases the risk of intestinal barrier failure as AP progresses [38].
Collectively, the net biological output of STAT3 in AP reflects a stimulus and cell type specific integration of competing upstream signals. The IL-6/STAT3 axis primarily acts as a pro-inflammatory driver in acinar cells and recruited neutrophils, where it contributes to the escalation of local injury into a systemic cytokine storm [31,33]. In contrast, IL-22-mediated STAT3 signaling functions as a protective pathway in acinar and intestinal epithelial cells, playing a role in maintaining barrier integrity and promoting tissue repair [37,38]. Furthermore, while IL-10 is traditionally recognized as a potent anti-inflammatory cytokine, its role via STAT3 can be paradoxically pro-inflammatory depending on the pathological context, such as in certain viral models [36]. These multifaceted roles identify STAT3 as a key regulator that balances detrimental inflammatory responses with beneficial tissue repair based on the molecular stimulus and cellular environment.
2.1.3. Dynamic activation patterns of STATs across disease stages
STAT activation evolves dynamically. In the hyperacute phase, TNF-α enhances the nuclear binding of STAT1 and STAT3 within 30 minutes [39]. Simultaneously, hydrostatic pressure can directly enhance NF-κB p65 and STAT3 signaling, thereby inducing inflammation, which suggests a critical regulatory role of STAT3 in sensing mechanical stress within a 24 hour experimental timeframe [40]. Subsequently, in pressure-induced or biliary pancreatitis, IL-6 trans-signaling drives pathogenic responses via dual STAT3 phosphorylation (Tyr705/Ser727) [31,41]. While Tyr705 phosphorylation is essential for STAT3 dimerization, the phosphorylation at Ser727 is particularly critical for enhancing transcriptional efficacy and maintaining mitochondrial bioenergetics, which may help preserve acinar cell integrity during early injury [42,43]. This dual activation also induces C-X-C motif chemokine ligand 1 and promotes neutrophil recruitment [31]. In viral AP, STAT3 and STAT1 exhibit an antagonistic relationship where excessive STAT3 activation might impair viral clearance [44]. Temporally, while moderate STAT3 activation promotes autophagic flux and alleviate edema, sustained activation drives chronic fibrosis and multi-organ injury [45].
2.2 Downstream effectors of STATs signaling
STAT3 executes a broad transcriptional program constrained by intrinsic feedback. Inhibition of the JAK2/STAT3 pathway typically downregulates pro-inflammatory cytokines, including IL-6, IL-1β, and TNF-α, mitigating SAP severity and systemic injury [46,47]. The IL-6/STAT3 signaling axis serves as a critical upstream pathway initiating these downstream inflammatory responses, directly promoting the expression of pro-inflammatory cytokines such as TNF-α [48,49]. Simultaneously, this axis forms a STAT3/SOCS3 negative feedback module [50]. The downstream consequences of STAT3 activation are notably stage-dependent. In the acute phase, the neutralization of IL-6 may increase cellular apoptosis to mitigate SAP severity [33]. However, as the disease enters the resolution phase, p-STAT3 activation appears to trigger the acinar-to-ductal metaplasia (ADM) process, which is increasingly recognized as a self-protective mechanism that reduces acinar necrosis and initiates tissue remodeling [51]. Mechanistically, STAT3 is also considered indispensable for the production of pancreatitis-associated protein 1 (PAP1), which is a key protein that suppresses necrosis and facilitates pancreatic repair [52].
Furthermore, the STAT3-dependent chemokine network links acinar signaling to immune cell recruitment, and its persistent activation in the chronic phase correlates with tissue fibrosis [53]. In obesity-related contexts, this sustained activation may even favor a protumorigenic microenvironment through the induction of matrix metalloproteinase-7 [32]. To maintain homeostasis, SOCS3 provides self-limiting negative feedback to control inflammation. Specifically, PAP/hepatocarcinoma-intestine-pancreas (HIP) proteins induce STAT3-mediated SOCS3 transcription, which subsequently inhibits JAK phosphorylation and NF-κB activation [54,55]. Similarly, sirtuin 1 (SIRT1)-mediated STAT3 deacetylation reduces its transcriptional activity and decreases inflammatory cytokine expression, indicating that SIRT1 helps mitigate AP by inactivating STAT3 [56].
2.3 Pathological mechanisms mediated by STATs signaling
During the progression of AP, early local inflammation can rapidly escalate into a SIRS, which is frequently characterized by a cytokine storm. STAT family members, particularly STAT1 and STAT3, are involved in orchestrating these inflammatory circuits by regulating various pro-inflammatory cytokines, chemokines, and immune cell functions. STAT3 appears to promote inflammation via the activation of the IL-6/STAT3 pathway [33] or the IL-10/STAT3 axis [36], which is particularly relevant in viral AP. This process includes the induction of chemokines [56,57] and a potential synergistic interaction with NF-κB [58], which collectively enhance neutrophil and macrophage infiltration [59] and exacerbate pancreatic injury [60,61]. STAT1 similarly amplifies inflammation through transcriptional coordination with NF-κB [62,63]. However, in addition to its pro-inflammatory role, STAT3 exerts protective effects in AP through multiple mechanisms, including the induction of anti-inflammatory PAP1 [52], and mediation of protection related to T-cell protein tyrosine phosphatase deficiency [64].
Beyond inflammation, STATs signaling drives parenchymal damage. In acinar cells, TNF-α-induced STAT1/STAT3 activation leads to premature digestive enzyme secretion such as amylase and trypsinogen [39]. The IL-6/JAK2/STAT3 axis is suggested to be a relevant node for acinar injury, as the inhibition of this pathway has been shown to reduce serum levels of amylase and lipase while alleviating tissue edema [26,65]. STAT3 also mediates lipotoxic signals and may promote ferroptosis via the CLDN4/JAK2/STAT3 axis [66,67]. Furthermore, the temporal control of STAT3 signaling is likely important for tissue homeostasis because uncontrolled signaling, which can result from IL-22BP deficiency, is associated with persistent ADM and delayed tissue recovery [45]. Conversely, site-specific phosphorylation (Tyr705/Ser727) via the FAK/STAT3 axis preserves mitochondrial fitness and limit apoptotic demise [42,43]. This parenchymal damage extends to endocrine islets, where the IFN-γ/STAT1 axis drives β-cell dysfunction and reduced insulin biosynthesis [68,69]. Although basal STAT3 preserves β-cell integrity [70], inflammatory STAT3 hyperactivation impairs insulin secretion [71,72].
As the disease progresses toward its severe form, STAT signaling facilitates the dissemination of pathological signals to extra-pancreatic organs, leading to systemic complications and chronic sequelae. During the transition to severe AP, extra-pancreatic organs are often among the earliest affected [73,74]. The IL-22/STAT3 axis potentially facilitates the shift from local injury to systemic complications by regulating epithelial autophagy and immune responses [45]. The IL-6/STAT3 pathway is closely linked to multi-organ fibrosis, potentially through fibroblast activation that drives structural remodeling of the kidney, lung, and vasculature [75-77]. Abnormal JAK/STAT3 activation via ADAM17-mediated IL-6 trans-signaling facilitates the spread of pathological signals beyond the pancreas [75,77]. Recombinant IL-22 and runt-associated transcription factor 3 have been suggested to mitigate pulmonary injury through mechanisms involving STAT3 signaling [47,78]. Similarly, the IL-22/STAT3 and IL-10/STAT3 axes are thought to contribute to the preservation of the intestinal barrier and blood-brain barrier integrity [38,79,80]. Furthermore, in the context of obesity-related AP, IL-6 may prolong the inflammatory response and potentially promote a tumorigenic microenvironment through sustained STAT3 activation [32]. Essentially, this indicates that when STAT3 remains active, the initial acute damage transitions into a long-term problem that reshapes the pancreatic tissue, making the local environment more vulnerable to cancer development. However, despite the extensive documentation of the STAT1/3 axes, the roles of other family members in pancreatic pathology are often overlooked. For instance, the involvement of lesser-studied members such as STAT2 and STAT4 in coordinating the Th1/Th17 response during the acute-to-chronic transition remains a critical knowledge gap. Moreover, while acinar cell injury is well-characterized, how STAT signaling, particularly underdeveloped members such as STAT5, regulates ductal bicarbonate secretion to mitigate protease-driven injury is yet to be elucidated. Bridging these gaps in isoform-specific signaling across different cell types is essential for a holistic understanding of STAT-mediated regulation in AP.
3. Pharmacological studies
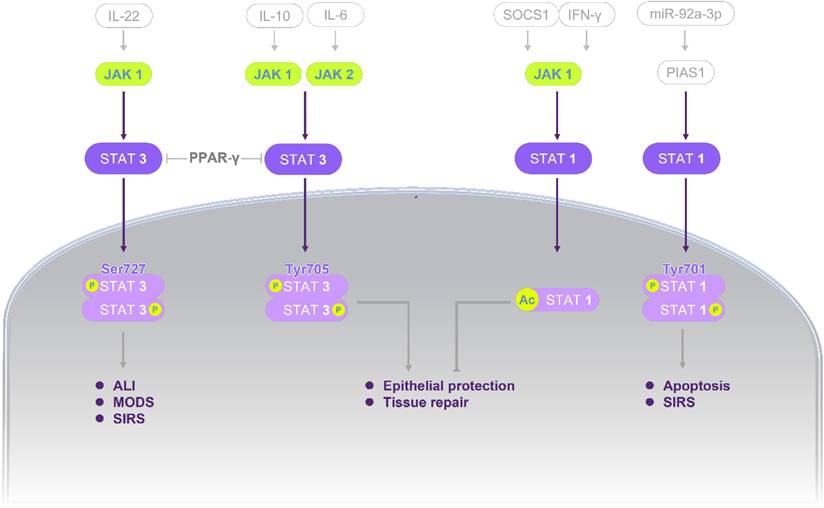
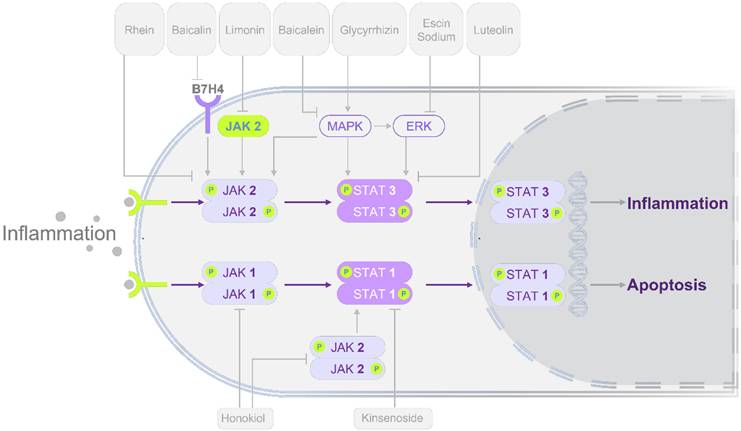
Building on mechanistic insights, pharmacological studies have demonstrated that modulation of STATs signaling can alleviate pancreatic injury and systemic complications. These interventions span multiple categories, including multi-component herbal formulas, bioactive natural compounds, and chemical agents, each with distinct regulatory effects on STATs activity. An overview of these pharmacological findings is presented in Figure 3 and Tables 3-4, which highlight both shared and unique therapeutic mechanisms.
Modulation of STATs signaling pathways by herbal-derived single compounds in alleviating AP. Most compounds exert protective effects primarily through inhibition of the STAT3 pathway, while a subset acts via STAT1 modulation. In terms of STAT3 regulation, the majority of monomers act through the JAK2/STAT3 or MAPK/ERK/STAT3 pathways. Rhein reduces pancreatic injury by directly inhibiting phosphorylation of molecules in the JAK2/STAT3 pathway. Baicalein and baicalin act by suppressing upstream regulators B7H4 and MAPK, thereby modulating the JAK2/STAT3 cascade. In contrast, glycyrrhizin alleviates AP by inhibiting phosphorylation along the MAPK/ERK/STAT3/AKT axis. Similarly, escin sodium exerts protective effects by suppressing ERK/STAT3 pathway. Direct STAT3-targeting compounds include luteolin, daphnetin, and colchicine. At the STAT1 level, honokiol reduces AP by inhibiting JAK/STAT1 phosphorylation, while kinsenoside protects against AP by binding TLR4 and downregulating p-STAT1. These regulatory mechanisms provide potential therapeutic targets for AP. Standard arrows denote positive regulation (upregulation/activation) and T-shaped lines denote negative regulation (downregulation/inhibition). Abbreviations: AKT, protein kinase B; AP, acute pancreatitis; B7H4, B7 homolog 4; ERK, extracellular signal-regulated kinase; JAK, janus kinase; MAPK, mitogen-activated protein kinase; PPAR-γ, peroxisome proliferator-activated receptor gamma; Ser, serine; SOCS3, suppressor of cytokine signaling 3; STAT, signal transducer and activator of transcription; TLR4, toll-like receptor 4.
