Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(5):1685-1706. doi:10.7150/ijms.130156 This issue Cite
Research Paper
Prognostic Factors for Mortality in Patients with Pyogenic Liver Abscess: A Systematic Review and Meta-Analysis
1. Department of Emergency Medicine, Taipei Veterans General Hospital, Taipei, 112201, Taiwan.
2. Institute of Emergency and Critical Care Medicine, National Yang Ming Chiao Tung University, Taipei, 112304, Taiwan.
3. School of Medicine, National Yang Ming Chiao Tung University, Taipei, 112304, Taiwan.
4. Department of Emergency Medicine, China Medical University Hsinchu Hospital, Hsinchu, 302, Taiwan.
5. Department of Nursing, Taipei Veterans General Hospital, Taipei, 112201, Taiwan.
6. Division of Faculty Development, Department of Medical Education, Taipei Veterans General Hospital, Taipei, 112201, Taiwan.
7. Ph.D. Program of Interdisciplinary Medicine, National Yang Ming Chiao Tung University, Taipei, 112304, Taiwan.
8. Institute of Public Health, National Yang Ming Chiao Tung University, Taipei, 112304, Taiwan.
9. Institute of Health and Welfare Policy, National Yang Ming Chiao Tung University, Taipei, 112304, Taiwan.
10. Kinmen Hospital, Ministry of Health and Welfare, Kinmen, 891, Taiwan.
11. Institute of Clinical Nursing, National Yang Ming Chiao Tung University, Taipei, 112304, Taiwan.
Received 2025-12-18; Accepted 2026-3-18; Published 2026-3-25
Abstract
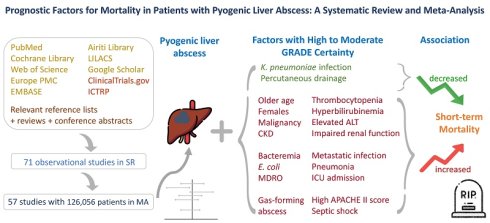
Background: Pyogenic liver abscess (PLA) is an infectious and potentially fatal disease, yet no systematic reviews have comprehensively analyzed the prognostic factors associated with mortality. This study aims to identify prognostic factors associated with mortality in PLA.
Methods: Databases including PubMed, Cochrane Library, Web of Science, Europe PMC, EMBASE, Airiti Library, LILACS, Google Scholar, ClinicalTrials.gov, and ICTRP were searched from inception to July 31, 2023. Reference lists, relevant reviews, and conference abstracts were also screened. Studies investigating predictors of mortality in PLA were included, with short-term mortality as the primary outcome. Pooled estimates were calculated using a random-effects model. Subgroup, meta-regression, and sensitivity analyses were performed.
Results: Seventy-one observational studies were included in this systematic review, and 57 studies involving 126,056 patients contributed to the meta-analyses. Pooled adjusted estimates identified significant mortality predictors: older age, female sex, malignancy, chronic kidney disease, septic shock, higher APACHE II score, gas-formation, metastatic infection, anemia, thrombocytopenia, hypoalbuminemia, hyperbilirubinemia, elevated ALT, impaired renal function, bacteremia, Escherichia coli infection, anaerobic infection, multidrug-resistant organism infection, pneumonia, and ICU admission. Fever, Klebsiella pneumoniae infection, and percutaneous drainage were associated with lower short-term mortality.
Conclusion: In PLA, significant mortality predictors included demographic, comorbidity, clinical, laboratory, radiographic, microbiological, and complication-related factors. Percutaneous drainage was associated with lower short-term mortality in selected patients, although this association should not be interpreted as causal, and treatment decisions should be individualized according to the underlying etiology and clinical context. Future high-quality prospective studies to identify etiology-specific prognostic factors are warranted.
Keywords: pyogenic liver abscess, mortality, prognostic factors, drainage, sepsis
Introduction
Although diagnostic tools and clinical care have improved, pyogenic liver abscess (PLA) remains a potentially life-threatening infectious disease. The literature has reported a wide range of short-term mortality rates, with in-hospital mortality ranging from 0.6% to 11% [1-3].
Numerous studies have identified various prognostic factors for mortality among patients with PLA. Older age has been highlighted as a significant predictor of mortality [4], while some literature has found no significant correlation [5]. Malignancy has been recognized as an important prognostic factor for fatal outcomes, but the estimates vary [6]. Moreover, diabetes mellitus has been characterized as a key comorbidity in infection, but its role in clinical outcomes of PLA remains inconsistent [7]. Additionally, the relationship between causative pathogens, their patterns of drug resistance, and disease prognosis has been explored, with heterogeneous findings observed [8, 9]. While drainage has been acknowledged as a principal treatment modality, the comparative effects of percutaneous and surgical approaches on PLA remain uncertain [10].
Given the limitations of existing evidence and the paucity of high-quality and comprehensive research on prognostic predictors in PLA, this review sought to identify adjusted prognostic factors for mortality across different timeframes.
Methods
This work adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (Table S1) and the Checklist for Critical Appraisal and Data Extraction for Systematic Reviews of Prognostic Factor Studies (CHARMS-PF) guidelines [11, 12]. The study protocol has been registered in INPLASY with the identifier INPLASY202380006.
Search strategy
Two reviewers independently searched eight databases (PubMed, Cochrane Library, Web of Science, Europe PMC, EMBASE, Airiti Library, LILACS, and Google Scholar) and two trial registries (ClinicalTrials.gov and the WHO International Clinical Trials Registry Platform [ICTRP]) from their inception through July 31, 2023. The reference lists of included studies and six related systematic reviews, as well as conference abstracts from three major annual meetings were also screened. The search strategy in detail was shown in Table S2.
Eligibility criteria
Studies fulfilling the following criteria were enrolled: (1) studies of patients with PLA; (2) studies reporting factors associated with mortality and analyzed using logistic regression or Cox proportional hazards regression; and (3) observational designs. Furthermore, duplicate records, non-relevant studies, studies without full-text availability, non-human studies, case reports, studies not addressing the outcome of interest, and those reporting combined outcomes were excluded. There were no restrictions on language, publication period, or publication status. Preprints were eligible if they met the predefined inclusion criteria and provided sufficient methodological details and extractable data.
Study selection
Based on the predefined eligibility criteria, two reviewers independently assessed article titles/abstracts and subsequently the full texts of the retrieved articles. Any discrepancies were resolved in consultation with a third reviewer. Publication timeframe, publication status, and language were not restricted.
Outcomes
The primary outcome was short-term mortality, including in-hospital mortality, intensive care unit (ICU) mortality, 28-day or 30-day mortality, and post-discharge 30-day mortality. Secondary outcomes included other measures of long-term mortality.
Data extraction
Two reviewers independently extracted relevant study data from each article. Any discrepancies were resolved in consultation with a third reviewer. To minimize potential duplication of patient populations, the characteristics of potentially related studies, including disease context, recruitment period, and study institutions, were examined. When potential overlap between studies was suspected for the same prognostic factor, only the study with the largest sample size was selected for inclusion in the meta-analysis. Studies assessing different prognostic factors from the same cohort were retained because they did not lead to duplication within each factor-specific analysis. Study characteristics were collected, and adjusted and unadjusted prognostic factors with their estimates were documented. When multiple models were available, we preferentially extracted adjusted estimates from the most fully adjusted multivariable model that accounted for most of the predefined core confounders—age, sex, chronic kidney disease (CKD), malignancy, and diabetes mellitus.
Risk of bias assessment
The quality assessment of included studies was performed using the Quality in Prognostic Studies (QUIPS) tool by two independent reviewers [13]. Any discrepancies were resolved in consultation with a third reviewer. The six domains for assessment are: (1) bias due to participation; (2) bias due to attrition; (3) bias due to prognostic factor measurement; (4) bias due to outcome measurement; (5) bias due to confounding; and (6) bias in statistical analysis and reporting. In the domain of bias due to confounding, the core confounders—specified as a predefined set of prognostic factors for adjustment—denote five major covariates: age, sex, CKD, malignancy, and diabetes mellitus, which were accounted for in the multivariate analysis [12]. Based on these six domains, an overall high risk of bias was assigned if any domain was rated high risk or if four or more domains were rated moderate risk. For studies without any high-risk domains, the overall risk was classified as moderate when two to three domains were rated moderate risk, whereas those with at most one moderate-risk domain were classified as low risk.
Statistical analysis
All statistical analyses were performed using Review Manager version 5.3.5 (The Nordic Cochrane Centre) and R version 4.3.1 within RStudio. Adjusted estimates of prognostic factors contributing to mortality from each study were pooled as the main results using a random-effects model with the inverse variance method in this meta-analysis. Outcomes were expressed as odds ratios (ORs) or hazard ratios (HRs) with corresponding 95% confidence intervals (CIs), based on the statistics reported in the included studies. For each prognostic factor evaluated using different regression models, separate meta-analyses were conducted for studies reporting ORs and HRs. Conference abstracts lacking sufficient methodological information to allow risk-of-bias assessment were excluded from the quantitative synthesis. Cochran's Q test and the I² statistic were used to examine heterogeneity. Egger's test was used to assess small-study effects only for meta-analyses including at least 10 studies. Funnel plots were also used to assess publication bias by examining the symmetry of the included studies. A p-value of less than 0.05 indicated statistical significance, and all analyses were conducted using two-sided tests.
Subgroup analyses of the primary outcome were conducted. For each prognostic factor, pooled unadjusted estimates were reported in sensitivity analyses when available. Univariate random-effects meta-regression analysis was performed to determine the association between study characteristics and the effect of malignancy on mortality.
Certainty of evidence
The certainty of evidence for each prognostic factor was addressed separately through application of the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) system by two independent reviewers [14, 15]. For this systematic review of prognostic factors, a body of evidence from observational studies began as high certainty initially. The five GRADE domains for rating down certainty of evidence were risk of bias, imprecision, inconsistency, indirectness, and publication bias. Certainty could be rated up for two additional domains: dose-response gradient and large effect. Ultimately, certainty was categorized as high, moderate, low, or very low. Any discrepancies were resolved in consultation with a third reviewer.
Results
Study selection
The PRISMA flowchart of this systematic review is shown in Figure S1. Initially, 3,596 studies were identified from databases and registers, and another 2,363 from reference lists and conference abstracts. After exclusions, 71 studies were included in the systematic review, and 57 studies involving 126,056 individuals contributed to the meta-analyses.
Study characteristics
Table 1 and Table S3 show the characteristics of these 71 observational studies, including 70 cohort studies and 1 case-control study. Their mean or median ages ranged from 50 to 72 years and the majority were male. Sixty-six articles reported primary outcomes with mortality rates ranging from 1% to 34%, and 5 studies described only secondary outcomes and could not be pooled in meta-analyses. Adjusted and unadjusted factors reported across the included 71 articles were provided in Table S4. Of them, 66 articles documented both adjusted and unadjusted estimates of prognostic factors, and 5 presented only unadjusted results. Among the 66 articles employing multivariate analyses, 65% incorporated adjustments for at least two core confounders. Fifty-seven studies contributed to the quantitative synthesis, and all pooled meta-analyses were derived from studies reporting ORs as effect measures. Studies reporting HRs addressed different prognostic factors or timeframes and therefore could not be pooled quantitatively.
Characteristics of the included studies
| Author, year | Journal | Study design | Country | Population | Sample size | Age (mean or median) | Mortality outcome | Mortality rate | Significant adjusted factors identified |
|---|---|---|---|---|---|---|---|---|---|
| Lee 1991 [52] | World J Surg | cohort study | Taiwan | PLA | 73 | 50 | in-hospital mortality | 19% | leukocytosis, albumin, pleural effusion |
| Mischinger 1994 [53] | World J Surg | cohort study | Austria | PLA | 46 | 51 | in-hospital mortality | 17% | serum WBC, hemoglobin, malignancy, APACHE II |
| Chu 1996 [54] | Arch Surg | cohort study | China | PLA | 83 | 72 | in-hospital mortality | 18% | malignancy, hyperbilirubinemia, and elevated APTT |
| Barakate 1999 [55] | Aust NZ J Surg | cohort study | Australia | PLA | 98 | 54 | in-hospital mortality | 8% | NA |
| Lee 2001 [56] | Dig Surg | cohort study | Taiwan | PLA | 133 | 53 | in-hospital mortality | 6% | sepsis |
| Molle 2001 [4] | Gut | cohort study | Denmark | PLA | 665 | NA | 30-day mortality | 28% | liver cirrhosis, CCI, age, sex |
| Pérez 2001 [57] | Am J Surg | cohort study | Spain | PLA | 133 | 60 | 30-day or in-hospital mortality | 14% | biliary origin, shock, multiple abscess, hemoglobin, BUN |
| Wong 2002 [6] | J Gastroenterol Hepatol | cohort study | China | PLA | 80 | 63 | in-hospital mortality | 6% | malignancy |
| Ng 2002 [58] | Aliment Pharmacol Ther | cohort study | China | PLA | 66 | 64 | in-hospital mortality | 3% | NA |
| Chen 2005a [59]* | Wien Klin Wochenschr | cohort study | Taiwan | E. coli PLA | 72 | 66 | in-hospital mortality | 26% | malignancy, hypoalbuminemia, multiple abscess |
| Chen 2005b [60]* | Swiss Med Wkly | cohort study | Taiwan | PLA | 86 | 60 | in-hospital mortality | 10% | NA |
| Jepsen 2005 [61] | Aliment Pharmacol Ther | cohort study | Denmark | PLA | 1,448 | 64 | 30-day mortality | 19% | sex |
| Chen 2006 [62] | Swiss Med Wkly | cohort study | Taiwan | PLA | 225 | 60 | in-hospital mortality | 10% | metastatic infection |
| Hsieh 2006 [63] | Dig Liver Dis | cohort study | Taiwan | PLA | 314 | 57 | in-hospital mortality | 8% | APACHE II score, primary liver cancer history |
| Chen 2007 [64] | Am J Med Sci | cohort study | Taiwan | E. coli and K. pneumoniae PLA | 202 | 58 | in-hospital mortality or 30-day mortality | 10% | APACHE II score, right-lobar abscess involvement, malignancy, uremia, MDRO |
| Ruiz-Hernandez 2007 [65] | Eur J Gastroenterol Hepatol | cohort study | Spain | PLA | 84 | 64 | in-hospital mortality | 19% | septic shock |
| Thomsen 2007 [7] | Clin Infect Dis | case-control study | Denmark | PLA | 1,442 | 64 | 30-day post-discharge mortality | 19% | NA |
| Chen 2008a [66]* | Neth J Med | cohort study | Taiwan | PLA | 253 | 56 | in-hospital mortality | 9% | gas-forming liver abscess, MDRO, anaerobic infection, BUN > 7.86 mmol/l, APACHE II score ≥ 15 |
| Chen 2008b [67]* | Crit Care Med | cohort study | Taiwan | PLA with ICU admission | 72 | 58 | in-hospital mortality | 28% | acute respiratory failure, APACHE II > 16 |
| Lee 2008 [49] | Clin Infect Dis | cohort study | Taiwan | K. pneumoniae PLA | 110 | 62 | in-hospital mortality | 10% | APACHE II score, metastatic infection, septic shock, acute respiratory failure, gas formation on imaging, pigtail drainage |
| Ng 2008 [68] | Surg Pract | cohort study | China | PLA | 143 | 68 | in-hospital mortality | 13% | size of abscess, BUN, APTT |
| Tsai 2008 [33] | Emerg Infect Dis | cohort study | Taiwan | PLA | 29,703 | 59 | in-hospital mortality | 11% | age, DM, abscess drainage, biliary procedure, peptic ulcer, urinary tract infection, renal disease, hypertension, cholelithiasis, hepatobiliary malignancy, other malignancy, pneumonia, active viral hepatitis, heart disease |
| Chen 2009a [32]* | Age Ageing | cohort study | Taiwan | PLA in elderly patients | 339 | 59 | in-hospital mortality | 10% | age, APACHE II score, K. pneumonia infection |
| Chen 2009b [42]* | Am J Surg | cohort study | Taiwan | PLA | 298 | 57 | in-hospital mortality | 10% | APACHE II score at admission, SAPS II score at admission, gas-forming liver abscess, anaerobic infection |