Herbal formulas modulating the STATs pathway for AP therapy.
| Formulas | Herbs | Preparation Method | Standardization | Cell/Animal Study | Pathways | Mechanism | Ref |
|---|---|---|---|---|---|---|---|
| Da Cheng Qi Decoction | Rheum palmatum L. (Da Huang), Magnolia officinalis Rehd. et Wils. (Hou Po), Citrus aurantium L. (Zhi Shi), and Natrii Sulfas (Mang Xiao) | Water decoction with ethanol precipitation Reconstituted spray-dried powders | HPLC fingerprinting Chinese Pharmacopoeia standards | CER-AP AR42J cells NaT-AP rats | JAK2/STAT3 signaling pathway | ↓p-JAK2, ↓p-STAT3 (Tyr705) | 65 82 |
| Chaiqin Chengqi Decoction | Bupleurum chinense DC. (Chai Hu), Scutellaria baicalensis Georgi (Huang Qin), Magnolia officinalis Rehd. et Wils. (Hou Po), Citrus aurantium L. (Zhi Shi), Gardenia jasminoides Ellis (Zhi Zi), Rheum palmatum L. (Da Huang), Natrii Sulfas (Mang Xiao), Corydalis yanhusuo W.T. Wang (Yan Hu Suo), Ligusticum chuanxiong Hort. (Chuan Xiong), and Aucklandia lappa Decne. (Mu Xiang) | Water decoction | Hospital-authenticated raw materials | CER-AP AR42J cells CER-AP rats | JAK2/STAT3 signaling pathway | ↓p-JAK2, ↓p-STAT3 | 83 |
| Qingyi Granules | Scutellaria baicalensis Georgi (Huang Qin), Gardenia jasminoides Ellis (Zhi Zi), Corydalis yanhusuo W.T. Wang (Yan Hu Suo), Bupleurum chinense DC. (Chai Hu), Rheum palmatum L. (Da Huang), Paeonia lactiflora Pall. (Bai Shao), Natrii Sulfas (Mang Xiao), and Aucklandia lappa Decne. (Mu Xiang) | Standardized granules; reconstituted for intragastric administration | UHPLC-QE-MS serum pharmacochemistry | NaT-AP rats | IL-6/STAT3 signaling pathway | ↓IL-6, ↓p-STAT3 | 84 |
| Chaihuang Qingyi Huoxue Granule | Bupleurum chinense DC. (Chai Hu), Magnolia officinalis Rehd. et Wils. (Hou Po), Paeonia lactiflora Pall. (Chi Shao), Rheum palmatum L. (Da Huang), Prunus persica (L.) Batsch (Tao Ren), Salvia miltiorrhiza Bge. (Dan Shen), Glycyrrhiza uralensis Fisch. (Gan Cao), Corydalis yanhusuo W.T. Wang (Yan Hu Suo), Astragalus membranaceus (Fisch.) Bge. (Huang Qi), Scutellaria baicalensis Georgi (Huang Qin), Citrus aurantium L. (Zhi Shi), Gardenia jasminoides Ellis (Zhi Zi), Paeonia lactiflora Pall. (Bai Shao), Taraxacum mongolicum Hand.-Mazz. (Pu Gong Ying) | Commercial granules; saline decoction | Standardized commercial formulation | NaT-AP rats | PI3K/AKT signaling pathway | ↓p-PI3K, ↓p-AKT | 85 |
| Qingyi Decoction | Rheum palmatum L. (Da Huang), Bupleurum chinense DC. (Chai Hu), Aucklandia lappa Decne. (Mu Xiang), Corydalis yanhusuo W.T. Wang (Yan Hu Suo), Paeonia lactiflora Pall. (Bai Shao), Scutellaria baicalensis Georgi (Huang Qin), Coptis chinensis Franch. (Huang Lian) | Water decoction; lyophilized powder | UHPLC-Orbitrap-MS/MS profiling | CER-AP mice | NF-κB/NLRP3 signaling pathway | ↓NF-κB, ↓NLRP3 | 86 |
| Da-Huang-Fu-Zi-Tang | Rheum palmatum L. (Da Huang), Aconitum carmichaelii Debx. (Fu Zi), Asarum heterotropoides F. Schmidt (Xi Xin) | Water decoction; vacuum concentration | HPLC-ESI-MS/UV profiling | NaT-AP rats | JAK2/STAT3 signaling pathway | ↓p-JAK2, ↓p-STAT3 | 87 |
| Shenmai Injection | Panax ginseng C.A. Mey. (Ren Shen) and Ophiopogon japonicus (L.f.) Ker Gawl. (Mai Dong) | Commercial patent drug | UHPLC-QTOF/MS analysis | NaT-AP 266-6 cells NaT-AP mice | IL-6/STAT3 signaling pathway | ↓IL-6, ↓p-STAT3 (Tyr705) | 89 |
| Rheum palmatum L. and Salvia miltiorrhiza Bge. | Rheum palmatum L. (Da Huang) and Salvia miltiorrhiza Bge. (Dan Shen) | DH-DS herbal pair (1:1 ratio); oral gavage (crude drug equivalent) | Batch-controlled; authenticated by specialist | CER-AP rats | JAK2/STAT3/IL-17A signaling pathway | ↓JAK2, ↓p-JAK2 (Tyr1007/1008), ↓STAT3, ↓p-STAT3 (Tyr705), ↓IL-17A | 90 |
Note: All Latin binomials are derived from http://herb.ac.cn, with the exception of Rheum palmatum L. and Salvia miltiorrhiza Bge.82. Abbreviations: AKT, protein kinase B; AP, acute pancreatitis; CER, cerulein; DH-DS, Da Huang and Dan Shen; HPLC, high-performance liquid chromatography; HPLC-ESI-MS/UV, high-performance liquid chromatography-electrospray ionization-mass spectrometry/ultraviolet detector; IL, interleukin; JAK2, janus kinase 2; NaT, sodium taurocholate; NF-κB, nuclear factor kappa-B; NLRP3, NOD-, LRR- and pyrin domain-containing protein 3; PI3K, phosphoinositide 3-kinase; p-STAT, phosphorylated signal transduction and transcriptional activation factor; STAT, signal transducer and activator of transcription; Tyr, tyrosine; UHPLC-Orbitrap-MS/MS, ultra-high-performance liquid chromatography-Orbitrap tandem mass spectrometry; UHPLC-QE-MS, ultra-high-performance liquid chromatography-Q Exactive mass spectrometry; UHPLC-QTOF/MS, ultra-high-performance liquid chromatography-quadrupole time-of-flight mass spectrometry.
Herbal-derived single compounds targeting the STATs pathway for AP treatment.
| Compounds | Classification | Source | Preparation Method | Standardization | Cell/Animal Study | Mechanism | Function | Ref |
|---|---|---|---|---|---|---|---|---|
| Baicalein | Polyphenols | Scutellaria baicalensis Georgi (Huang Qin) | Normal saline solution (20 mg/kg, i.p.) | Purity > 98% (endotoxin-free); commercial source | CER-AP mice | ↓p-p38, ↓p-ERK, ↓p-JNK, ↓p-JAK2, ↓p-STAT3 | Inhibition of the MAPK/JAK2/STAT3 pathway alleviates pancreatic damage and reshapes the immune microenvironment | 91 |
| Baicalin | Polyphenols | Scutellaria baicalensis Georgi (Huang Qin) | Intravenous solution (20 mg/kg, i.v.) | Commercial source | ARG-AP HTG mice | ↓B7H4, ↓p-JAK2, ↓p-JAK2 (Tyr1007/1008), ↓p-STAT3 (Tyr705) | Inhibition of the B7H4/JAK2/STAT3 axis mitigates oxidative stress and protects against pancreatic damage | 92 |
| Luteolin | Polyphenols | Chrysanthemum indicum L. (Ye Ju Hua) | DMSO-saline solution (2 mg/kg, i.p.) | Commercial source | CER-AP mice | ↓p-STAT3 (Tyr705) | Targeting the STAT3 pathway to reduce pancreatic injury and inhibit inflammation-driven tumorigenesis | 93 |
| Daphnetin | Polyphenols | Daphne odora Thunb. (Rui Xiang) | 5% DMSO solution (4 mg/kg, i.p.) | Purity 99.23%; commercial source | ARG-AP mice | ↓p-JAK2, ↓p-JAK2 (Tyr1007/1008), ↓ p-STAT3 (Tyr705) | Inhibiting the JAK2/STAT3 pathway alleviates pancreatic and pulmonary injury while modulating immune responses | 94 |
| Honokiol | Polyphenols | Magnolia officinalis Rehd. et Wils. (Hou Po) | Saline solution (5 mg/kg, i.p.) | Commercial source | CER+LPS-AP mice | ↓p-JAK1 (Tyr1034/1035), ↓p-JAK2 (Tyr1007/1008), ↓p-STAT1 (Ser727) | Targeting the JAK1/2-STAT1 pathway to alleviate pancreatic and intestinal tissue injury | 95 |
| Butyrate | Short-chain fatty acid | - | PBS solution (i.g. for 7 days) | Commercial source; GC-MS quantified | CER-AP mice | ↓HDAC1, ↓p-STAT1 (Ser727), ↓NLRP3 | Modulation of the HDAC1/STAT1/NLRP3 pathway to alleviate pancreatic and intestinal tissue injury | 96 |
| Curcumin | Polyphenols | Curcuma longa L. (Jiang Huang) | Saline solution (100 mg/kg, i.p.) | Commercial source | NaT-AP rats | ↓p-JAK2, ↓p-STAT3 (Tyr705) | Inhibiting the JAK2/STAT3 pathway to alleviate pancreatic and renal tissue injury | 97 |
| DSC | Polyphenols | Salvia miltiorrhiza Bge. (Dan Shen) | Corn oil with <1% DMSO (25-100 mg/kg, i.p.) | Lab-synthesized; HPLC, 1H NMR, and ESI-HRMS identified | CER-AP 266-6 cells CER-AP mice | ↓p-STAT3 (Tyr705), ↓NLRP3 | Suppressing the STAT3/NLRP3 pathway to alleviate pancreatic injury and modulate immune responses | 98 |
| (R)-TML104 | Polyphenols | Veratrum album L. (Bi Li Lu) | Saline solution (5, 10, 20 mg/kg, i.p.) | Lab-synthesized | CER-AP mice | ↑SIRT1, ↓Ac-STAT3, ↓IL-6, ↓p-STAT3 (Tyr705) | Supressing the IL-6/STAT3 pathway to alleviate pancreatic injury | 56 |
| Selenium | Essential trace element | - | Saline solution (0.25-1 mg/kg, i.p.) | Commercial source | CER+LPS-AP mice | ↓p-ERK, ↓p-JNK, ↓p-p38, ↓p-NF-κB, ↓p-STAT3 | Targeting the MAPK/NF-κB/STAT3 pathway to attenuate pancreatic and pulmonary tissue injury | 99 |
| Picroside II | Terpenoids | Neopicrorhiza scrophulariiflora (Pennell) Hong (Hu Huang Lian) | Saline solution (25 mg/kg, i.p.) | Purity > 98%; commercial source | NaT-AP rats | ↓p-JAK2, ↓p-STAT3 | Inhibition of the JAK2/STAT3 pathway to alleviate pancreatic and hepatic tissue injury | 100 |
| Limonin | Terpenoids | Citrus limon (L.) Burm. f. (Ning Meng) | Tail vein injection (25, 50, 100 mg/kg, i.v.) | Commercial source | CER-AP mice ARG-AP mice | ↓JAK2, ↓p-STAT3 (Tyr705) | Regulation of the JAK2/STAT3 pathway to alleviate pancreatic injury in both MAP and SAP model | 101 |
| Lutein | Terpenoids | Green leafy vegetables, fruits, and egg yolk | DMSO solution (1-5 μM) | Commercial source | CER-AP AR42J cells | ↑PPAR-γ, ↑SOCS3, ↓p-JAK2 (Tyr1007/1008), ↓p-STAT3 (Tyr705) | Targeting the PPAR-γ/SOCS3/JAK2/STAT3 pathway to alleviate pancreatic injury | 102 |
| Escin Sodium | Terpenoids | Aesculus chinensis Bunge (Qi Ye Shu) | Saline solution (1-6 mg/kg, i.p.) | Commercial source | NaT-AP AR42J cells NaT-AP rats | ↓p-ERK, ↓p-STAT3 (Tyr705) | Suppression of the ERK/STAT3 pathway to alleviate pancreatic injury | 103 |
| Glycyrrhizin | Terpenoids | Glycyrrhiza uralensis Fisch. (Gan Cao) | - | Commercial source | NaT-AP PACs NaT-AP mice | ↓p-ERK1/2, ↓p-STAT3, ↓p-AKT | Modulation of the MAPK/ERK/STAT3/AKT pathway to mitigate pancreatic injury | 104 |
| Rhein | Anthraquinones | Rheum palmatum L. (Da Huang) | Saline solution (30 mg/kg, i.g.) | Purity > 98%; commercial source | CER-AP AR42J cells NaT-AP rats | ↓p-JAK2 (Tyr1007/1008), ↓JAK2; ↓p-STAT3 (Tyr705), ↓STAT3 | Inhibition of the JAK2/STAT3 pathway attenuates pancreatic damage | 81 |
| Colchicine | Alkaloids | Colchicum autumnale L. (Qiu Shui Xian) | Saline solution (0.5 mg/kg, i.g.) | Commercial source | NaT-AP rats | ↓p-STAT3 (Tyr705) | Suppression of STAT3 signaling alleviates pancreatic and pulmonary tissue injury | 107 |
| Rutaecarpine | Alkaloids | Euodia rutaecarpa (Juss.) Benth. (Wu Zhu Yu) | DMSO solution (25, 50, 100 mg/kg, i.g.) | Purity > 98%; commercial source | CER+LPS-AP mice CER-AP AR42J cells | ↓p-p38, ↓p-ERK, ↓p-JNK, ↓p-STAT3 (Ser727) | Modulation of the MAPK/STAT3 pathway to mitigate pancreatic injury | 108 |
| Kinsenoside | Glycosides | Anoectochilus roxburghii (Wall.) Lindl. (Jin Xian Lian) | Saline solution (2.5, 5, 10 mg/kg, i.p.) | Purity 99.91%; Commercial source | CER+LPS-AP mice NaT-AP mice | ↓p-STAT1 (Tyr701) | Targeting the STAT1 pathway alleviates pancreatic injury while modulating immune responses | 109 |
| Lactate | Metabolic byproduct | Endogenous metabolite | PBS solution (150 mM, i.p.) | Commercial source | CER-AP PACs CER-AP mice | ↓p-JAK2 (Tyr1007/1008), ↓p-STAT1 (Tyr701) | Inhibiting the JAK2/STAT1 pathway alleviates pancreatic injury while modulating immune responses | 110 |
Note: All Latin names are from http://herb.ac.cn, except those marked with an asterisk (*), which follow the 2020 Pharmacopoeia of the People's Republic of China. Abbreviations: Ac, acetylated; AKT, protein kinase B; AP, acute pancreatitis; ARG, arginine; B7H4, B7 homolog 4; CER, cerulein; DSC, 4-(2-acetoxy-3-((R)-3-(benzylthio)-1-methoxy-1-oxopropan-2-ylamino)-3-oxopropyl)-1,2-phenylene diacetate; EGFR, epidermal growth factor receptor; ERK, extracellular signal-regulated kinase; HDAC1, histone deacetylase 1; HTG, hypertriglyceridemia; i.g., intragastric; i.p., intraperitoneal; i.v., intravenous; JAK, janus kinase; JNK, c-Jun N-terminal kinase; LPS, lipopolysaccharide; MAP, mild acute pancreatitis; MAPK, mitogen-activated protein kinase; NaT, sodium taurocholate; NF-κB, nuclear factor kappa-B; NLRP3, NOD-, LRR- and Pyrin domain-containing protein 3; PACs, pancreatic acinar cells; PPAR-γ, peroxisome proliferator-activated receptor gamma; p-STAT, phosphorylated signal transduction and transcriptional activation factor; (R)-TML104, (R)-4,6-dimethoxy-3-(4-methoxy phenyl)-2,3-dihydro-1H-indanone; SAP: severe acute pancreatitis; Ser, serine; SIRT1, sirtuin 1; SOCS3, suppressor of cytokine signaling 3; SOX9, SRY-box transcription factor 9; STAT, signal transducer and activator of transcription; Tyr, tyrosine.
3.1 Herbal formulas
Da Cheng Qi Decoction (DCQD), a classical formula recorded in Shang Han Lun, consists of Rheum palmatum L. (Da Huang), Magnolia officinalis Rehd. et Wils. (Hou Po), Citrus aurantium L. (Zhi Shi), and Natrii Sulfas (Mang Xiao). Experimental studies have demonstrated that DCQD markedly reduces the protein expression of p-JAK2 (Tyr1007/1008) and p-STAT3 (Tyr705) in vitro, with an effect comparable to that of specific JAK2 or STAT3 inhibitors, suggesting that its therapeutic action primarily involves antagonism of the JAK2/STAT3 pathway [65]. High-performance liquid chromatography (HPLC) identified nine major active components in DCQD, among which rhein has been confirmed to confer protection against AP through modulation of the STAT3 signaling pathway [65,81,82]. Moreover, DCQD-mediated inhibition of JAK2/STAT3 signaling also attenuated SAP-associated intestinal injury in vivo [82].
Chaiqin Chengqi Decoction (CQCQD) and Qingyi Granules (QYG), both derived from the combination of DCQD and Dachaihu Decoction, have been shown to modulate STAT3-mediated signaling in AP [83,84]. CQCQD suppresses p-JAK2 and p-STAT3 expression in vitro and in vivo, thereby ameliorating CER-induced pancreatic injury via inhibition of the JAK2/STAT3 pathway [83]. Early administration of QYG in AP patients with acute respiratory distress syndrome significantly reduced hospital stay and mortality [84]. Consistent with these clinical findings, QYG was also reported to inhibit the IL-6/STAT3 signaling axis in a sodium taurocholate (NaT)-induced SAP rat model, mitigating both pancreatic and pulmonary injury [84]. Additional related formulations, including Chaihuang Qingyi Huoxue Granule and Qingyi Decoction, similarly attenuate AP progression through downregulation of STAT3 signaling, primarily involving the phosphoinositide 3-kinase /protein kinase B (PI3K/AKT) and NF-κB/nucleotide-binding oligomerization domain like receptor (NLR) family pyrin domain containing 3 (NLRP3) pathways [85,86].
Da-Huang-Fu-Zi-Tang (DHFZT), a classical formula originally described in Synopsis of Golden Chamber (Jin Kui Yao Lue), is composed of Rheum palmatum L. (Da Huang), Aconitum carmichaelii Debx. (Fu Zi), Asarum heterotropoides F. Schmidt (Xi Xin). Previous studies have shown that DHFZT alleviates inflammatory responses, promotes intestinal motility, and exerts protective effects in SAP [87]. A multicenter randomized controlled trial further demonstrated that DHFZT effectively reduced hospital stay, decreased mortality, and improved overall clinical outcomes in SAP patients [88]. Mechanistic investigations in a NaT-induced SAP rat model revealed that DHFZT significantly suppresses inflammatory factor expression and inhibits the activation of p-JAK2 and p-STAT3, suggesting that its therapeutic effects involve antagonism of the JAK2/STAT3 signaling pathway, thereby mitigating AP progression and alleviating SAP-associated liver injury [87].
Shenmai injection (SMI), a sterile preparation derived from Panax ginseng C.A. Mey. (Ren Shen) and Ophiopogon japonicus (L.f.) Ker Gawl. (Mai Dong), contains four major active components identified by ultra-high-performance liquid chromatography-mass spectrometry: ginsenoside Rb1, ginsenoside Rg1, ginsenoside Re, and ophiopogonin D [89]. In vitro studies indicated that these compounds enhance the viability of 266-6 cells [89]. Further investigations in both cell-based and animal models of AP demonstrated that SMI confers protective effects by inhibiting the IL-6/STAT3 signaling pathway, thereby attenuating pancreatic injury and associated inflammatory responses [89].
Rheum palmatum L. and Salvia miltiorrhiza Bge. (Dahuang and Danshen, DH-DS), recorded in Shennong's Classic of Materia Medica (Shen Nong Ben Cao Jing) for their properties of clearing dampness-heat and resolving stagnation, have been investigated for their potential to modulate the pathogenesis of AP. Experimental studies demonstrated that DH-DS significantly reduces inflammatory cytokine levels and suppresses the expression of p-JAK2, p-STAT3, and IL-17A in the CER-induced AP model. These effects indicate that DH-DS exerts protective actions by modulating the JAK2/STAT3/IL-17A signaling axis [90]. Mechanistically, upregulation of IL-17A promotes Th17 cell differentiation, thereby amplifying inflammatory responses. In addition, molecular docking and dynamics simulations confirmed a strong binding affinity between DH-DS, particularly its active component Danshexinkum D, and the STAT3 protein, supporting the molecular basis for its regulatory effects on the STAT3 pathway [90].
Traditional chinese medicine (TCM) formulations exert multi-component, multi-target regulatory effects in the treatment of AP. Studies have demonstrated that commonly used formulas, including DCQD, CQCQD, QYG, and DHFZT, can downregulate inflammatory cytokines, inhibit the STAT3 signaling pathway, reduce acinar cell apoptosis, and protect against AP-associated intestinal and pulmonary injuries. Mechanistic studies have further identified active components of DCQD, QYG, DHFZT, and SMI via HPLC analysis, and research on DCQD and CQCQD has employed specific JAK2/STAT3 inhibitors to provide pharmacological validation of their effects. Despite these advances, several limitations remain. First, the experimental models employed are relatively homogeneous, and investigations have largely focused on STAT3, with limited systematic exploration of other STATs family members, restricting a comprehensive understanding of the STATs network in AP. Second, genetic editing approaches, such as CRISPR-Cas9, have not been applied to directly validate putative targets, limiting mechanistic depth. Third, chemical profiling does not equate to mechanistic verification. Although most formulas undergo HPLC analysis to identify constituents, the functional link between these specific compounds and STAT3 inhibition is rarely established experimentally. Their relevance is often merely predicted via network pharmacology or molecular docking, without direct evidence delineating which components actually drive the pathway modulation. Consequently, the active principles remain largely speculative rather than validated, representing a critical gap between phytochemical characterization and mechanistic proof. Moreover, clinical safety profiles and potential adverse effects of these formulations remain inadequately characterized.
3.2 Herbal-derived single compounds
3.2.1. Polyphenols
Polyphenolic monomers, which are abundant in TCM herbs such as Scutellaria baicalensis Georgi (Huang Qin) and Magnolia officinalis Rehd. et Wils. (Hou Po), mainly comprise flavonoids and phenylpropanoids. Among flavonoid monomers, those shown to modulate the STATs signaling pathway in AP include baicalein (5,6,7-trihydroxyflavone), baicalin (7-D-glucuronic acid-5,6-dihydroxyflavone), and luteolin (3',4',5,7-tetrahydroxyflavone). Similarly, phenylpropanoid monomers with reported STATs-regulatory activity include daphnetin (7,8-dihydroxy-2H-1-benzopyran-2-one) and honokiol (3',5-di(prop-2-en-1-yl)[1,1'-biphenyl]-2,4'-diol). Moreover, curcumin (1,6-Heptadiene-3,5-dione, 1,7-bis(4-hydroxy-3-methoxyphenyl)-, (E,E)-), 4-(2-acetoxy-3-((R)-3-(benzylthio)-1-methoxy-1-oxopropan-2-ylamino)-3-oxopropyl)-1,2-phenylene diacetate (DSC), a danshensu-derived compound and (R)-4,6-dimethoxy-3-(4-methoxy phenyl)-2,3-dihydro-1H-indanone [(R)-TML104] have also been shown to possess relevant regulatory effects on the STATs pathway. Current studies indicate that most monomeric compounds confer protective effects against AP primarily via modulation of the STAT3 signaling pathway. Honokiol is notable as the only compound reported to influence the STAT1 pathway. Baicalein and baicalin have been shown to downregulate the protein expression of p-JAK2 and p-STAT3, thereby inhibiting JAK2/STAT3 signaling and attenuating AP-related injury [91,92]. Interestingly, their upstream mechanisms differ: baicalein regulates mitogen-activated protein kinase (MAPK) activity [91], whereas baicalin modulates the expression of B7 homolog 4, an established biomarker of type I diabetes progression [92]. Notably, luteolin downregulates p-STAT3 (Tyr705), the inflammatory transcription factor SRY-box 9, and phosphorylated epidermal growth factor receptor, thereby suppressing ADM, reversing epithelial-mesenchymal transition, and blocking the progression from pancreatitis to pancreatic cancer [93]. Several studies have investigated how monomeric compounds protect extra-pancreatic organs in AP. For example, daphnetin downregulated the expression of p-JAK2 (Tyr1007) and p-STAT3 (Tyr705) in lung tissue, thereby attenuating pancreatic and pulmonary injury in an arginine (ARG)-induced AP model [94]. Honokiol suppressed high-mobility group protein B1 (HMGB1), p-JAK1 (Tyr1034/1035), p-JAK2 (Tyr1007/1008), and p-STAT1 (Ser727) protein levels, leading to inhibition of the JAK1/2-STAT1 pathway and amelioration of SAP-associated intestinal injury [95]. Under inflammatory conditions, macrophages release substantial amounts of HMGB1 into the intestinal tract, thereby compromising gut barrier integrity and exacerbating SAP. Similarly, butyrate, a short-chain fatty acid, alleviates intestinal inflammation by inhibiting the GPR109A-NLRP3 axis and suppressing the histone deacetylase 1 (HDAC1)/STAT1/NLRP3 pathway in the pancreas, resulting in reduced pancreatic and intestinal damage in the CER-AP model [96]. Additionally, curcumin downregulated renal p-JAK2 and p-STAT3 expression, thereby mitigating pancreatic and renal injury in a NaT-AP model [97]. Moreover, both in vivo and in vitro models of CER-induced AP have demonstrated that DSC downregulates p-STAT3 (Tyr705) and the NLRP3 inflammasome, thereby inhibiting the STAT3/NLRP3 signaling axis and modulating macrophage polarization, which ultimately attenuates pancreatic injury [98]. As another novel compound, (R)-TML104 has been shown in CER-induced AP to markedly enhance the interaction between SIRT1 and STAT3, leading to reduced STAT3 acetylation. Consequently, it attenuates the IL-6/STAT3 pathway mediated inflammatory response, decreases chemokine expression, modulates the immune microenvironment, and ultimately mitigates pancreatic injury [56]. In addition, sodium selenite, commonly supplemented in AP patients with selenium deficiency, has been shown to suppress the MAPK/NF-κB/STAT3 pathway, conferring protection against AP [99]. Overall, polyphenolic monomers modulate STATs signaling through distinct patterns: baicalein, baicalin, daphnetin and curcumin downregulate both p-JAK2 and p-STAT3; luteolin, DSC and (R)-TML104 reduce p-STAT3 levels without documented JAK2 involvement; honokiol uniquely inhibits the JAK1/2-STAT1 axis. However, a critical gap remains across this class of compounds: direct evidence of physical interaction with STAT proteins is largely absent. To date, molecular docking predictions or biophysical binding assays are missing for the majority of these polyphenols. While functional causality has been supported by inhibitor-based studies, genetic knockout models, or protein interaction assays for compounds such as honokiol, butyrate, and (R)-TML104, the link between the remaining monomers and STAT3 inhibition remains primarily correlative. Consequently, future research requires definitive validation using genetic loss-of-function models to confirm these targets.
3.2.2. Terpenoids
Terpenoids monomers, commonly found in traditional medicinal herbs, have been shown to exert anti-inflammatory effects in AP primarily by modulating the STAT3 signaling pathway. Studies indicate that picroside II (6-vanilloylcatalpol) and lutein suppress the phosphorylation of JAK2 and STAT3, while limonin (7,16-dioxo-7,16-dideoxylimondiol) reduces JAK2 and p-STAT3 activation [100-102]. These effects converge on inhibition of the JAK2/STAT3 pathway, thereby ameliorating AP. Specifically, picroside II [100] and limonin [101] directly suppress the activation of this pathway, whereas lutein [102] indirectly modulates its activity by upregulating PPAR-γ and SOCS3. In addition, escin sodium and glycyrrhizin attenuate pancreatic injury by reducing p-extracellular signal-regulated kinase (ERK)1/2 and p-STAT3 levels, thereby inhibiting the ERK/STAT3 pathway [103,104]. Notably, computational docking and molecular dynamics simulations suggest that glycyrrhizin potentially binds to MAPK3 to form a stable complex, thereby implying a suppressive effect on the MAPK/ERK/STAT3/AKT signaling axis and a subsequent reduction of necrosis in pancreatic acinar cells (PACs) [104]. Notably, glycyrrhizin is the only terpenoid in this group with a proposed binding mechanism. However, this interaction remains in silico and awaits physical validation. For all other terpenoids, the molecular basis of STAT3 inhibition remains entirely undefined. Without any target engagement data—even at the predictive level—their suppressive effects on STAT3 signaling are inferred solely from observed changes in phosphorylation, which should be considered correlative rather than causative.
3.2.3. Anthraquinones
Rhein (4,5-dihydroxyanthraquinone-2-carboxylic acid), a major active component of DCQD, has been shown to exert notable anti-inflammatory effects [105]. Previous studies have demonstrated that rhein alleviates CER plus lipopolysaccharide-induced SAP by modulating glycerophospholipid metabolism in pancreatic tissue [106]. Furthermore, recent studies consistently indicate that rhein downregulates the expression of p-JAK2 (Tyr1007/1008) and p-STAT3 (Tyr705), thereby suppressing the JAK2/STAT3 signaling pathway and attenuating AP [81]. Notably, molecular docking studies suggest that rhein potentially binds to JAK2 with binding sites that partially overlap the ATP pocket, which provides a predictive structural basis for its inhibitory role [81]. However, this interaction awaits biophysical validation to confirm actual target engagement. Moreover, the specific quantitative contribution of rhein to the overall efficacy of the DCQD formula remains undefined, and its clinical translation for AP treatment has yet to be initiated.