| Chou 2009 [34] | Gastroenterol J Taiwan | cohort study | Taiwan | PLA | 357 | NA | in-hospital mortality | 9% | age, albumin |
| Foo 2010 [69] | Am J Gastroenterol | cohort study | Taiwan | PLA | 377 | 60 | in-hospital mortality | 6% | gas forming abscess, creatinine > 1.3mg/dl |
| Kuo 2010 [70] | Gastroenterol J Taiwan | cohort study | Taiwan | PLA | 120 | NA | in-hospital mortality | 14% | hypoalbuminemia, renal impairment |
| Lou 2010 [71] | Gastroenterol J Taiwan | cohort study | Taiwan | PLA with ICU admission | 35 | 64 | ICU mortality | 34% | APACHE II score, abscess size |
| Meddings 2010 [43] | Am J Gastroenterol | cohort study | USA | PLA | 17,787 | NA | in-hospital mortality | 6% | age, health insurance, hospital characteristics, Elixhauser comorbidity score, cholecystectomy, bacteremia, bacteria classified elsewhere, liver aspiration |
| Chen 2011 [72] | J Gastrointest Surg | cohort study | Taiwan | PLA with underlying malignancy | 85 | 66 | in-hospital mortality | 33% | APACHE II score, multiloculated abscess, polymicrobial infection |
| Kang 2011 [1] | J Chin Med Assoc | cohort study | Taiwan | PLA | 2,319 | 60 | in-hospital mortality | 2% | nephropathy, gastroenterological cancers, acute low respiratory conditions |
| Law 2011 [73] | Eur J Gastroenterol Hepatol | cohort study | China | PLA | 319 | 66 | in-hospital mortality | 16% | malignancy, hypoalbuminemia, DIC, and acute coronary syndrome |
| Law 2012 [10] | World J Gastroenterol | cohort study | China | PLA | 318 | 66 | in-hospital mortality | 16% | malignancy, hypoalbuminemia, DIC, acute coronary syndrome |
| Tian 2012 [9] | Clin Microbiol Infect | cohort study | China | PLA | 357 | 60 | in-hospital mortality | 7% | uremia, gas-forming abscess, MDRO, stay in ICU |
| Kuo 2013 [8] | Am J Emerg Med | cohort study | Taiwan | PLA | 431 | 57 | in-hospital mortality | 15% | higher MEDS scores on admission, malignancy, multiple abscesses, anaerobic infection, TSB > 1.3 mg/dL, serum creatinine > 1.3 mg/dL |
| Law 2013 [74] | Int J Infect Dis | cohort study | China | PLA | 319 | 66 | in-hospital mortality | 16% | age, active hepatic malignancy, serum albumin < 28g/dl, TSB > 30umol/l, DIC |
| Law 2014 [75] | Eur J Gastroenterol Hepatol | cohort study | China | PLA | 359 | 66 | in-hospital mortality | 13% | active hepatic malignancy, DIC, CRP ratio > 0.5 by week 3 |
| Yoon 2014 [76] | Scand J Infect Dis | cohort study | Korea | K. pneumoniae PLA | 161 | 61 | in-hospital mortality | 2% | APACHE II score |
| Chen 2014 [77] | BMC Gastroenterology | cohort study | Taiwan | PLA | 134 | 59 | in-hospital mortality | 4% | NA |
| Hong 2014 [78] | PLoS One | cohort study | Taiwan | PLA in ESRD | 447 | NA | long-term mortality | NA | DM, chronic liver disease |
| Li 2015 [79] | Chin J Crit Care Med | cohort study | China | PLA | 272 | 61 | in-hospital mortality | 5% | septic shock, malignancy |
| Czerwonko 2016 [80] | HPB | cohort study | Argentina | PLA | 142 | 62 | in-hospital mortality | 8% | TSB >5 mg/dl, bilobular involvement |
| Shelat 2016 [81] | Hepatobiliary Pancreat Dis Int | cohort study | Singapore | culture-negative PLA | 264 | 59 | in-hospital or 30-day mortality | 14% | age, fever, INR, BUN |
| Sohn 2016 [82] | Korean J Gastroenterol | cohort study | Korea | PLA | 231 | 64 | in-hospital mortality | 7% | recurrence, anemia, hyperbilirubinemia, thrombocytopenia |
| Mucke 2017 [83] | BMC Infect Dis | cohort study | Germany | PLA | 86 | 62 | in-hospital mortality | 16% | malignancy, TSB |
| Bettinger 2018 [84] | Aliment Pharmacol Ther | cohort study | Germany | PLA | 181 | NA | in-hospital mortality | 22% | Intrahepatic abscess expansion (solitary vs multifocal), bile duct compression, intensive care treatment, treatment with proton pump inhibitors, CCI |
| Chen 2018 [85] | J Int Med Res | cohort study | China | PLA | 178 | 55 | 28-day mortality | 4% | NA |
| Sharma 2018 [86] | Mayo Clin Proc Innov Qual Outcomes | cohort study | USA | PLA | 72 | 63 | 3-month mortality | 17% | biliary disease, liver disease, malignancy, immunosuppression, cardiovascular disease |
| Park 2019 [87] | Eur J Trauma Emerg Surg | cohort study | Korea | PLA | 102 | 65 | in-hospital mortality | 10% | neutrophil-to-lymphocyte ratio |
| Xu 2019a [88]* | Front Endocrinol | cohort study | China | PLA | 240 | 68 | in-hospital mortality | 9% | platelet, GNRI < 90, low T3 syndrome |
| Xu 2019b [89]* | BMC Geriatr | cohort study | China | PLA | 240 | 68 | in-hospital mortality | 9% | platelet, GNRI < 90, PT > 14.8s |
| Dai 2020 [16] | preprint | cohort study | China | PLA | 240 | 68 | in-hospital mortality | 9% | albumin, PT > 14.8s, AST/ALT ratio |
| Lee 2020 [90] | Abdom Radiol | cohort study | Korea | PLA | 219 | 61 | in-hospital mortality | 4% | DM, multiple abscess, internal gas bubble, metastatic infection, septic shock |
| Ruiz-Hernández 2020 [5] | Irish J Med Sci | cohort study | Spain | PLA | 193 | 67 | in-hospital mortality | 16% | E. coli PLA |
| Du 2020 [91] | BMC Infect Dis | cohort study | China | PLA, excluding malignancy | 227 | 56 | 6-month mortality | 11% | DM |
| Yoo 2021 [2] | Liver Int | cohort study | Korea | PLA | 30,690 | 65 | in-hospital mortality | 10% | sex, age, DM, cancer, CKD, ICU admission |
| Faridi 2021 [92] | Int Surg J | cohort study | India | ruptured PLA | 40 | NA | in-hospital or 30-day mortality | 28% | left lobe abscess, shock at presentation, time of presentation, APACHE II score |
| Lee 2021a [93]* | BMC Infect Dis | cohort study | Korea | PLA | 648 | 66 | in-hospital mortality | 2% | maximal abscess diameter, AKI at admission |
| Lee 2021b [94]* | J Clin Med | cohort study | Taiwan | PLA | 324 | 58 | 30-day mortality | 1% | APACHE II score, concomitant infections |
| Losie 2021 [95] | BMC Infect Dis | cohort study | Canada | PLA | 136 | 61 | 30-day mortality | 7% | polymicrobial bacteremia, no drainage performed, history of congestive heart failure, history of liver disease, total bilirubin |
| Yu 2021 [96] | Saudi J Gastroenterol | cohort study | China | PLA | 239 | 64 | in-hospital mortality | 4% | malignancy |
| Große 2021 [97] | Sci Rep | cohort study | Germany | PLA | 133 | 65 | 1-year mortality | 9% | enterococcal infection |
| Chan 2022 [98] | Malays J Med Sci. | cohort study | Singapore | PLA | 213 | 62 | in-hospital mortality | 13% | age |
| Myeong 2022 [99] | J. Infect. Public Health | cohort study | Korea | PLA | 30,690 | 65 | in-hospital mortality | 10% | sex, age, antibiotics, DM, colon cancer, other cancer, CKD, endophthalmitis, ICU admission |
| Park 2022 [46] | Journal of Clinical Medicine | cohort study | Korea | PLA | 833 | 62 | in-hospital mortality | 4% | inadequate antibiotics, use of inotropic agents, ICU admission |
| Wu 2022 [100] | Chin J Crit Care Med | cohort study | China | PLA with sepsis | 120 | 65 | in-hospital mortality | 9% | malignancy, liver failure, septic shock |
| Meister 2022 [101] | Langenbecks Arch Surg | cohort study | Australia | cryptogenic PLA | 98 | 56 | in-hospital mortality | 18% | NA |
| Rossi 2022 [102] | Infection | cohort study | France | PLA | 302 | 62 | 3-month mortality | 11% | CCI, portal thrombosis, MDRO, drainage |
| Jiménez-Romero 2023 [103] | Clin Transplant | cohort study | Spain | PLA following liver transplantation | 289 | NA | in-hospital mortality | 10% | Liver transplantation |
| Li 2023 [104] | Front Surg | cohort study | China | PLA | 458 | 53 | in-hospital mortality | 3% | platelet, hemoglobin, albumin, TSB, creatinine, ARDS, presence of gas |
| Liu 2023 [105] | Kaohsiung J Med Sci | cohort study | China | PLA | 48 | 58 | In-hospital mortality | 15% | NA |
*This asterisk symbol denotes two different publications that share the same author surname and publication year; in this article, they are labeled with “a” and “b” following the author surname and year to distinguish them.
PLA: Pyogenic liver abscess; ESRD: end-stage renal disease; CNS: central nervous system; WBC: white blood cell count; APACHE II: Acute Physiology and Chronic Health Evaluation II; NA: not applicable; CCI: Charlson Comorbidity Index; SAPS II: Simplified Acute Physiology Score II; MDRO: multidrug-resistant organism; BUN: blood urea nitrogen; TSB: total serum bilirubin; GNRI: Geriatric Nutritional Risk Index; E. coli: Escherichia coli; K. pneumoniae: Klebsiella pneumoniae; DM: diabetes mellitus; AST: aspartate aminotransferase; ALT: alanine aminotransferase; CKD: chronic kidney disease; ICU: intensive care unit; APTT: activated partial thromboplastin time; ARDS: acute respiratory distress syndrome; DIC: disseminated intravascular coagulation; MEDS: Mortality in Emergency Department Sepsis score; AKI: acute kidney injury.
Risk of bias assessment
The comprehensive appraisal of risk of bias for individual studies is illustrated in detail in Figure S2. Assessment using the QUIPS tool revealed that 34% of studies had moderate or high risk of bias from participation, 58% from prognostic factor measurement, 37% from confounding, and 80% from statistical analysis and reporting. Only 6% and 15% of studies were judged as having moderate risk exclusively due to attrition and outcome measurement, respectively, with no studies rated as high risk in these two domains. In terms of overall judgment, 21% of studies were rated as low risk of bias, 48% as moderate, and 31% as high.
Prognostic factors for primary outcome
A total of 66 enrolled studies reported predictors of the primary outcome, and 57 of them were meta-analyzed for different factors. Table 2 presents the pooled estimates of all factors for short-term mortality among patients with PLA, and Table 3 provides the GRADE summary of findings. Subgroup analyses are summarized in Table S5, and sensitivity analyses according to adjustment status are detailed in Table S6.
Summary of pooled estimates of all factors for short-term mortality among patients with pyogenic liver abscess
| Factor | Number of studies | Sample size | aOR [95% CI], p value (Inverse variance method and random-effects model) | Heterogeneity (I2, p value) | Egger's test** (p value) |
|---|---|---|---|---|---|
| Age (older vs younger) | 9 | 3,048 | 2.33 [1.29, 4.19], p = 0.005* | 77%, p < 0.001 | NA |
| Age (per 1-year increase) | 4 | 30,559 | 1.02 [1.01, 1.04], p = 0.01* | 27%, p = 0.25 | NA |
| Female sex | 6 | 80,403 | 1.18 [1.04, 1.33], p = 0.01* | 66%, p = 0.01 | NA |
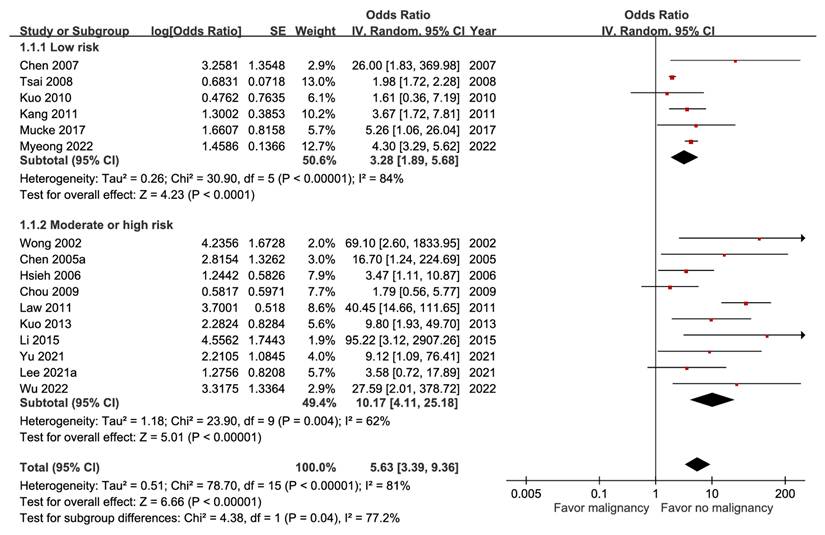
| Malignancy | 16 | 65,972 | 5.63 [3.39, 9.36], p < 0.00001* | 81%, p < 0.00001 | 0.0064 |
| Chronic kidney disease | 8 | 64,077 | 2.41 [1.42, 4.07], p = 0.001* | 93%, p < 0.00001 | NA |
| Diabetes mellitus | 6 | 62,349 | 1.06 [0.83, 1.36], p = 0.64 | 80%, p = 0.0001 | NA |
| Liver cirrhosis | 2 | 31,355 | 1.95 [0.45, 8.49], p = 0.37 | 89%, p = 0.003 | NA |
| Hypertension | 2 | 30,027 | 0.94 [0.17, 5.23], p = 0.94 | 86%, p = 0.007 | NA |
| Fever | 3 | 880 | 0.29 [0.13, 0.68], p = 0.004* | 0%, p = 0.65 | NA |
| Higher APACHE II score | 4 | 1,026 | 12.42 [5.85, 26.37], p < 0.00001* | 0%, p = 0.88 | NA |
| APACHE II score (per 1-point increase) | 4 | 696 | 1.34 [1.21, 1.48], p < 0.00001* | 0%, p = 0.64 | NA |
| Septic shock | 7 | 2,046 | 9.14 [4.54, 18.42], p < 0.00001* | 0%, p = 0.68 | NA |
| Jaundice | 2 | 734 | 1.08 [0.01, 137.40], p = 0.98 | 66%, p = 0.09 | NA |
| Altered mental status | 2 | 1,025 | 0.85 [0.04, 18.51], p = 0.92 | 46%, p = 0.17 | NA |
| Biliary origin | 5 | 1,202 | 1.57 [0.66, 3.73], p = 0.31 | 46%, p = 0.12 | NA |
| Abscess size (per 1-cm increase) | 3 | 280 | 1.64 [0.77, 3.51], p = 0.20 | 97%, p < 0.00001 | NA |
| Multiple abscesses | 8 | 2,044 | 2.95 [0.89, 9.78], p = 0.08 | 80%, p < 0.00001 | NA |
| Gas-forming abscess | 5 | 1,262 | 10.16 [4.01, 25.71], p < 0.00001* | 15%, p = 0.32 | NA |
| Serum WBC count (per 10⁹/L increase) | 2 | 245 | 1.02 [0.99, 1.05], p = 0.21 | 43%, p = 0.18 | NA |
| Leukocytosis | 4 | 1,218 | 1.87 [0.30, 11.68], p = 0.50 | 77%, p = 0.004 | NA |
| Anemia | 5 | 1,706 | 4.33 [1.05, 17.91], p = 0.04* | 68%, p = 0.01 | NA |
| Thrombocytopenia | 3 | 929 | 4.18 [2.05, 8.50], p < 0.0001* | 0%, p = 0.51 | NA |
| Hypoalbuminemia | 8 | 2,186 | 4.12 [2.60, 6.53], p < 0.00001* | 0%, p = 0.73 | NA |
| Hyperbilirubinemia | 6 | 1,752 | 8.36 [2.35, 29.72], p = 0.001* | 0%, p = 0.43 | NA |
| Elevated ALT | 2 | 1,025 | 5.40 [1.32, 22.19], p = 0.02* | 0%, p = 0.77 | NA |
| Azotemia | 4 | 1,027 | 5.12 [0.84, 31.24], p = 0.08 | 80%, p = 0.002 | NA |
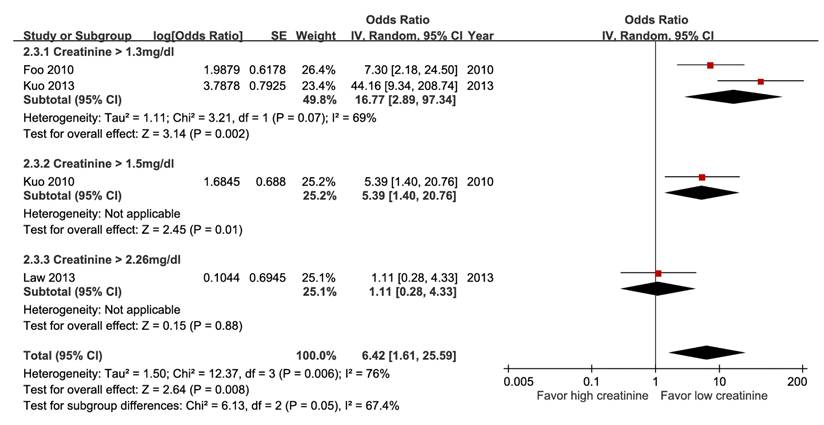
| Impaired renal function | 4 | 1,207 | 6.42 [1.61, 25.59], p = 0.008* | 76%, p = 0.006 | NA |
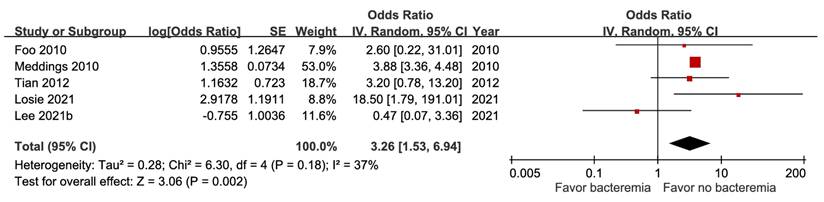