3.2.4. Others
Alkaloid and glycoside constituents similarly modulate inflammation in AP through regulation of the STATs signaling pathway. Representative compounds include the alkaloids colchicine (N-Acetyl trimethylcolchicinic acid methylether) and rutaecarpine (8,13-dihydroindolo(2',3':3,4)pyrido(2,1-b)quinazolin-5(7H)-one), along with the glycoside kinsenoside ((3R)-5-oxooxolan-3-yl beta-D-glucopyranoside). Colchicine has been shown to inhibit p-STAT3 expression in pancreatic and lung tissues, reduce inflammatory cytokine levels, and attenuate oxidative stress and apoptosis, thereby mitigating pancreatic and pulmonary injury in a NaT-induced SAP rat model [107]. Likewise, rutaecarpine downregulates p-STAT3 expression and suppresses associated inflammatory pathways, including MAPK and NF-κB, thereby exerting protective effects against AP [108]. Distinct from the previously discussed monomers, both in vivo and in vitro studies have shown that kinsenoside downregulates p-STAT1 and reduces inflammatory cytokine levels, inhibits the toll-like receptor 4 (TLR4)-STAT1 pathway, and modulates M1 macrophage polarization, thereby attenuating pancreatic and pulmonary injury in a SAP mouse model and decreasing apoptosis in CER-treated PACs [109]. Network pharmacology analysis identified TLR4 as a key target mediating kinsenoside's protective effects in AP, a conclusion further supported by molecular docking results showing strong binding affinity between kinsenoside and TLR4 [109]. Similarly, lactate (2-hydroxypropanoate), a glycolytic end-product, promotes the shift of macrophages from the M1 to M2 phenotype, suppresses the GPR132 (a macrophage membrane receptor)/JAK2/STAT1 signaling axis, reduces inflammatory cytokine secretion, and improves the immune microenvironment, ultimately conferring protection against AP [110]. Colchicine and rutaecarpine further expand the range of potential STAT3 inhibitors, while kinsenoside and lactate appear to shift the regulatory focus toward STAT1 through suggested upstream targets such as TLR4 and GPR132 [107-110]. However, the current evidentiary basis for these compounds remains preliminary, as it has not yet been demonstrated that any of these agents directly interact with components of the STATs signaling pathways. Furthermore, because no inhibitor-based experiments or genetic loss-of-function studies have been reported, the functional causality between these compounds and their observed effects remains largely untested. The STAT1-targeting candidates, despite findings from molecular docking or pharmacological inferences, currently lack biophysical validation and causal confirmation, which represents a research gap similar to the limitations previously observed for STAT3-oriented monomers.
Monomeric compounds exert multi-target regulatory effects on inflammatory signaling pathways in AP. Current studies indicate that a majority of investigated monomers alleviate AP-associated inflammation by downregulating JAK2/STAT3 pathway activity, including inhibition of JAK2 and STAT3 phosphorylation and reduction of STAT3 nuclear translocation. Notably, some monomers confer protection against extra-pancreatic organ injury. Daphnetin, colchicine, and kinsenoside ameliorate AP-associated lung damage via STATs signaling inhibition, curcumin mitigates AP-related renal injury through suppression of the JAK2/STAT3 pathway, honokiol and butyrate alleviate intestinal injury via inhibition of STAT1 signaling. Particularly, luteolin has revealed a potential role for STAT3 in the transition from AP inflammation to carcinogenesis. Furthermore, DSC, kinsenoside, and lactate promote M1-to-M2 macrophage polarization via STATs pathway modulation, thereby regulating the immune microenvironment and mitigating AP progression. Nevertheless, current studies are largely constrained by the reliance on single animal models and the absence of dynamic mechanistic analyses across multiple models and time points. Consistent with research on TCM formulas, studies on monomeric compounds have predominantly focused on the STAT3 pathway, with only a few, including honokiol, butyrate, and kinsenoside, examining STAT1 regulation. Systematic investigations into other STATs family members remain limited. Mechanistically, few studies have employed pharmacological interventions using small-molecule inhibitors or agonists, and genetic editing approaches have rarely been applied to functionally validate specific STATs pathway targets. Furthermore, most experimental studies tend to utilize prophylactic dosing regimens, which frequently deviate from the actual clinical therapeutic window where treatment usually commences only after the onset of systemic inflammation. This methodological discrepancy may restrict the translational medicine value of the existing evidence, as it does not fully reflect the therapeutic challenges encountered in clinical practice. Future research should therefore prioritize a more comprehensive exploration of these downstream effects, such as DNA binding and epigenetic regulation, while focusing on administration strategies that are more clinically relevant to thoroughly assess the therapeutic potential of monomeric compounds in AP therapy.
Despite their potent regulatory effects on STAT signaling, the clinical translation of these monomers is largely dictated by their pharmacokinetic (PK) profiles. A primary challenge shared by polyphenols (e.g., curcumin, baicalein), terpenoids (e.g., limonin, picroside II) and anthraquinones (rhein) is limited oral bioavailability, often resulting from poor aqueous solubility, extensive first-pass metabolism, or active efflux mediated by P-glycoprotein (P-gp) [111-114]. Despite these barriers, monomers achieve therapeutic thresholds in the pancreas through several distinct mechanisms. Many monomers exist primarily as metabolites in vivo, which may serve as the actual pharmacodynamic basis. Rhein is rapidly absorbed and converted into glucuronide and sulfate forms, which undergo enterohepatic circulation; its linear PK properties ensure consistent systemic exposure despite low solubility [113,115]. Similarly, rutaecarpine undergoes extensive phase I and II metabolism (hydroxylation and conjugation), maintaining its potent induction of hepatic CYP1A2 even when parent plasma levels are nearly undetectable (below 10 ng/mL) [116,117]. In contrast, kinsenoside is characterized by rapid assimilation and clearance, with its stability significantly influenced by chemical hydrolysis of its lactone group rather than enzymatic degradation [118,119]. Effective modulation of the JAK/STAT axis requires drugs to penetrate the blood-pancreatic barrier. While colchicine (derived from colchicum autumnale) exhibits a low oral bioavailability (< 8%), its high volume of distribution suggests extensive tissue uptake, though its use is constrained by a narrow therapeutic window and potential hepatotoxicity [120]. For rhein, structural modifications have proven successful in enhancing targeting; specifically, ester and amide derivatives like HPDM-rhein have demonstrated a superior ability to cross the blood-pancreatic barrier and accumulate in the lungs and pancreas, directly addressing the systemic complications of AP [113]. To overcome the inherent PK limitations of natural products, innovative delivery systems and synergistic pharmacological strategies are employed. Nano-formulations, such as PEGylated liposomes for curcumin and PLGA-nanoparticles for lutein, can increase the AUC by 3- to 77-fold [121,122]. Furthermore, glycyrrhizin acts as a complex PK modulator; it can reduce the systemic exposure of co-administered drugs like paeoniflorin via P-gp induction, yet it may also delay the clearance of others, leading to increased toxicity [123,124]. Conversely, kinsenoside shows minimal interference with CYP450 enzymes, suggesting a higher safety profile for combination therapies [118]. In summary, the translational potential of these monomers relies on a multidimensional PK strategy: utilizing metabolic activation, leveraging vascular permeability, and employing advanced delivery platforms to ensure that STAT-modulating agents reach sufficient concentrations within the diseased pancreatic microenvironment.
3.3 Chemical drugs
Glucocorticoids, including dexamethasone (DEX), are widely employed for their potent anti-inflammatory and immunosuppressive effects. In both MAP and SAP rat models, DEX has been demonstrated to attenuate pancreatic injury by downregulating inflammatory cytokines and chemokines, disrupting STAT3-DNA binding, and suppressing the MAPK/NF-κB/STAT3 signaling pathway [125]. Compared to the model group, DEX administration resulted in pronounced reductions in MAPK kinase activity and IL-1β levels during the early phase of AP (3 hours), whereas NF-κB activity and STAT3 expression were significantly suppressed at later stages in MAP (12 hours) and at the early phase in SAP (3 hours), respectively, indicating a time-dependent therapeutic effect [125]. Notably, another study demonstrated that DEX mitigates SAP-associated lung injury by downregulating intercellular adhesion molecule-1, JAK2, STAT3, and pro-inflammatory cytokines, thereby suppressing the JAK2/STAT3 signaling axis [126]. These findings collectively suggest that DEX may protect against both pancreatic and extra-pancreatic organ damage in a temporally regulated manner during AP progression.
Thiamine (vitamin B1, VB1) is an essential micronutrient that plays a pivotal role in cellular energy metabolism. PACs primarily depend on thiamine transporter-1 and -2 (THTR-1 and THTR-2) for VB1 uptake from the circulation. Exposure of PACs to elevated levels of inflammatory cytokines (IL-6, IL-1β, TNF-α) for 24 hours has been shown to markedly impair VB1 uptake [127]. Treatment with the STAT3-specific inhibitor S3I-201 downregulated p-STAT3 (Tyr705) levels, thereby inhibiting the IL-6/STAT3/THTR pathway and restoring IL-6-induced reductions in VB1 uptake in 266-6 cells [127]. These findings indicate that modulation of the STAT3 pathway may represent a potential strategy to enhance energy metabolism in PACs, although in vivo validation remains lacking.
Alprostadil, a prostaglandin E1 analog, is widely used in the management of urological disorders and has recently attracted attention for its potential therapeutic application in inflammatory conditions. In an ARG-induced SAP rat model, levels of malondialdehyde (MDA), myeloperoxidase (MPO), and pro-inflammatory cytokines (IL-6, IL-1β, TNF-α) were significantly elevated, whereas superoxide dismutase activity was reduced, reflecting exacerbated oxidative stress and inflammatory responses [128]. Administration of alprostadil markedly reversed these changes and concurrently downregulated the expression of JAK2 and STAT3 proteins. Notably, the therapeutic efficacy of alprostadil was comparable to that of the JAK2-specific inhibitor AG490, indicating that alprostadil mitigates SAP progression via inhibition of the JAK2/STAT3 signaling pathway [128].
Carvedilol, a non-selective β-adrenergic blocker, has been shown to exert protective effects by attenuating oxidative stress and inflammatory responses. In an ARG-induced AP rat model, oxidative stress markers, including MDA, were significantly elevated, whereas antioxidant factors such as glutathione and catalase were decreased. Concurrently, inflammatory mediators, including C-reactive protein, IL-1β, TNF-α, and MPO, were markedly increased [129]. Administration of carvedilol ameliorated these alterations and markedly downregulated the mRNA expression of PAP2 and platelet-activating factor, as well as the protein levels of NF-κB p65, MAPK-p38, and STAT1. These findings indicate that carvedilol mitigates SAP progression primarily through inhibition of the MAPK/NF-κB/STAT1 signaling pathway [129].
According to the 2025 revised guidelines, the routine management of AP mainly comprises analgesia, fluid resuscitation, and nutritional support, as no specific pharmacological therapy has been firmly established [130]. Nonetheless, recent preclinical studies have demonstrated that several western drugs, including DEX, VB1, alprostadil, and carvedilol, can attenuate inflammation, enhance microcirculation, and modulate metabolic dysfunction in AP models, primarily through modulation of STATs-related signaling pathways. These findings offer valuable experimental evidence for optimizing therapeutic windows and designing intervention strategies. Nevertheless, current research remains largely confined to STAT3-associated pathways, and the functional roles of other STATs family members remain insufficiently characterized. Notably, except for DEX pharmacological studies, which employed both MAP and SAP models at multiple time points, most investigations are limited by the use of single models and static observational time points. Such an approach does not allow a systematic evaluation of drug effects across diverse etiologies and disease stages. Importantly, the lack of established positive control drugs not only diminishes the comparability and persuasiveness of the results but also underscores the current absence of targeted therapeutics for SAP. Furthermore, since these drugs have not been incorporated into clinical practice for AP, their efficacy and safety profiles require further validation through large-scale prospective studies and clinical trials. Consequently, their inclusion in international guidelines and standard treatment protocols remains a long-term objective.
4. Clinical Evidence
Beyond experimental and pharmacological studies, clinical evidence highlights the pivotal role of STATs signaling in AP pathophysiology. Altered STATs activity has been observed in immune cells of SAP patients, correlating with inflammatory status, disease progression, and systemic complications. A concise overview of these findings is presented in Figure 4, which serve as references for the subsequent discussion.
Abnormal alterations of the STATs family in immune cells of AP patients. Members of the STATs family exhibit dysregulated signaling in monocytes, lymphocytes, and neutrophils of AP patients, further modulating immune function and inflammation. In monocytes, IL-6 stimulation results in reduced levels of p-STAT3 (Tyr705) and p-STAT1 (Tyr701). This reduction, combined with downregulation of HLA-DR expression, contributes to an “immunoparalysis” phenotype. However, normal p-STAT5 (Tyr694) expression under GM-CSF stimulation can partially alleviate this immunosuppressed state. Moreover, decreased p-STAT3 (Tyr705) disrupts the balance between pro-inflammatory cytokines (TNF-α, IL-1β) and the anti-inflammatory cytokine IL-10. In neutrophils, p-STAT5 (Tyr694) remains largely unchanged upon GM-CSF stimulation; in contrast, IL-6 induces an increase in p-STAT3 (Tyr705), whereas IL-4 leads to decreased p-STAT6 (Tyr641). These alterations are associated with inflammation, organ dysfunction, and mortality in AP patients. In lymphocytes, p-STAT3 (Tyr705) remains constitutively active under unstimulated conditions; following IL-6 stimulation, p-STAT1 (Tyr701) activation is impaired, while IL-4 stimulation enhances p-STAT6 (Tyr641) activation. This signaling imbalance contributes to an increased risk of secondary infections in AP patients. Abbreviations: AP, acute pancreatitis; GM-CSF, granulocyte-macrophage colony-stimulating factor; HLA-DR, human leukocyte antigen-DR; IL, interleukin; STAT, signal transducer and activator of transcription; TNF-α, tumor necrosis factor-α; Tyr, tyrosine.
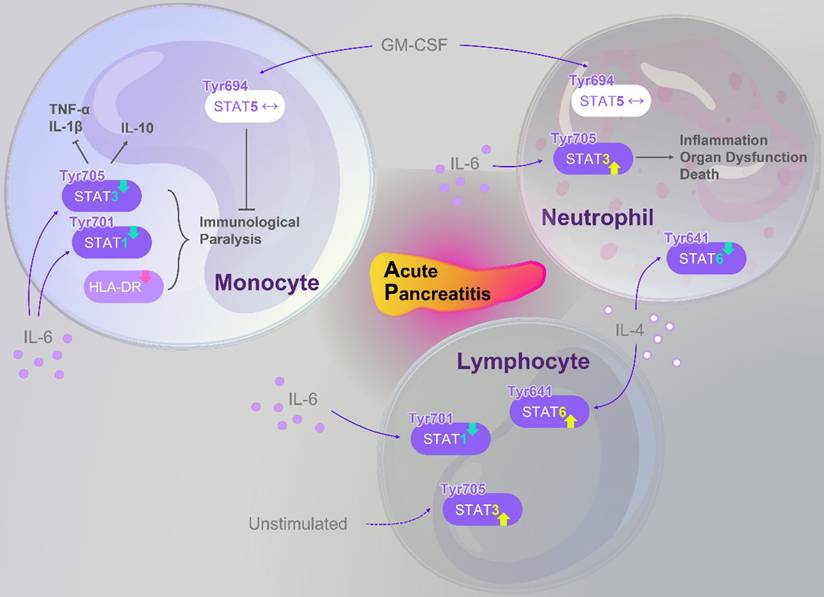
4.1 Abnormal STATs signaling in immune cells of SAP patients
Current clinical research on STAT signaling in AP is predominantly based on observational profiling, with direct mechanistic insights remaining exceptionally rare. Based on observational clinical data, studies have demonstrated that the STATs family exhibits a certain common regulatory pattern in different immune cells (lymphocytes, monocytes, and neutrophils) of patients with SAP complicated by MODS. As upstream signaling molecules of the STATs family, p-ERK1/2 (Thr202/Tyr204) and NF-κB p-p65 (Ser529) are downregulated in lymphocytes, monocytes, and neutrophils isolated from SAP patients [131-133]. Compared with healthy individuals, the expression level of p-STAT1 (Tyr701) in lymphocytes and monocytes of SAP patients is significantly reduced following stimulation with IL-6 [131,132]. However, no significant changes are observed in the level of MAPK-p-p38 (Thr180/Tyr182) in monocytes and neutrophils [132,133]. Similarly, no significant alteration is detected in the level of p-STAT5 (Tyr694) in these cells following stimulation with granulocyte-macrophage colony-stimulating factor [132,133]. These shared alterations suggest that the STATs family and its associated signaling pathways may exhibit global regulatory abnormalities in the immune dysregulation of SAP.