| Bacteremia | 5 | 18,981 | 3.26 [1.53, 6.94], p = 0.002* | 37%, p = 0.18 | NA |
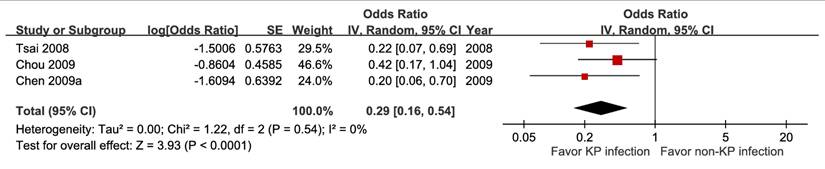
| Klebsiella pneumoniae infection | 3 | 30,399 | 0.29 [0.16, 0.54], p < 0.0001* | 0%, p = 0.54 | NA |
| Escherichia coli infection | 3 | 772 | 2.84 [1.30, 6.21], p = 0.009* | 0%, p = 0.90 | NA |
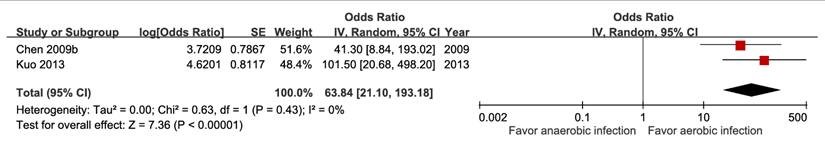
| Anaerobic infection | 2 | 729 | 63.84 [21.10, 193.18], p < 0.00001* | 0%, p = 0.43 | NA |
| Polymicrobial infection | 4 | 1,043 | 2.09 [0.73, 5.97], p = 0.17 | 47%, p = 0.13 | NA |
| Multidrug-resistant organism | 3 | 812 | 8.43 [2.90, 24.53], p < 0.0001* | 0%, p = 0.41 | NA |
| Pneumonia | 2 | 29,823 | 1.52 [1.33, 1.72], p < 0.00001* | 0%, p = 0.96 | NA |
| ICU admission | 5 | 32,380 | 5.12 [3.84, 6.83], p < 0.00001* | 10%, p = 0.35 | NA |
| Metastatic infection | 4 | 878 | 5.34 [2.32, 12.32], p < 0.0001* | 0%, p = 0.46 | NA |
| Ascites | 2 | 497 | 0.84 [0.06, 11.07], p = 0.89 | 54%, p = 0.14 | NA |
| Percutaneous drainage | 4 | 47,736 | 0.48 [0.37, 0.63], p < 0.00001* | 76%, p = 0.007 | NA |
| Surgical drainage | 2 | 18,105 | 0.92 [0.74, 1.15], p = 0.46 | 0%, p = 0.32 | NA |
* Significance with p value less than 0.05
** Egger's test was performed only for meta-analyses including at least 10 studies; otherwise, it was not conducted because of limited statistical power.
CI: confidence interval; NA: Not Applicable; APACHE II: Acute Physiology and Chronic Health Evaluation II; WBC: white blood cell; ALT: alanine aminotransferase; ICU: intensive care unit.
GRADE summary of findings on prognostic factors for short-term mortality among patients with liver abscess.
| Factors | Study number1 | Patient number | Risk of bias2 | Inconsistency3 | Indirectness4 | Imprecision5 | Large effect6 | Other considerations7 | Baseline risk8 | Relative effect (aOR) with 95% CI | Absolute effect with 95% CI9 | GRADE Certainty10 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age(older vs younger) | 9 | 3,048 | 0 | 0 | 0 | 0 | +1 | 0 | 8.5% | 2.33 [1.29, 4.19] | 87 more per 1,000(from 22 more to 195 more) | ⨁⨁⨁⨁High |
| Age (per 1-year increase) | 4 | 30,559 | 0 | 0 | 0 | 0 | 0 | 0 | 8.5% | 1.02 [1.01, 1.04] | 2 more per 1,000(from 1 more to 3 more) | ⨁⨁⨁⨁High |
| Female sex | 6 | 80,403 | 0 | 0 | 0 | -1f | 0 | 0 | 9.0% | 1.18 [1.04, 1.33] | 15 more per 1,000(from 3 more to 26 more) | ⨁⨁⨁Moderate |
| Malignancy | 16 | 65,972 | 0 | 0 | 0 | -2g | +2 | Publication bias (-1)i | 9.1% | 5.63 [3.39, 9.36] | 269 more per 1,000(from 162 more to 393 more) | ⨁⨁⨁Moderate |
| Chronic kidney disease | 8 | 64,077 | 0 | 0 | 0 | 0 | +1 | Dose response (+1)j | 5.8% | 2.41 [1.42, 4.07] | 71 more per 1,000(from 22 more to 142 more) | ⨁⨁⨁⨁High |
| Diabetes mellitus | 6 | 62,349 | 0 | -1c, e | 0 | -2h | 0 | 0 | 13.2% | 1.06 [0.83, 1.36] | 7 more per 1,000(from 20 fewer to 39 more) | ⨁ Very low |
| Liver cirrhosis | 2 | 31,355 | 0 | -1c, d | 0 | -2h | 0 | 0 | 26.9% | 1.95 [0.45, 8.49] | 149 more per 1,000(from 127 fewer to 489 more) | ⨁ Very low |
| Hypertension | 2 | 30,027 | 0 | -1c, d, e | 0 | -2h | 0 | 0 | 10.9% | 0.94 [0.17, 5.23] | 6 fewer per 1,000(from 88 fewer to 281 more) | ⨁ Very low |
| Fever | 3 | 880 | -1a | 0 | 0 | -2g | +1 | 0 | 25.3% | 0.29 [0.13, 0.68] | 164 fewer per 1,000(from 211 fewer to 66 fewer) | ⨁⨁ Low |
| High APACHE II score | 4 | 1,026 | 0 | 0 | 0 | -2g | +2 | Dose response (+1)j | 2.8% | 12.42 [5.85, 26.27] | 234 more per 1,000(from 115 more to 401 more) | ⨁⨁⨁⨁High |
| APACHE II score (per 1-point increase) | 4 | 696 | 0 | 0 | 0 | 0 | 0 | Publication bias (-1)i | 2.8% | 1.34 [1.21, 1.48] | 9 more per 1,000(from 6 more to 13 more) | ⨁⨁⨁Moderate |
| Septic shock | 7 | 2,046 | 0 | 0 | 0 | -2g | +2 | 0 | 4.9% | 9.14 [4.54, 18.42] | 270 more per 1,000(from 140 more to 436 more) | ⨁⨁⨁⨁High |
| Jaundice | 2 | 734 | -1a | -1c, e | 0 | -2h | 0 | 0 | 5.6% | 1.08 [0.01, 137.40] | 4 more per 1,000(from 55 fewer to 834 more) | ⨁ Very low |
| Altered mental status | 2 | 1,025 | -1a | -1c, e | 0 | -2h | 0 | 0 | 3.6% | 0.85 [0.04, 18.51] | 5 fewer per 1,000(from 35 fewer to 373 more) | ⨁ Very low |
| Biliary origin | 5 | 1,202 | -1a | 0 | 0 | -2h | 0 | 0 | 7.3% | 1.57 [0.66, 3.73] | 37 more per 1,000(from 24 fewer to 154 more) | ⨁ Very low |
| Abscess size (per 1-cm increase) | 3 | 280 | 0 | 0 | 0 | -2h | 0 | 0 | 14.3% | 1.64 [0.77, 3.51] | 72 more per 1,000(from 29 fewer to 226 more) | ⨁⨁ Low |
| Multiple abscesses | 8 | 2,044 | 0 | 0 | 0 | -1f | 0 | 0 | 7.3% | 2.95 [0.89, 9.78] | 115 more per 1,000(from 7 fewer to 362 more) | ⨁⨁⨁Moderate |
| Gas-forming abscess | 5 | 1,262 | 0 | 0 | 0 | -2g | +1 | 0 | 7.1% | 10.16 [4.01, 25.71] | 366 more per 1,000(from 164 more to 592 more) | ⨁⨁⨁Moderate |
| Serum WBC count (per 10⁹/L increase) | 2 | 245 | -1a, b | 0 | 0 | -1f | 0 | 0 | 7.5% | 1.02 [0.99, 1.05] | 1 more per 1,000(from 1 fewer to 3 more) | ⨁⨁ Low |
| Leukocytosis | 4 | 1,218 | -1a | -1c, d, e | 0 | -2h | 0 | 0 | 7.5% | 1.87 [0.30, 11.68] | 57 more per 1,000(from 51 fewer to 412 more) | ⨁ Very low |
| Anemia | 5 | 1,746 | -1a | -1c, d, e | 0 | -2g | +1 | 0 | 4.9% | 4.33 [1.05, 17.91] | 133 more per 1,000(from 2 more to 430 more) | ⨁ Very low |
| Thrombocytopenia | 3 | 929 | 0 | 0 | 0 | 0 | +1 | 0 | 1.1% | 4.18 [2.05, 8.50] | 33 more per 1,000(from 11 more to 75 more) | ⨁⨁⨁⨁High |
| Hypoalbuminemia | 8 | 2,186 | 0 | 0 | 0 | -2g | +1 | Publication bias (-1)i | 5.9% | 4.12 [2.60, 6.53] | 145 more per 1,000(from 81 more to 230 more) | ⨁⨁ Low |
| Hyperbilirubinemia | 6 | 1,752 | 0 | 0 | 0 | -2g | 0 | Dose response (+1)j | 3.8% | 8.36 [2.35, 29.72] | 211 more per 1,000(from 47 more to 502 more) | ⨁⨁⨁Moderate |
| Elevated ALT | 2 | 1,025 | -1a | 0 | 0 | -2g | +2 | 0 | 3.6% | 5.40 [1.32, 22.19] | 132 more per 1,000(from 11 more to 418 more) | ⨁⨁⨁Moderate |
| Azotemia | 4 | 1,027 | -1a, b | -1c, d, e | 0 | -2h | 0 | 0 | 6.5% | 5.12 [0.84, 31.24] | 198 more per 1,000(from 10 fewer to 621 more) | ⨁ Very low |
| Impaired renal function | 4 | 1,247 | 0 | 0 | 0 | -2g | +1 | 0 | 7.0% | 6.42 [1.61, 25.59] | 256 more per 1,000(from 38 more to 588 more) | ⨁⨁⨁Moderate |
| Bacteremia | 5 | 18,981 | 0 | 0 | 0 | 0 | +1 | 0 | 2.8% | 3.26 [1.53, 6.94] | 58 more per 1,000(from 14 more to 139 more) | ⨁⨁⨁⨁High |
| Klebsiella pneumoniae infection | 3 | 30,399 | 0 | 0 | 0 | 0 | +1 | 0 | 13.5% | 0.29 [0.16, 0.54] | 91 fewer per 1,000(from 110 fewer to 57 fewer) | ⨁⨁⨁⨁High |
| Escherichia coli infection | 3 | 772 | 0 | 0 | 0 | 0 | +1 | 0 | 4.1% | 2.84 [1.30, 6.21] | 67 more per 1,000(from 12 more to 168 more) | ⨁⨁⨁⨁High |
| Anaerobic infection | 2 | 729 | 0 | 0 | 0 | -2g | 0 | 0 | 9.7% | 63.84 [21.10, 193.18] | 776 more per 1,000(from 597 more to 857 more) | ⨁⨁Low |
| Polymicrobial infection | 4 | 1,043 | -1a | 0 | 0 | -2h | 0 | 0 | 6.5% | 2.09 [0.73, 5.97] | 62 more per 1,000(from 17 fewer to 229 more) | ⨁ Very low |
| Multidrug-resistant organism | 3 | 812 | 0 | 0 | 0 | -2g | +1 | 0 | 4.8% | 8.43 [2.90, 24.53] | 250 more per 1,000(from 79 more to 505 more) | ⨁⨁⨁Moderate |
| Pneumonia | 2 | 29,823 | 0 | 0 | 0 | -1f | 0 | 0 | 2.9% | 1.52 [1.33, 1.72] | 15 more per 1,000(from 9 more to 20 more) | ⨁⨁⨁Moderate |
| ICU admission | 5 | 32,380 | 0 | 0 | 0 | 0 | +2 | 0 | 5.3% | 5.12 [3.84, 6.83] | 170 more per 1,000(from 124 more to 224 more) | ⨁⨁⨁⨁High |
| Metastatic infection | 4 | 878 | 0 | 0 | 0 | -2g | +1 | 0 | 6.2% | 5.34 [2.32, 12.32] | 198 more per 1,000(from 71 more to 385 more) | ⨁⨁⨁Moderate |
| Ascites | 2 | 497 | -1a | -1c, e | 0 | -2h | 0 | 0 | 5.7% | 0.84 [0.06, 11.07] | 9 fewer per 1,000(from 53 fewer to 343 more) | ⨁ Very low |
| Percutaneous drainage | 4 | 47,736 | 0 | 0 | 0 | 0 | +1 | 0 | 7.5% | 0.48 [0.37, 0.63] | 37 fewer per 1,000(from 46 fewer to 26 fewer) | ⨁⨁⨁⨁High |
| Surgical drainage | 2 | 18,105 | 0 | 0 | 0 | -1f | 0 | 0 | 5.6% | 0.92 [0.74, 1.15] | 4 fewer per 1,000(from 14 fewer to 8 more) | ⨁⨁⨁Moderate |
1 Study type was all observational studies.
2 The risk of bias domain was downgraded by one level if:
a More than 60% of the studies had a high overall risk of bias, or fewer than 40% of the studies had a low overall risk of bias.
b A discrepancy between studies at high risk of bias and those at low risk of bias was observed in the subgroup analysis, if available.
3 The inconsistency domain was downgraded by one level if any of the following criteria were met:
c There was an important difference in the point estimates across studies.
d There was limited overlap in their CIs.
e A substantial proportion of point estimates fell on opposite sides of the null effect threshold.
4 The indirectness domain was downgraded by one level if there was a considerable mismatch between the PICO elements in the included studies and the target research question.
5 For the assessment of the imprecision domain, the minimally important difference (MID) was defined as follows:
For categorical variables, the MID for benefit was set at an absolute risk reduction of 10 deaths per 1,000 patients (1.0%), and the MID for harm was set at an absolute risk increase of 10 deaths per 1,000 patients (1.0%).
For continuous variables, including laboratory values or age increments per unit, the MID for benefit was set at an absolute risk reduction of 1 death per 1,000 patients (0.1%), and the MID for harm was set at an absolute risk increase of 1 death per 1,000 patients (0.1%).
The threshold for a large effect of absolute risk for any variable was defined as 20 times the corresponding minimally important difference (MID).