In terms of cell type-specific manifestations, lymphocytes exhibit distinct abnormalities in STATs signaling. Under basal conditions without exogenous cytokine stimulation, the proportion of p-STAT3 (Tyr705)-positive lymphocytes in SAP patients reaches 42.0%±4.7%, which is significantly higher than that in healthy controls (2.6%±0.1%, P < 0.001), and STAT3 shows sustained activation [131]—a phenomenon not observed in monocytes [132]. In addition, lymphocytes display an imbalance between STAT1 and STAT6 signaling, characterized by downregulation of p-STAT1 (Tyr701) upon IL-6 stimulation and upregulation of p-STAT6 (Tyr641) upon IL-4 stimulation [131]. Given that STAT1 is a key regulator of Th1 cell differentiation and effector function, whereas STAT6 is critical for Th2 cell differentiation, this signaling imbalance reflects suppression of Th1-mediated cellular immunity and activation of Th2-mediated humoral responses, thereby creating a gap in cellular immune defense and increasing the risk of secondary infections in SAP patients.
In contrast to the activation pattern of STAT3 in lymphocytes, monocytes exhibit distinct STATs signaling features upon IL-6 stimulation. Compared with healthy controls, the proportion of p-STAT3 (Tyr705)-positive monocytes in SAP patients is significantly reduced (75.3%±5.7% vs 97.3%±0.3%) [132]. STAT3 activation in monocytes normally promotes the production of the anti-inflammatory cytokine IL-10 and suppresses pro-inflammatory cytokines such as TNF-α and IL-1β [134,135]. Disruption of this activation impairs the anti-inflammatory function of monocytes, rendering the STAT3 signaling pathway in these cells more severely compromised than in lymphocytes. Notably, monocytes from SAP patients also show reduced p-STAT1 (Tyr701) levels and marked downregulation of human leukocyte antigen-DR expression (55.0%±4.1% vs 93.1%±3.4%, P < 0.001) compared with healthy controls [132]. Together with weakened STAT3 phosphorylation, these alterations constitute a hallmark of monocyte “immunoparalysis”, reflecting diminished responsiveness to pathogens and damage signals. This immunological state, typically observed in patients classified as SAP according to the Atlanta classification [132], represents a key factor contributing to the development of intra-abdominal abscesses and sepsis, which occurred in 69.2% of the patients within the observed sample [132].
Further comparison of STATs alterations across different immune cell types reveals opposing trends in monocytes and neutrophils regarding p-STAT3 (Tyr705) levels. In SAP patients, the proportion of p-STAT3-positive neutrophils following IL-6 stimulation was significantly increased compared with healthy controls (38.1%±8.4% vs 10.7%±2.2%, P = 0.016) [133], suggesting a potential role in inflammation and mortality-associated signaling. Additionally, neutrophil STAT6 abnormalities differed markedly from those in lymphocytes. Upon IL-4 stimulation, p-STAT6 (Tyr641) fluorescence intensity in neutrophils from SAP patients was significantly reduced relative to healthy controls [133]. Given that STAT6 indirectly regulates anti-inflammatory responses and the clearance of apoptotic cells, its impaired activation may compromise inflammation resolution in SAP patients. Clinical data show reduced STAT6 in SAP, but it remains unclear whether therapeutically restoring STAT6 might paradoxically interfere with phagocytic clearance of translocated bacteria while suppressing inflammation.
4.2 Roles of STATs signaling in severity evaluation, prognosis prediction, and complication risk of AP
To ensure clinical diagnostic rigor, the studies discussed herein primarily defined the severity of AP according to the Atlanta or revised Atlanta classification systems [132,136,137]. Acting as observational biomarkers, Turunen et al. demonstrated that the dynamic evolution of STAT3 and STAT1 phosphorylation reflects differences in clinical outcomes among AP patients. At the STAT3 level, upon admission, monocytes and neutrophils from AP patients exhibited elevated constitutive STAT3 phosphorylation compared with healthy controls, regardless of the presence of persistent organ dysfunction (OD) [136]. In this context, persistent OD was rigorously defined by the Revised Atlanta Classification as a Modified Marshall Score (MMS) ≥ 2 lasting more than 48 hours [136]. Moreover, IL-6-stimulated p-STAT3 (Tyr705) levels in monocytes from both patient groups (OD- group representing moderately severe AP; OD+ group representing severe AP) were lower than those in healthy controls [136]. Between days 5 and 8 after admission, constitutive STAT3 phosphorylation in both monocytes and neutrophils declined in both groups, although monocyte levels remained higher than in controls (P = 0.007) [136]. Notably, IL-6-stimulated monocyte p-STAT3 (Tyr705) in the OD- group showed partial recovery (still below control levels), suggesting a gradual restoration of IL-6 responsiveness, which correlates with the absence of persistent OD (MMS < 2) and zero 30-day mortality in this group [136]. At the STAT1 level, IL-6-stimulated monocyte p-STAT1 (Tyr701) was lower in both patient groups compared with controls upon admission, while lymphocytes showed no significant difference [136]. This reduced monocyte p-STAT1 (Tyr701) expression persisted at days 5-8, potentially due to miR-30a-mediated targeting of STAT1 [138] or downregulation of the IL-6 receptor [139], reflecting sustained impairment of monocyte anti-infective signaling pathways in AP patients.
From the perspective of observational biomarkers for disease severity and complication prediction, the clinical relevance of aberrant STAT3, STAT1, and STAT6 phosphorylation is increasingly evident. Regarding STAT3, basal p-STAT3 (Tyr705) levels in AP patients are significantly higher than in healthy controls and correlate positively with disease severity as assessed by standardized scoring systems [140,141]. In patients sampled before the onset of OD, elevated p-STAT3 (Tyr705) levels can predict the development of persistent OD, defined by a Modified Marshall Score ≥ 2 or an acute increase in the Sequential Organ Failure Assessment score of two or more points [140,141]. In the study by Ezzat et al. [137], patients with necrotizing AP exhibited significantly higher levels of STAT3 mRNA expression compared with those with non-necrotizing AP. This elevated STAT3 expression was found to correlate with several indicators of disease severity, including the SIRS and the computed tomography severity index, as well as the presence of pancreatic necrosis [137]. However, it is important to note that STAT3 mRNA levels did not demonstrate a significant correlation with BISAP scores in this cohort. Furthermore, while receiver operating characteristic curve analysis was employed to assess its diagnostic potential, STAT3 mRNA did not show statistical significance as an independent predictor of pancreatic necrosis, yielding a P value of 0.141 [137]. Instead, the analysis focused on its ability to identify the combination of moderate and severe AP, although the resulting area under the curve of 0.64 also lacked statistical significance. These findings suggest that while STAT3 mRNA levels reflect certain inflammatory and necrotic processes in AP, their current utility as standalone prognostic biomarkers remains limited and requires further clinical validation.
Moving beyond observational correlations to mechanistic causality and therapeutic interventions, genetic and pharmacological studies provide direct evidence of STATs signaling in driving clinical outcomes. For instance, the risk allele C of the γ-glutamyltransferase 1 single nucleotide polymorphism rs5751901 increases the risk of post-endoscopic retrograde cholangiopancreatography pancreatitis by enhancing STAT3 activation [142], whereas QYG combined with DEX can effectively reduce complication incidence in AP patients by inhibiting the IL-6/STAT3 pathway, highlighting p-STAT3 as a potential preventive and therapeutic target [84]. Returning to prognostic biomarkers, changes in STAT1 display an opposite trend to STAT3. IL-6-stimulated p-STAT1 (Tyr701) levels in monocytes and lymphocytes decrease with increasing AP severity [140,141], which may result from downregulation of the IL-6 receptor [139] or reduced phosphorylation of glycoprotein 130 [143]. This hypo-responsiveness of STAT1 can predict the risk of secondary infections, providing a reference for early identification of high-risk patients. In addition, IL-4-stimulated p-STAT6 (Tyr641) levels in monocytes and neutrophils from AP patients are lower than in healthy controls [141]. Although p-STAT6 is not significantly associated with disease severity, in patients with existing OD, low p-STAT6 (Tyr641) can predict persistent OD, offering a novel molecular marker for assessing prognosis of organ failure [141]. In summary, the integration of molecular STAT signaling profiles with established clinical metrics like BISAP, APACHE II, and Marshall scores offers a more comprehensive framework for early risk stratification and prognosis prediction in AP.
Current clinical studies indicate that STATs signaling plays a crucial role in immune regulation during AP. However, several limitations remain. First, while specific alterations have been observed in lymphocytes, monocytes, and neutrophils, the complex correlations and signaling crosstalk between these distinct immune cell populations remain to be elucidated. Research remains focused on STAT1, STAT3, and STAT6, while the roles and mechanisms of STAT2, STAT4, and STAT5 remain largely unexplored. Second, current clinical evidence is disproportionately skewed toward peripheral immune cell profiling, whereas direct investigations into the STAT signaling dynamics within human pancreatic parenchymal cells remain nearly non-existent. This lack of “in-situ” clinical data hinders our understanding of the direct interplay between immune dysregulation and acinar/ductal injury. Third, the vast majority of existing data is derived from observational profiling, with a critical shortage of interventional studies, such as pharmacological modulation or genetic risk-allele analysis (e.g., SNP studies), that could definitively establish mechanistic causality. Future investigations should therefore aim to: (1) expand research beyond STAT1/3 to include the entire STAT family, focusing specifically on the coordinated crosstalk between immune and parenchymal cells to establish a holistic map of the AP microenvironment, (2) promote the development and implementation of rapid bedside detection technologies, such as integrated microfluidic chips, and (3) design large-scale, multicenter, stratified, and longitudinal clinical studies, coupled with rigorous bidirectional validation between basic and clinical research, to facilitate the transition from descriptive inflammatory profiling (observational) to precise immune modulation (mechanistic).
5. Translational Challenges and Precision Therapeutic Strategies
The functional duality of STATs, particularly STAT3, represents a significant hurdle in translating experimental findings into clinical efficacy. The recent clinical failure of the STAT3 antisense oligonucleotide danvatirsen underscores the risks of global inhibition, which can lead to unintended immune suppression [144]. Because STAT3 is indispensable for maintaining intestinal epithelial homeostasis and supporting pancreatic acinar cell regeneration during the resolution phase, indiscriminate blockade may inadvertently compromise innate repair mechanisms or exacerbate the risk of secondary infections [145,146]. Furthermore, the optimization of the therapeutic window through strategic timing is equally critical. Because the IL-22/STAT3 axis promotes essential anti-apoptotic and pro-regenerative effects during the resolution phase, a “one-size-fits-all” inhibitory approach may inadvertently delay tissue repair [147,148]. Consequently, future therapeutic efforts must move toward cell-type, organelle, or pathway selective modulation through strategic timing. A biphasic model that inhibits pro-inflammatory STATs signaling during the hyperacute phase while selectively activating restorative pathways, such as through IL-22 mRNA loaded lipid-nanoparticles, may optimize the therapeutic window [149]. Advanced delivery systems further offer a promising solution to these compartmentalized roles; for instance, fibroblast activation protein alpha targeted nanocarriers or pH responsive micelles can concentrate therapeutic agents within activated fibroblasts or damaged acinar cells while sparing systemic immune functions [150,151]. Such technologies are especially vital for TCM monomers, as their clinical utility is often hampered by low oral bioavailability and rapid systemic metabolism [152].
6. Concluding Remarks and Future Directions
In summary, this review has synthesized the critical role of the STATs family as a regulatory hub that orchestrates the complex transition from local injury to systemic inflammation in AP. While these insights provide a robust conceptual framework, translating them into therapeutic reality requires overcoming the current reliance on idealized rodent models [153,154] and the limitations of correlational protein expression analysis. To bridge this gap and establish definitive causal relationships, the field must now transition from broad observation toward genetic rescue experiments and cell-type specific investigations.
Moving forward, the field must test specific mechanistically driven hypotheses to bridge the gaps identified in the preceding sections. Primarily, it remains to be determined whether STAT3 activation in pancreatic stellate cells during early AP contributes to ductal pressure regulation by altering pancreatic architectural stiffness [155]. Simultaneously, research should investigate if pharmacological STAT6 activation, while intended to promote inflammation resolution, might inadvertently compromise the host ability to clear translocated gut bacteria [156,157]. Thirdly, studies should explore whether the STAT4 mediated Th1/Th17 balance dictates the transition from acute injury to chronic fibro inflammatory progression [158]. Finally, given that STAT1 and STAT3 activation drives premature protease activation in acinar cells [39], it remains to be seen whether activating distinct isoforms, such as STAT5 in ductal cells, can mitigate bicarbonate secretion impairment and thereby restore the alkaline environment necessary to prevent this injury.
While TCM monomers and synthetic modulators show significant potential, more rigorous preclinical studies and higher levels of clinical evidence are necessary to firmly establish their efficacy [159]. Mechanistic investigations utilizing spatiotemporal analysis will be essential to clarify stage-specific roles, while pharmacological research must focus on dose-response relationships and extra-pancreatic organ protection. Ultimately, integrating these molecular insights with precision delivery technologies that can sense the pathological microenvironment will be essential to realize individualized therapy and improve patient outcomes in AP.