The imprecision domain was downgraded under the following conditions:
f By one level if the CI crossed any MID.
g By two levels if the CI crossed any large effect threshold and the aOR CI ratio was greater than 2.5 for binary outcomes.
h By two levels if the CI crossed both MIDs for benefit and harm.6 The large effect domain was assessed using the pooled point estimate of the aOR for relative risk.:
The domain was upgraded by one level if the estimated aOR was > 2 or < 0.5. The domain was upgraded by two levels if the estimated aOR was > 5 or < 0.2. No or limited upgrading was applied when substantial downgrading was present in the risk of bias, inconsistency, or imprecision domains.
7 Other considerations included the publication bias domain and the dose-response domain:
i The publication bias domain was downgraded by one level if a significant result in Egger's test or an asymmetric funnel plot was observed.
j The dose-response domain was upgraded by one level if a dose-response gradient was identified in subgroup analyses.
8 Baseline risks were calculated based on the number of events and total participants in the control groups (i.e., those without the prognostic factor) of studies examining the factor of interest.
9 Outcome was short-term mortality, including in-hospital mortality, 28-day mortality, or 30-day mortality. Absolute effects were calculated using baseline risks and relative effects via GRADEpro (https://www.gradepro.org/).
10 All certainty ratings began at high certainty because this was a meta-analysis of prognostic factor studies.
aOR: adjusted odds ratio; GRADE: grading of recommendations, assessment, development and evaluation; CI: confidence interval; No: number; APACHE II: Acute Physiology and Chronic Health Evaluation II; WBC: white blood cell; ALT: alanine aminotransferase; ICU: intensive care unit.
Demographic data
For age as a categorical variable, older patients had a higher pooled estimate of short-term mortality compared with younger patients, based on 9 studies enrolling 3,048 individuals (aOR 2.33, 95% CI 1.29-4.19) (Figure S3A). Cochran's Q test for heterogeneity disclosed an I² statistic of 77% with p < 0.001. The subgroup analyses showed no significant differences for age threshold, outcome type, risk of bias, or country, but significant differences for year of publication and sample size. The sensitivity analysis demonstrated that the pooled result from unadjusted estimates (OR 1.97, 95% CI 1.29-3.01) was similar to that from adjusted estimates. The funnel plot showed no obvious asymmetry (Figure S3C). Its GRADE certainty was rated high. For age as a continuous variable (Figure S3B), the aOR per 1-year increase in age was 1.02 (95% CI 1.01-1.04).
A total of six studies with 80,403 participants were included in the meta-analysis of sex as a predictor of mortality. Females had a higher mortality rate than males (aOR 1.18, 95% CI 1.04-1.33, p = 0.01) (Figure S4). Statistical significance of heterogeneity was observed among these trials (I² = 66%, p = 0.01), but the majority of point estimates lay on the same side of the null effect threshold. The GRADE certainty was downgraded to moderate due to imprecision.
Comorbidities
The forest plot of malignancy predicting mortality from 16 studies including 65,972 patients showed an aOR of 5.63 (95% CI, 3.39-9.36) (Figure 1). Univariate random-effects meta-regression for the prognostic effect of malignancy (Table S7) revealed that low risk of bias and younger age were both significant predictors of its association with decreased mortality. The funnel plot (Figure S5) showed obvious asymmetry and the GRADE certainty was moderate.
Forest plot of the association between malignancy (subgroup by risk of bias) and short-term mortality in pyogenic liver abscess.
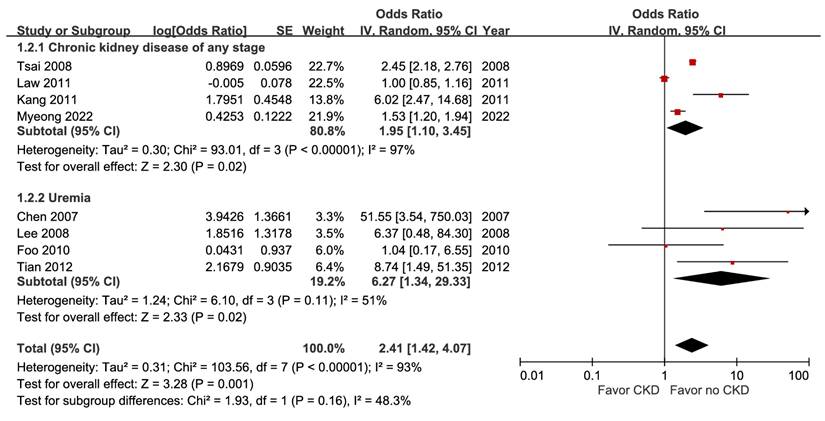
A pooled analysis of eight studies with 64,077 subjects indicated a statistically significant association between CKD and mortality (aOR 2.41, 95% CI 1.42-4.07, p = 0.001). Subgroup analysis identified a disease-severity-related gradient in point estimates (Figure 2), and the certainty of evidence was high.
Forest plot of the association between chronic kidney disease (subgroup by severity) and short-term mortality in pyogenic liver abscess.
Six studies including 62,349 patients examined the association between diabetes mellitus and mortality, and no significant effect was observed (aOR 1.06, 95% CI 0.83-1.36) (Figure S6). The heterogeneity was significant (I2 = 80%, p = 0.0001). The certainty of evidence was very low. There was also no significant association between cirrhosis or hypertension and mortality (Figure S7 and Figure S8).
Clinical presentation and severity score
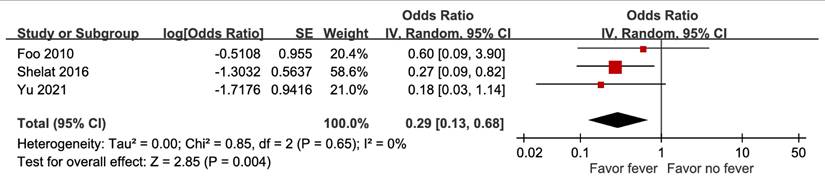
Data from three trials showed that fever was a prognostic factor associated with decreased mortality, with an aOR of 0.29 (95% CI 95% CI 0.13-0.68, p = 0.004) (Figure 3). A high percentage of moderate-to-high risk of bias was noted, and sensitivity analysis based on adjustment revealed a discrepancy. Hence, the certainty of evidence was low.
Forest plot of the association between fever and short-term mortality in pyogenic liver abscess.
Irrespective of whether the Acute Physiology and Chronic Health Evaluation II (APACHE II) score was treated as a categorical or continuous factor, a higher score was associated with increased mortality (aOR 12.42, 95% CI 5.85-26.37; and aOR 1.34, 95% CI 1.21-1.48) (Figure 4 and Figure S9A). As a categorical variable, the cut-off value showed a dose-response gradient relationship (Figure 4), whereas as a continuous variable, the funnel plot illustrated asymmetry (Figure S9B). As a result, their GRADE certainties were rated as high and moderate, respectively.
Forest plot of the association between APACHE II score (subgroup by cut-off value) and short-term mortality in pyogenic liver abscess.
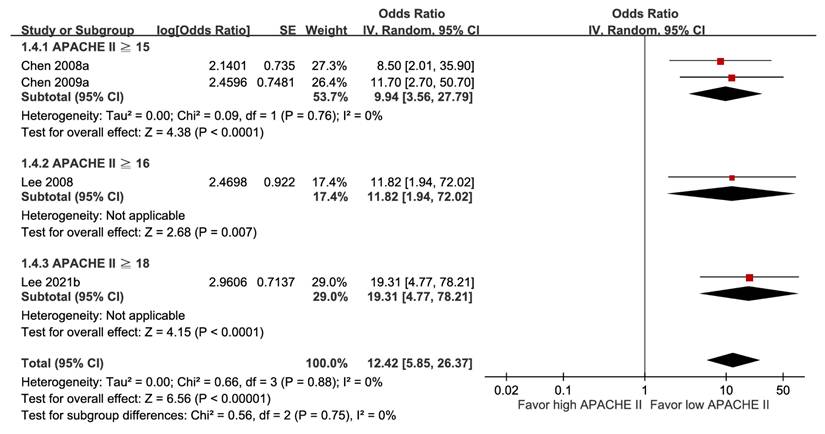
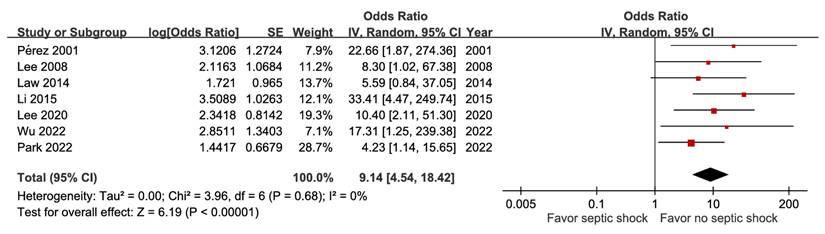
A total of seven studies including 2,046 participants demonstrated a positive relationship between septic shock and poor outcome (aOR 9.14, 95% CI 4.54-18.42), without evidence of heterogeneity (I² = 0%) (Figure 5). The GRADE certainty was rated as high. The combined results showed that jaundice and altered mental status did not correlate with mortality (Figure S10 and Figure S11).