Abbreviations
ADM: acinar-to-ductal metaplasia; AKT: protein kinase B; AP: acute pancreatitis; ARG: arginine; BISAP: Bedside Index of Severity in Acute Pancreatitis; CER: cerulein; CQCQD: Chaiqin Chengqi Decoction; DCQD: Da Cheng Qi Decoction; DSC: 4-(2-acetoxy-3-((R)-3-(benzylthio)-1-methoxy-1-oxopropan-2-ylamino)-3-oxopropyl)-1:2-phenylene diacetate; DH-DS: Dahuang and Danshen; DEX: dexamethasone; DHFZT: Da-Huang-Fu-Zi-Tang; ERK: extracellular signal-regulated kinase; HIP: Hepatocarcinoma-intestine-pancreas; HPLC: high-performance liquid chromatography; HMGB1: high-mobility group protein B1; IFN-γ: interferon gamma; IL: interleukin; JAK: janus kinase; MAP: mild acute pancreatitis; MAPK: mitogen-activated protein kinase; MDA: malondialdehyde; MODS: multiple organ dysfunction syndrome; MPO: myeloperoxidase; NaT: sodium taurocholate; NF-κB: nuclear factor kappa-B; NLRP3: nucleotide-binding oligomerization domain like receptor (NLR) family pyrin domain containing 3; OD: organ dysfunction; PACs: pancreatic acinar cells; PAP: pancreatitis-associated protein; P-gp: P-glycoprotein; PI3K: phosphoinositide 3-kinase; PK: pharmacokinetic; PPAR-γ: peroxisome proliferator-activated receptor gamma; PSCs: pancreatic stellate cells; QYG: Qingyi Granules; (R)-TML104: (R)-4:6-dimethoxy-3-(4-methoxy phenyl)-2:3-dihydro-1H-indanone; SAP: severe acute pancreatitis; SIRS: systemic inflammatory response syndrome; SIRT1: sirtuin 1; SMI: Shenmai injection; SOCS: suppressor of cytokine signaling; STAT: signal transducer and activator of transcription; TCM: traditional chinese medicine; THTR: thiamine transporter; TLR4: toll-like receptor 4; TNF-α: tumor necrosis factor alpha; VB1: vitamin B1.
Acknowledgements
Funding
This research was supported by the National Natural Science Foundation of China (No. 82305076), the China Postdoctoral Science Foundation (No. 2023MD744128), the Sichuan Provincial Natural Science Foundation (No. 2025ZNSFSC1825), and the Postdoctoral Science Foundation of Chengdu University of Traditional Chinese Medicine (No. BSH2023004). The authors thank all funding agencies mentioned above for their generous support.
Author contributions
YP, YT and YFM designed the study; YP, AEL, FNY, YMT collected and analyzed the data; All authors assessed the methodological quality and drew the figures; YP, AEL, FNY, YMT wrote the manuscript. All authors read and approved the final manuscript.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Trikudanathan G, Yazici C, Evans Phillips A, Forsmark CE. Diagnosis and Management of Acute Pancreatitis. Gastroenterology. 2024;167:673-688
2. Iannuzzi JP, King JA, Leong JH, Quan J, Windsor JW, Tanyingoh D. et al. Global Incidence of Acute Pancreatitis Is Increasing Over Time: A Systematic Review and Meta-Analysis. Gastroenterology. 2022;162:122-134
3. Xia C-C, Chen H-T, Deng H, Huang Y-T, Xu G-Q. Reactive oxygen species and oxidative stress in acute pancreatitis: Pathogenesis and new therapeutic interventions. World J Gastroenterol. 2024;30:4771-4780
4. Zhou Y, Huang X, Jin Y, Qiu M, Ambe PC, Basharat Z. et al. The role of mitochondrial damage-associated molecular patterns in acute pancreatitis. Biomed Pharmacother. 2024;175:116690
5. Loh C-Y, Arya A, Naema AF, Wong WF, Sethi G, Looi CY. Signal Transducer and Activator of Transcription (STATs) Proteins in Cancer and Inflammation: Functions and Therapeutic Implication. Front Oncol. 2019;9:48
6. Morris R, Kershaw NJ, Babon JJ. The molecular details of cytokine signaling via the JAK/STAT pathway. Protein Sci. 2018;27:1984-2009
7. Philips RL, Wang Y, Cheon H, Kanno Y, Gadina M, Sartorelli V. et al. The JAK-STAT pathway at 30: Much learned, much more to do. Cell. 2022;185:3857-3876
8. Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9:798-809
9. McBride KM, Banninger G, McDonald C, Reich NC. Regulated nuclear import of the STAT1 transcription factor by direct binding of importin-alpha. EMBO J. 2002;21:1754-1763
10. Cimica V, Chen H-C, Iyer JK, Reich NC. Dynamics of the STAT3 transcription factor: nuclear import dependent on Ran and importin-β1. PLoS One. 2011;6:e20188
11. Tsai M-H, Pai L-M, Lee C-K. Fine-Tuning of Type I Interferon Response by STAT3. Front Immunol. 2019;10:1448
12. Xue B, Li H, Liu S, Feng Q, Xu Y, Deng R. et al. The redox cycling of STAT2 maintains innate immune homeostasis. Cell Rep. 2022;40:111215
13. Deng F, Yang R, Yang Y, Li X, Hou J, Liu Y. et al. Visible light accelerates skin wound healing and alleviates scar formation in mice by adjusting STAT3 signaling. Commun Biol. 2024;7:1266
14. Wei L, Laurence A, O'Shea JJ. New insights into the roles of Stat5a/b and Stat3 in T cell development and differentiation. Semin Cell Dev Biol. 2008;19:394-400
15. Smith MR, Satter LRF, Vargas-Hernández A. STAT5b: A master regulator of key biological pathways. Front Immunol. 2022;13:1025373
16. Tolomeo M, Cascio A. STAT4 and STAT6, their role in cellular and humoral immunity and in diverse human diseases. Int Rev Immunol. 2024;43:394-418
17. Closa D, Motoo Y, Iovanna JL. Pancreatitis-associated protein: from a lectin to an anti-inflammatory cytokine. World J Gastroenterol. 2007;13:170-174
18. Yu JH, Kim H. Role of janus kinase/signal transducers and activators of transcription in the pathogenesis of pancreatitis and pancreatic cancer. Gut Liver. 2012;6:417-422
19. Chen P, Huang L, Zhang Y, Qiao M, Yao W, Yuan Y. The antagonist of the JAK-1/STAT-1 signaling pathway improves the severity of cerulein-stimulated pancreatic injury via inhibition of NF-κB activity. Int J Mol Med. 2011;27:731-738
20. Huang L, Chen P, Xu L, Zhou Y, Zhang Y, Yuan Y. Fractalkine upregulates inflammation through CX3CR1 and the Jak-Stat pathway in severe acute pancreatitis rat model. Inflammation. 2012;35:1023-1030
21. Hayashi T, Ishida Y, Kimura A, Iwakura Y, Mukaida N, Kondo T. IFN-gamma protects cerulein-induced acute pancreatitis by repressing NF-kappa B activation. J Immunol. 2007;178:7385-7394
22. Wang D, Tang M, Zong P, Liu H, Zhang T, Liu Y. et al. MiRNA-155 Regulates the Th17/Treg Ratio by Targeting SOCS1 in Severe Acute Pancreatitis. Front Physiol. 2018;9:686
23. Qiu Z, Xu F, Wang Z, Yang P, Bu Z, Cheng F. et al. Blockade of JAK2 signaling produces immunomodulatory effect to preserve pancreatic homeostasis in severe acute pancreatitis. Biochem Biophys Rep. 2021;28:101133
24. Ju KD, Lim JW, Kim KH, Kim H. Potential role of NADPH oxidase-mediated activation of Jak2/Stat3 and mitogen-activated protein kinases and expression of TGF-β1 in the pathophysiology of acute pancreatitis. Inflamm Res. 2011;60:791-800
25. Ding L, Li J. FXYD domain containing ion transport regulator 5 (FXYD5) silencing promotes cell viability and alleviates inflammatory response in cerulein-induced AR42J cells by blocking JAK2/STAT3 signaling pathway. Bioengineered. 2022;13:2639-2647
26. Longevity OMAC. Retracted: Hydrostatin-SN10 Ameliorates Pancreatitis-Induced Lung Injury by Affecting IL-6-Induced JAK2/STAT3-Associated Inflammation and Oxidative Stress. Oxid Med Cell Longev. 2021;2021:8541783
27. Ansari RA, Husain K, Rizvi SAA. Role of Transcription Factors in Steatohepatitis and Hypertension after Ethanol: The Epicenter of Metabolism. Biomolecules. 2016;6:29
28. Ju KD, Lim JW, Kim H. Peroxisome Proliferator-activated Receptor-gamma Inhibits the Activation of STAT3 in Cerulein-stimulated Pancreatic Acinar Cells. J Cancer Prev. 2017;22:189-194
29. Ramnath RD, Sun J, Bhatia M. Involvement of SRC family kinases in substance P-induced chemokine production in mouse pancreatic acinar cells and its significance in acute pancreatitis. J Pharmacol Exp Ther. 2009;329:418-428
30. Mateu A, De Dios I, Manso MA, Ramudo L. Oxidized phospholipids exert a dual effect on bile acid-induced CCL2 expression in pancreatic acini. Pancreatology. 2017;17:372-380
31. Zhang H, Neuhöfer P, Song L, Rabe B, Lesina M, Kurkowski MU. et al. IL-6 trans-signaling promotes pancreatitis-associated lung injury and lethality. J Clin Invest. 2013;123:1019-1031
32. Pini M, Rhodes DH, Castellanos KJ, Hall AR, Cabay RJ, Chennuri R. et al. Role of IL-6 in the resolution of pancreatitis in obese mice. J Leukoc Biol. 2012;91:957-966
33. Chao KC, Chao KF, Chuang CC, Liu SH. Blockade of interleukin 6 accelerates acinar cell apoptosis and attenuates experimental acute pancreatitis in vivo. Br J Surg. 2006;93:332-338
34. Miao B, Qi W-J, Zhang S-W, Wang H, Wang C, Hu L. et al. miR-148a suppresses autophagy by down-regulation of IL-6/STAT3 signaling in cerulein-induced acute pancreatitis. Pancreatology. 2019;19:557-565
35. Wei H, Zhao H, Cheng D, Zhu Z, Xia Z, Lu D. et al. miR-148a and miR-551b-5p regulate inflammatory responses via regulating autophagy in acute pancreatitis. Int Immunopharmacol. 2024;127:111438
36. Yang Y, Sun Z, Li J, Song Y, Xu W. Neutrophil-derived IL-10 increases CVB3-induced acute pancreatitis pathology via suppressing CD8+T cell activation while increasing macrophage STAT3-IL-6 cascade. Cytokine. 2024;184:156784
37. Xue J, Nguyen DTC, Habtezion A. Aryl hydrocarbon receptor regulates pancreatic IL-22 production and protects mice from acute pancreatitis. Gastroenterology. 2012;143:1670-1680
38. Jin M, Zhang H, Wu M, Wang Z, Chen X, Guo M. et al. Colonic interleukin-22 protects intestinal mucosal barrier and microbiota abundance in severe acute pancreatitis. FASEB J. 2022;36:e22174
39. Robinson K, Vona-Davis L, Riggs D, Jackson B, McFadden D. Peptide YY attenuates STAT1 and STAT3 activation induced by TNF-alpha in acinar cell line AR42J. J Am Coll Surg. 2006;202:788-796
40. Zhang D, Man X, Li L, Tang J, Liu F. Radiocontrast agent and intraductal pressure promote the progression of post-ERCP pancreatitis by regulating inflammatory response, cellular apoptosis, and tight junction integrity. Pancreatology. 2022;22:74-82
41. Wen L, Javed TA, Yimlamai D, Mukherjee A, Xiao X, Husain SZ. Transient High Pressure in Pancreatic Ducts Promotes Inflammation and Alters Tight Junctions via Calcineurin Signaling in Mice. Gastroenterology. 2018;155:1250-1263.e5
42. Ren Y, Liu W, Zhang J, Bi J, Fan M, Lv Y. et al. MFG-E8 Maintains Cellular Homeostasis by Suppressing Endoplasmic Reticulum Stress in Pancreatic Exocrine Acinar Cells. Front Cell Dev Biol. 2021;9:803876
43. Ren Y, Liu W, Zhang L, Zhang J, Bi J, Wang T. et al. Milk fat globule EGF factor 8 restores mitochondrial function via integrin-medicated activation of the FAK-STAT3 signaling pathway in acute pancreatitis. Clin Transl Med. 2021;11:e295
44. Liang T, Zhang Z, Bai Z, Xu L, Xu W. STAT3 Increases CVB3 Replication and Acute Pancreatitis and Myocarditis Pathology via Impeding Nuclear Translocation of STAT1 and Interferon-Stimulated Gene Expression. Int J Mol Sci. 2024;25:9007
45. Sun L, Spiteri AG, Griffith BD, Zhang Y, Di Magliano MP, Olivei AC. et al. IL-22BP Modulates Injury in Acute Pancreatitis but Delays Tissue Recovery in Chronic Pancreatitis. Cell Mol Gastroenterol Hepatol. 2025;19:101520
46. Li M, Zhang X, Wang B, Xu X, Wu X, Guo M. et al. Effect of JAK2/STAT3 signaling pathway on liver injury associated with severe acute pancreatitis in rats. Exp Ther Med. 2018;16:2013-2021
47. Li S, Cui H-Z, Xu C-M, Sun Z-W, Tang Z-K, Chen H-L. RUNX3 protects against acute lung injury by inhibiting the JAK2/STAT3 pathway in rats with severe acute pancreatitis. Eur Rev Med Pharmacol Sci. 2019;23:5382-5391
48. Lv J, Fang M, Sun S, Wang G, Fu S, Sun B. et al. Blockade of the Arid5a/IL-6/STAT3 axis underlies the anti-inflammatory effect of Rbpjl in acute pancreatitis. Cell Biosci. 2022;12:95
49. Chen W-D, Zhang J-L, Wang X-Y, Hu Z-W, Qian Y-B. The JAK2/STAT3 signaling pathway is required for inflammation and cell death induced by cerulein in AR42J cells. Eur Rev Med Pharmacol Sci. 2019;23:1770-1777
50. Qin M-Z, Qin M-B, Liang Z-H, Tang G-D. Effect of SOCS3 on lung injury in rats with severe acute pancreatitis through regulating JAK2/STAT3 signaling pathway. Eur Rev Med Pharmacol Sci. 2019;23:10123-10131
51. Xu W, Cai W, Wu J, Hou X, Qi X, Wei Z. et al. Mesenchymal stem cell-derived apoptotic extracellular vesicles loaded with Prussian blue nanoparticles attenuate severe acute pancreatitis via neutrophil extracellular traps resolution and acinar-ductal metaplasia promotion. J Nanobiotechnology. 2025;23:804
52. Shigekawa M, Hikita H, Kodama T, Shimizu S, Li W, Uemura A. et al. Pancreatic STAT3 protects mice against caerulein-induced pancreatitis via PAP1 induction. Am J Pathol. 2012;181:2105-2113
53. Saad MI, Weng T, Lundy J, Gearing LJ, West AC, Harpur CM. et al. Blockade of the protease ADAM17 ameliorates experimental pancreatitis. Proc Natl Acad Sci U S A. 2022;119:e2213744119
54. Marimuthu MK, Moorthy A, Ramasamy T. Diallyl Disulfide Attenuates STAT3 and NF-κB Pathway Through PPAR-γ Activation in Cerulein-Induced Acute Pancreatitis and Associated Lung Injury in Mice. Inflammation. 2022;45:45-58
55. Gironella M, Folch-Puy E, LeGoffic A, Garcia S, Christa L, Smith A. et al. Experimental acute pancreatitis in PAP/HIP knock-out mice. Gut. 2007;56:1091-1097
56. Ren Z-N, Yang J, Zhang M-Y, Huang Y-W, Song D-X, Sun X. et al. A novel resveratrol analog upregulates sirtuin 1 and inhibits inflammatory cell infiltration in acute pancreatitis. Acta Pharmacol Sin. 2022;43:1264-1273
57. Yubero S, Ramudo L, Manso MA, De Dios I. The role of redox status on chemokine expression in acute pancreatitis. Biochim Biophys Acta. 2009;1792:148-154
58. Ferrero-Andrés A, Closa D, Roselló-Catafau J, Folch-Puy E. Polyethylene Glycol 35 (PEG35) Modulates Exosomal Uptake and Function. Polymers (Basel). 2020;12:3044
59. Li J, Pan X, Yang J, Jia L, Wu C, Liu H. et al. Enteral virus depletion modulates experimental acute pancreatitis via toll-like receptor 9 signaling. Biochem Pharmacol. 2020;171:113710
60. Zou L, Ke L, Wu C, Tong Z, Li W, Li N. et al. SEW2871 Alleviates the Severity of Caerulein-Induced Acute Pancreatitis in Mice. Biol Pharm Bull. 2015;38:1012-1019
61. Ramudo L, Yubero S, Manso MA, Vicente S, De Dios I. Signal transduction of MCP-1 expression induced by pancreatitis-associated ascitic fluid in pancreatic acinar cells. J Cell Mol Med. 2009;13:1314-1320
62. Chen H, Tu J, He L, Gao N, Yang W. Mmu_circ_0000037 inhibits the progression of acute pancreatitis by miR-92a-3p/Pias1 axis. Immun Inflamm Dis. 2023;11:e819
63. Zhang Y, Wang Y, Lu M, Qiao X, Sun B, Zhang W. et al. Modular analysis of bioinformatics demonstrates a critical role for NF-κB in macrophage activation. Inflammation. 2014;37:1240-1253
64. Bettaieb A, Xi Y, Hosein E, Coggins N, Bachaalany S, Wiede F. et al. Pancreatic T cell protein-tyrosine phosphatase deficiency ameliorates cerulein-induced acute pancreatitis. Cell Commun Signal. 2014;12:13
65. Zhou Z, Chen Y, Dong W, An R, Liang K, Wang X. Da Cheng Qi Decoction Alleviates Cerulein-Stimulated AR42J Pancreatic Acinar Cell Injury via the JAK2/STAT3 Signaling Pathway. Evid Based Complement Alternat Med. 2021;2021:6657036
66. Zheng C, Tao R, Wang Z, Zhang X, Jiang H, Ji Z. et al. CLDN4 promotes ferroptosis and inflammation involving JAK2/STAT3 pathway in acute pancreatitis. Funct Integr Genomics. 2025;25:184
67. Mateu A, Ramudo L, Manso MA, Closa D, De Dios I. Acinar inflammatory response to lipid derivatives generated in necrotic fat during acute pancreatitis. Biochim Biophys Acta. 2014;1842:1879-1886
68. Gonzalez-Moro I, Olazagoitia-Garmendia A, Colli ML, Cobo-Vuilleumier N, Postler TS, Marselli L. et al. The T1D-associated lncRNA Lnc13 modulates human pancreatic β cell inflammation by allele-specific stabilization of STAT1 mRNA. Proc Natl Acad Sci U S A. 2020;117:9022-9031
69. Moore F, Naamane N, Colli ML, Bouckenooghe T, Ortis F, Gurzov EN. et al. STAT1 is a master regulator of pancreatic {beta}-cell apoptosis and islet inflammation. J Biol Chem. 2011;286:929-941
70. De Groef S, Renmans D, Cai Y, Leuckx G, Roels S, Staels W. et al. STAT3 modulates β-cell cycling in injured mouse pancreas and protects against DNA damage. Cell Death Dis. 2016;7:e2272
71. Liu J-Y, Zhang Y-C, Xie R-R, Song L-N, Yang W-L, Xin Z. et al. Nifuroxazide improves insulin secretion and attenuates high glucose-induced inflammation and apoptosis in INS-1 cells. Eur J Pharmacol. 2021;899:174042
72. Li Y, Duan B, Li Y, Yu S, Wang Y. The isoflavonoid calycosin inhibits inflammation and enhances beta cell function in gestational diabetes mellitus by suppressing RNF38 expression. Immunopharmacol Immunotoxicol. 2020;42:366-372
73. Liu Z, Qi M, Tian S, Yang Q, Liu J, Wang S. et al. Ubiquitin-Specific Protease 25 Aggravates Acute Pancreatitis and Acute Pancreatitis-Related Multiple Organ Injury by Destroying Tight Junctions Through Activation of The STAT3 Pathway. Front Cell Dev Biol. 2021;9:806850
74. Rius-Pérez S, Torres-Cuevas I, Monsalve M, Miranda FJ, Pérez S. Impairment of PGC-1 Alpha Up-Regulation Enhances Nitrosative Stress in the Liver during Acute Pancreatitis in Obese Mice. Antioxidants (Basel). 2020;9:887
75. Ge J, Tang L, Mu P, Zhu F, Xie L, Tang Y. Association of ADAM17 Expression Levels in Patients with Interstitial Lung Disease. Immunol Invest. 2020;49:134-145
76. Chen K-L, Lv Z-Y, Yang H-W, Liu Y, Long F-W, Zhou B. et al. Effects of Tocilizumab on Experimental Severe Acute Pancreatitis and Associated Acute Lung Injury. Crit Care Med. 2016;44:e664-677
77. Takayanagi T, Forrester SJ, Kawai T, Obama T, Tsuji T, Elliott KJ. et al. Vascular ADAM17 as a Novel Therapeutic Target in Mediating Cardiovascular Hypertrophy and Perivascular Fibrosis Induced by Angiotensin II. Hypertension. 2016;68:949-955
78. Qiao Y-Y, Liu X-Q, Xu C-Q, Zhang Z, Xu H-W. Interleukin-22 ameliorates acute severe pancreatitis-associated lung injury in mice. World J Gastroenterol. 2016;22:5023-5032
79. Bai J, Bai J, Yang M. Interleukin-22 Attenuates Acute Pancreatitis-Associated Intestinal Mucosa Injury in Mice via STAT3 Activation. Gut Liver. 2021;15:771-781
80. Lin R, Chen F, Wen S, Teng T, Pan Y, Huang H. Interleukin-10 attenuates impairment of the blood-brain barrier in a severe acute pancreatitis rat model. J Inflamm (Lond). 2018;15:4
81. Yang X, Geng H, You L, Yuan L, Meng J, Ma Y. et al. Rhein Protects Against Severe Acute Pancreatitis In vitro and In vivo by Regulating the JAK2/STAT3 Pathway. Front Pharmacol. 2022;13:778221
82. Jin W, Shen Y. Da-Cheng-Qi Decoction Alleviates Intestinal Injury in Rats with Severe Acute Pancreatitis by Inhibiting the JAK2-STAT3 Signaling Pathway. Evid Based Complement Alternat Med. 2019;2019:3909468
83. Dong J, Chen X, Song Y, Fei X. Chaiqin Chengqi decoction inhibits inflammatory mediators and attenuates acute pancreatitis through deactivation of janus kinase/signal transducer and activator of transcription signaling pathway. J Tradit Chin Med. 2019;39:166-173
84. Ge P, Luo Y, Zhang J, Liu J, Xu C, Guo H. et al. Mechanism Investigation and Clinical Retrospective Evaluation of Qingyi Granules: Pancreas Cleaner About Ameliorating Severe Acute Pancreatitis with Acute Respiratory Distress Syndrome. Drug Des Devel Ther. 2024;18:2043-2061
85. Yang J, Jiang Y-H, Zhou X, Yao J-Q, Wang Y-Y, Liu J-Q. et al. Material basis and molecular mechanisms of Chaihuang Qingyi Huoxue Granule in the treatment of acute pancreatitis based on network pharmacology and molecular docking-based strategy. Front Immunol. 2024;15:1353695
86. An Y, Tu Z, Wang A, Gou W, Yu H, Wang X. et al. Qingyi decoction and its active ingredients ameliorate acute pancreatitis by regulating acinar cells and macrophages via NF-κB/NLRP3/Caspase-1 pathways. Phytomedicine. 2025;139:156424
87. Wu L, Li H, Zheng S, Liu X, Cai H, Cai B. Da-Huang-Fu-Zi-Tang attenuates liver injury in rats with severe acute pancreatitis. J Ethnopharmacol. 2013;150:960-966
88. Lu X, Zhan L, Kang X, Liu L, Li Y, Yu J. et al. [Clinical research of Dahuang Fuzi decoction in auxiliary treatment of severe acute pancreatitis: a multi-center observation in 206 patients]. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue. 2010;22:723-728
89. He Y, Hu C, Liu S, Xu M, Liang G, Du D. et al. Anti-Inflammatory Effects and Molecular Mechanisms of Shenmai Injection in Treating Acute Pancreatitis: Network Pharmacology Analysis and Experimental Verification. Drug Des Devel Ther. 2022;16:2479-2495
90. Feng M-C, Luo F, Huang L-J, Li K, Chen Z-M, Li H. et al. Rheum palmatum L. and Salvia miltiorrhiza Bge. Alleviates Acute Pancreatitis by Regulating Th17 Cell Differentiation: An Integrated Network Pharmacology Analysis, Molecular Dynamics Simulation and Experimental Validation. Chin J Integr Med. 2024;30:408-420
91. Pu W-L, Bai R-Y, Zhou K, Peng Y-F, Zhang M-Y, Hottiger MO. et al. Baicalein attenuates pancreatic inflammatory injury through regulating MAPK, STAT 3 and NF-κB activation. Int Immunopharmacol. 2019;72:204-210
92. Yang J, Han F, Wu G, Dong Y, Su H, Xu J. et al. Dysregulated B7H4/JAK2/STAT3 Pathway Involves in Hypertriglyceridemia Acute Pancreatitis and Is Attenuated by Baicalin. Dig Dis Sci. 2023;68:478-486
93. Huang X, Bhugul PA, Fan G, Ye T, Huang S, Dai S. et al. Luteolin inhibits pancreatitis-induced acinar-ductal metaplasia, proliferation and epithelial-mesenchymal transition of acinar cells. Mol Med Rep. 2018;17:3681-3689
94. Yang S, Song Y, Wang Q, Liu Y, Wu Z, Duan X. et al. Daphnetin ameliorates acute lung injury in mice with severe acute pancreatitis by inhibiting the JAK2-STAT3 pathway. Sci Rep. 2021;11:11491
95. Li J, Chen Y-F, Gao L, Li Y-J, Feng D-X. Honokiol Prevents Intestinal Barrier Dysfunction in Mice with Severe Acute Pancreatitis and Inhibits JAK/STAT1 Pathway and Acetylation of HMGB1. Chin J Integr Med. 2024;30:534-542
96. Pan X, Fang X, Wang F, Li H, Niu W, Liang W. et al. Butyrate ameliorates caerulein-induced acute pancreatitis and associated intestinal injury by tissue-specific mechanisms. Br J Pharmacol. 2019;176:4446-4461
97. Zhu S, Zhang C, Weng Q, Ye B. Curcumin protects against acute renal injury by suppressing JAK2/STAT3 pathway in severe acute pancreatitis in rats. Exp Ther Med. 2017;14:1669-1674
98. Ren Z, Li H, Zhang M, Zhao Y, Fang X, Li X. et al. A Novel Derivative of the Natural Product Danshensu Suppresses Inflammatory Responses to Alleviate Caerulein-Induced Acute Pancreatitis. Front Immunol. 2018;9:2513
99. Swetha K, Indumathi MC, Kishan R, Siddappa S, Chen C-H, Marathe GK. Selenium Mitigates Caerulein and LPS-induced Severe Acute Pancreatitis by Inhibiting MAPK, NF-κB, and STAT3 Signaling via the Nrf2/HO-1 Pathway. Biol Trace Elem Res. 2025;203:4728-4750
100. Piao X, Sui X, Liu B, Cui T, Qi Z. Picroside II Improves Severe Acute Pancreatitis-Induced Hepatocellular Injury in Rats by Affecting JAK2/STAT3 Phosphorylation Signaling. Biomed Res Int. 2021;2021:9945149
101. Xia T, Gu Y, Shen J, Zheng L, Xu C. Limonin ameliorates acute pancreatitis by suppressing JAK2/STAT3 signaling pathway. Environ Toxicol. 2021;36:2392-2403
102. Ahn YJ, Lim JW, Kim H. Lutein inhibits IL-6 expression by inducing PPAR-γ activation and SOCS3 expression in cerulein-stimulated pancreatic acinar cells. Mol Med Rep. 2022;26:302
103. Zhang Q, Zhao C, Zhang L, Sun K, Yu L, Wang X. et al. Escin Sodium Improves the Prognosis of Acute Pancreatitis via Promoting Cell Apoptosis by Suppression of the ERK/STAT3 Signaling Pathway. Oxid Med Cell Longev. 2021;2021:9921839
104. Zhang R, Asikaer A, Chen Q, Wang F, Lan J, Liu Y. et al. Network pharmacology and in vitro experimental verification unveil glycyrrhizin from glycyrrhiza glabra alleviates acute pancreatitis via modulation of MAPK and STAT3 signaling pathways. BMC Complement Med Ther. 2024;24:58
105. Cheng L, Chen Q, Pi R, Chen J. A research update on the therapeutic potential of rhein and its derivatives. Eur J Pharmacol. 2021;899:173908
106. Huang W, Liu H, Li Y, Mai G. The Effects of Rhein and Honokiol on Metabolic Profiles in a Mouse Model of Acute Pancreatitis. Med Sci Monit. 2020;26:e925727
107. Zhang D, Li L, Li J, Wei Y, Tang J, Man X. et al. Colchicine improves severe acute pancreatitis-induced acute lung injury by suppressing inflammation, apoptosis and oxidative stress in rats. Biomed Pharmacother. 2022;153:113461
108. Huang H, Wang M, Guo Z, Wu D, Wang H, Jia Y. et al. Rutaecarpine alleviates acute pancreatitis in mice and AR42J cells by suppressing the MAPK and NF-κB signaling pathways via calcitonin gene-related peptide. Phytother Res. 2021;35:6472-6485
109. Wang R, Jiang J, Song P, Peng Q, Jin X, Li B. et al. Kinsenoside alleviates experimental acute pancreatitis by suppressing M1 macrophage polarization via the TLR4/STAT1 signaling pathway. J Ethnopharmacol. 2025;345:119551
110. Jiang J, Wang R, Song P, Peng Q, Jin X, Li B. et al. Lactate Facilitates Pancreatic Repair Following Acute Pancreatitis by Promoting Reparative Macrophage Polarization. Cell Mol Gastroenterol Hepatol. 2025;19:101535
111. Shehzad A, Wahid F, Lee YS. Curcumin in cancer chemoprevention: molecular targets, pharmacokinetics, bioavailability, and clinical trials. Arch Pharm (Weinheim). 2010;343:489-499
112. Fan S, Zhang C, Luo T, Wang J, Tang Y, Chen Z. et al. Limonin: A Review of Its Pharmacology, Toxicity, and Pharmacokinetics. Molecules. 2019;24:3679
113. Li G-M, Chen J-R, Zhang H-Q, Cao X-Y, Sun C, Peng F. et al. Update on Pharmacological Activities, Security, and Pharmacokinetics of Rhein. Evid Based Complement Alternat Med. 2021;2021:4582412
114. Wu J, Song Z, Cai N, Cao N, Wang Q, Xiao X. et al. Pharmacokinetics, tissue distribution and excretion of six bioactive components from total glucosides picrorhizae rhizoma, as simultaneous determined by a UHPLC-MS/MS method. J Chromatogr B Analyt Technol Biomed Life Sci. 2023;1227:123830
115. Fu Y, Yang L, Liu L, Kong L, Sun H, Sun Y. et al. Rhein: An Updated Review Concerning Its Biological Activity, Pharmacokinetics, Structure Optimization, and Future Pharmaceutical Applications. Pharmaceuticals (Basel). 2024;17:1665
116. Estari RK, Dong J, Chan WK, Park MS, Zhou Z. Time effect of rutaecarpine on caffeine pharmacokinetics in rats. Biochem Biophys Rep. 2021;28:101121
117. Cai W, Guan Y, Zhou Y, Wang Y, Ji H, Liu Z. Detection and characterization of the metabolites of rutaecarpine in rats based on ultra-high-performance liquid chromatography with linear ion trap-Orbitrap mass spectrometer. Pharm Biol. 2017;55:294-298
118. Lu L, Xiong Y, Lin Z, Chu X, Panayi AC, Hu Y. et al. Advances in the therapeutic application and pharmacological properties of kinsenoside against inflammation and oxidative stress-induced disorders. Front Pharmacol. 2022;13:1009550
119. Zhang X, Jin M, Liu Y, Chen Q, Si L, Li G. et al. Oral Bioavailability of Kinsenoside in Beagle Dogs Measured by LC-MS/MS: Improvement of Ex Vivo Stability of a Lactone-Containing Compound. Pharmaceutics. 2018;10:87
120. Al Nebaihi HM, Davies NM, Brocks DR. Evaluation of the pharmacokinetics, chylomicron inhibition, and toxicity of colchicine in rats given low doses. Eur J Pharm Biopharm. 2024;202:114392
121. Xu H, Li Y, Paxton JW, Wu Z. Co-Delivery Using pH-Sensitive Liposomes to Pancreatic Cancer Cells: the Effects of Curcumin on Cellular Concentration and Pharmacokinetics of Gemcitabine. Pharm Res. 2021;38:1209-1219
122. Kamil A, Smith DE, Blumberg JB, Astete C, Sabliov C, Oliver Chen C-Y. Bioavailability and biodistribution of nanodelivered lutein. Food Chem. 2016;192:915-923
123. Sun H, Wang J, Lv J. Effects of glycyrrhizin on the pharmacokinetics of paeoniflorin in rats and its potential mechanism. Pharm Biol. 2019;57:550-554
124. Abe K, Higurashi T, Takahashi M, Maeda-Minami A, Kawano Y, Miyazaki S. et al. Concomitant Use of High-dose Methotrexate and Glycyrrhizin Affects Pharmacokinetics of Methotrexate, Resulting in Hepatic Toxicity. In Vivo. 2021;35:2163-2169
125. Yubero S, Ramudo L, Manso MA, De Dios I. Mechanisms of dexamethasone-mediated chemokine down-regulation in mild and severe acute pancreatitis. Biochim Biophys Acta. 2009;1792:1205-1211
126. Han X, Wang Y, Chen H, Zhang J, Xu C, Li J. et al. Enhancement of ICAM-1 via the JAK2/STAT3 signaling pathway in a rat model of severe acute pancreatitis-associated lung injury. Exp Ther Med. 2016;11:788-796
127. Anandam KY, Srinivasan P, Yasujima T, Al-Juburi S, Said HM. Proinflammatory cytokines inhibit thiamin uptake by human and mouse pancreatic acinar cells: involvement of transcriptional mechanism(s). Am J Physiol Gastrointest Liver Physiol. 2021;320:G108-G116
128. Fei S, Li W, Xiang L, Xie X, Zhang L. Protective Effect of Alprostadil on Acute Pancreatitis in Rats via Inhibiting Janus Kinase 2 (JAK2)/STAT3 Signal Transduction Pathway. Med Sci Monit. 2019;25:7694-7701
129. El Morsy EM, Ahmed MAE. Carvedilol attenuates l-arginine induced acute pancreatitis in rats through modulation of oxidative stress and inflammatory mediators. Chem Biol Interact. 2020;327:109181
130. IAP/APA/EPC/IPC/JPS Working Group. Electronic address: info@internationalpancreatology.org, IAP/APA/EPC/IPC/JPS Working Group. International Association of Pancreatology Revised Guidelines on Acute Pancreatitis 2025: Supported and Endorsed by the American Pancreatic Association, European Pancreatic Club, Indian Pancreas Club, and Japan Pancreas Society. Pancreatology. 2025;25:770-814
131. Oiva J, Mustonen H, Kylänpää M-L, Kyhälä L, Kuuliala K, Siitonen S. et al. Acute pancreatitis with organ dysfunction associates with abnormal blood lymphocyte signaling: controlled laboratory study. Crit Care. 2010;14:R207
132. Oiva J, Mustonen H, Kylänpää M-L, Kyhälä L, Alanärä T, Aittomäki S. et al. Patients with acute pancreatitis complicated by organ failure show highly aberrant monocyte signaling profiles assessed by phospho-specific flow cytometry. Crit Care Med. 2010;38:1702-1708
133. Oiva J, Mustonen H, Kylänpää M-L, Kuuliala K, Siitonen S, Kemppainen E. et al. Patients with acute pancreatitis complicated by organ dysfunction show abnormal peripheral blood polymorphonuclear leukocyte signaling. Pancreatology. 2013;13:118-124
134. Murray PJ. Understanding and exploiting the endogenous interleukin-10/STAT3-mediated anti-inflammatory response. Curr Opin Pharmacol. 2006;6:379-386
135. Matsuda N, Hattori Y. Systemic inflammatory response syndrome (SIRS): molecular pathophysiology and gene therapy. J Pharmacol Sci. 2006;101:189-198
136. Turunen A, Kuuliala A, Penttilä A, Kaukonen K-M, Mustonen H, Pettilä V. et al. Time course of signaling profiles of blood leukocytes in acute pancreatitis and sepsis. Scand J Clin Lab Invest. 2020;80:114-123
137. Ezzat GM, Nageb SM, Haredi MA-A, El-Masry MA. Mitochondrial DNA Copy Number, a Damage-Associated Molecular Pattern Molecule, Can Predict Pancreatic Necrosis and Is Correlated with the Severity of Acute Pancreatitis. Dig Dis Sci. 2023;68:4175-4185
138. Wang Y, Li T, Wu B, Liu H, Luo J, Feng D. et al. STAT1 regulates MD-2 expression in monocytes of sepsis via miR-30a. Inflammation. 2014;37:1903-1911
139. Dekkers PE, Juffermans NP, ten Hove T, de Jonge E, van Deventer SJ, van der Poll T. Endotoxin down-regulates monocyte and granulocyte interleukin-6 receptors without influencing gp130 expression in humans. J Infect Dis. 2000;181:1055-1061
140. Kuuliala K, Penttilä AK, Kaukonen K-M, Mustonen H, Kuuliala A, Oiva J. et al. Signalling Profiles of Blood Leucocytes in Sepsis and in Acute Pancreatitis in Relation to Disease Severity. Scand J Immunol. 2018;87:88-98
141. Turunen A, Kuuliala A, Mustonen H, Puolakkainen P, Kylänpää L, Kuuliala K. Blood Leukocyte Signaling Pathways as Predictors of Severity of Acute Pancreatitis. Pancreas. 2021;50:710-718
142. Furukawa R, Kuwatani M, Jiang J-J, Tanaka Y, Hasebe R, Murakami K. et al. GGT1 is a SNP eQTL gene involved in STAT3 activation and associated with the development of Post-ERCP pancreatitis. Sci Rep. 2024;14:12224
143. Abcejo AS, Andrejko KM, Raj NR, Deutschman CS. Failed interleukin-6 signal transduction in murine sepsis: attenuation of hepatic glycoprotein 130 phosphorylation. Crit Care Med. 2009;37:1729-1734
144. Tang C, Hartley GP, Couillault C, Yuan Y, Lin H, Nicholas C. et al. Preclinical study and parallel phase II trial evaluating antisense STAT3 oligonucleotide and checkpoint blockade for advanced pancreatic, non-small cell lung cancer and mismatch repair-deficient colorectal cancer. BMJ Oncol. 2024;3:e000133
145. Würstle S, Hapfelmeier A, Wöhrle C, Dichtl K, Lahmer T, Rasch S. et al. Changes in pathogen spectrum and antimicrobial resistance development in the time-course of acute necrotizing pancreatitis. J Gastroenterol Hepatol. 2019;34:2096-2103
146. Overcast GR, Meibers HE, Eshleman EM, Saha I, Waggoner L, Patel KN. et al. IEC-intrinsic IL-1R signaling holds dual roles in regulating intestinal homeostasis and inflammation. J Exp Med. 2023;220:e20212523
147. Mühl H, Scheiermann P, Bachmann M, Härdle L, Heinrichs A, Pfeilschifter J. IL-22 in tissue-protective therapy. Br J Pharmacol. 2013;169:761-771
148. Khawar MB, Azam F, Sheikh N, Abdul Mujeeb K. How Does Interleukin-22 Mediate Liver Regeneration and Prevent Injury and Fibrosis? J Immunol Res. 2016;2016:2148129
149. Ouyang H, Wei S, Gao B, Qian X, Chen Y, Lu J. et al. Delivery of Synthetic Interleukin-22 mRNA to Hepatocytes via Lipid Nanoparticles Alleviates Liver Injury. Small. 2024;20:e2401499
150. Sun X, Cai W, Li H, Gao C, Ma X, Guo Y. et al. Endothelial-like cancer-associated fibroblasts facilitate pancreatic cancer metastasis via vasculogenic mimicry and paracrine signalling. Gut. 2025;74:1437-1451
151. Li C, Chen Q, Jiang C. Intelligent micelles for on-demand drug delivery targeting extracellular matrix of pancreatic cancer. J Control Release. 2024;373:879-889
152. Wang F, Deng Y, Yu L, Zhou A, Wang J, Jia J. et al. A Macrophage Membrane-Polymer Hybrid Biomimetic Nanoplatform for Therapeutic Delivery of Somatostatin Peptide to Chronic Pancreatitis. Pharmaceutics. 2022;14:2341
153. Jiang X, Zheng Y-W, Bao S, Zhang H, Chen R, Yao Q. et al. Drug discovery and formulation development for acute pancreatitis. Drug Deliv. 2020;27:1562-1580
154. Lowe ME, Goodman MT, Coté GA, Glesby MJ, Haupt M, Schork NJ. et al. Accelerating the Drug Delivery Pipeline for Acute and Chronic Pancreatitis: Summary of the Working Group on Drug Development and Trials in Recurrent Acute Pancreatitis at the National Institute of Diabetes and Digestive and Kidney Diseases Workshop. Pancreas. 2018;47:1193-1199
155. Bynigeri RR, Jakkampudi A, Jangala R, Subramanyam C, Sasikala M, Rao GV. et al. Pancreatic stellate cell: Pandora's box for pancreatic disease biology. World J Gastroenterol. 2017;23:382-405
156. Fang J, Ou Q, Wu B, Li S, Wu M, Qiu J. et al. TcpC Inhibits M1 but Promotes M2 Macrophage Polarization via Regulation of the MAPK/NF-κB and Akt/STAT6 Pathways in Urinary Tract Infection. Cells. 2022;11:2674
157. Wang Y, Xi J, Yi J, Meng C, Zhao X, Sun Z. et al. Brucella induces M1 to M2 polarization of macrophages through STAT6 signaling pathway to promote bacterial intracellular survival. Res Vet Sci. 2022;145:91-101
158. Hill NJ, Lyons PA, Armitage N, Todd JA, Wicker LS, Peterson LB. NOD Idd5 locus controls insulitis and diabetes and overlaps the orthologous CTLA4/IDDM12 and NRAMP1 loci in humans. Diabetes. 2000;49:1744-1747
159. Yao Q, Wei T, Qiu H, Cai Y, Yuan L, Liu X. et al. Epigenetic Effects of Natural Products in Inflammatory Diseases: Recent Findings. Phytother Res. 2025;39:90-137
Author contact
Corresponding authors: Yong Tang, Professor, Ph.D. International Collaborative Centre on Big Science Plan for Purinergic Signalling, Chengdu University of Traditional Chinese Medicine; School of Health and Rehabilitation, Chengdu University of Traditional Chinese Medicine, Chengdu 610075, China. Add: No. 1166 Liutai Avenue, Chengdu, 610075, Sichuan Province, P. R. China. Tel.: +86-028-87665716; E-mail: tangyongedu.cn. Yi-fan Miao, Ph.D. Department of Emergency Medicine, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu, China. Add: No. 39 Shi-er-qiao Road, Chengdu, 610072, Sichuan Province, P. R. China. Tel.: +86-028-87783271; Email: miaoyfedu.cn.

 Global reach, higher impact
Global reach, higher impact