Forest plot of the association between septic shock and short-term mortality in pyogenic liver abscess.
Abscess pattern
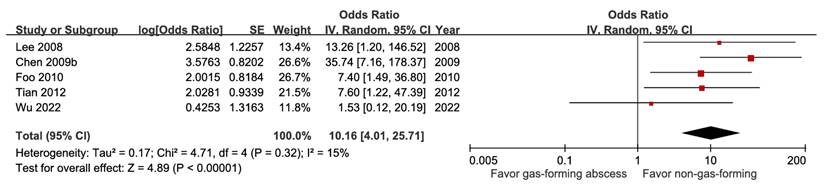
For biliary origin, abscess size, or multiple abscesses, there was no significant effect on prognosis (Figure S12, Figure S13, and Figure S14). However, year of publication was a significant subgroup factor for both abscess size and multiple abscesses. The GRADE certainties for these factors were downgraded to moderate to very low, mainly due to imprecision. Gas-forming abscess was identified as a prognostic factor for mortality, with an aOR of 10.16 (95% CI 4.01-25.7), and this evidence had a moderate degree of certainty (Figure 6).
Forest plot of the association between gas-forming abscess and short-term mortality in pyogenic liver abscess.
Laboratory findings
Anemia and thrombocytopenia were predictive of mortality, with aORs of 4.33 (95% CI 1.05-17.91) and 4.18 (95% CI 2.05-8.50) (Figures S17 and Figure S18), in which a large effect was observed for both GRADE ratings. Nevertheless, no significant association with mortality was observed for serum white blood cell (WBC) count, analyzed either continuously or categorically (Figures S15 and Figure S16).
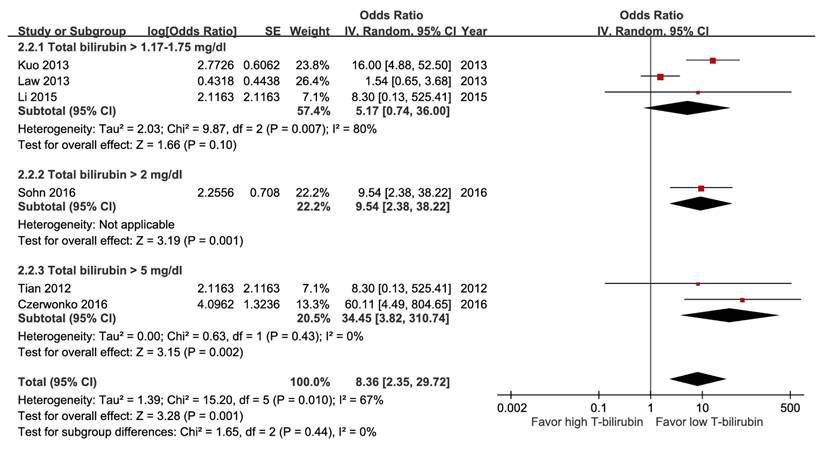
Among these meta-analyses, participants with hypoalbuminemia (Figure S19), hyperbilirubinemia (Figure 7), impaired renal function (Figure 8), elevated alanine aminotransferase (ALT) (Figure 9), or azotemia (Figure S20) had a higher risk of mortality than those without. The pooled estimate for hypoalbuminemia from eight studies was aOR 4.12 (95% CI, 2.60-6.53). In the sensitivity analysis excluding one preprint, the pooled result remained unchanged (aOR 4.05; 95% CI, 2.46-6.67) (Table S8) [16]. However, the funnel plot of the standard error of the log odds ratio versus the odds ratio for hypoalbuminemia illustrated an asymmetrical distribution on visual inspection (Figure S19). Nevertheless, subgroup analysis of hyperbilirubinemia using different cut-off values demonstrated a dose-response gradient (Figure 7).
Forest plot of the association between hyperbilirubinemia (subgroup: cut-off value) and short-term mortality in pyogenic liver abscess.
Forest plot of the association between impaired renal function (subgroup: cut-off value) and short-term mortality in pyogenic liver abscess.
Forest plot of the association between elevated ALT and short-term mortality in pyogenic liver abscess.

Microbiological factors
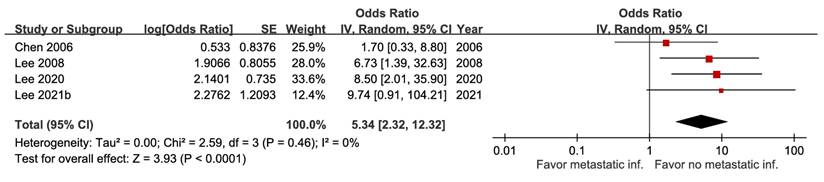
Five studies involving 18,981 patients demonstrated that bacteremia was associated with increased short-term mortality (aOR 3.26, 95% CI 1.53-6.94), with high certainty of evidence (Figure 10). The pooled estimates of 30,399 participants from three studies evaluating Klebsiella pneumoniae infection showed a significant inverse association (aOR 0.29, 95% CI 0.10-0.54) (Figure 11). In contrast, Escherichia coli infection (aOR 2.84, 95% CI 1.30-6.21) and anaerobic infection (aOR 63.84, 95% CI 21.10-193.18) were significantly associated with increased mortality (Figure S21 and Figure 12). Polymicrobial infection was not associated with mortality (aOR 2.09, 95% CI 0.73-5.90) (Figure S22), whereas infection with multidrug-resistant organisms was associated with higher mortality (aOR 8.43, 95% CI 2.90-24.50) (Figure 13).
Forest plot of the association between bacteremia and short-term mortality in pyogenic liver abscess.
Forest plot of the association between Klebsiella pneumoniae infection and short-term mortality in pyogenic liver abscess.
Forest plot of the association between anaerobic infection and short-term mortality in pyogenic liver abscess.
Forest plot of the association between multidrug-resistant organism and short-term mortality in pyogenic liver abscess.

Complications
Patients with pneumonia had a higher likelihood of fatal outcomes (aOR 1.52, 95% CI 1.33-1.72) (Figure S23), whereas both ICU admission and metastatic infection showed stronger associations (aOR 5.12, 95% CI 3.84-6.83, high certainty; aOR 5.34, 95% CI 2.32-12.32, moderate certainty) (Figure 14 and Figure 15). With very low certainty, ascites was not significantly correlated with mortality (Figure S24).
Forest plot of the association between ICU admission and short-term mortality in pyogenic liver abscess.
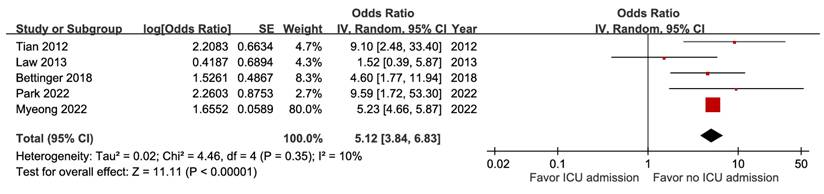
Forest plot of the association between metastatic infection and short-term mortality in pyogenic liver abscess.
Treatment modalities
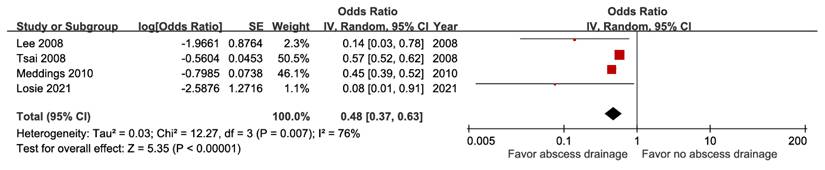
A meta-analysis of four studies including 47,736 subjects demonstrated a lower risk of mortality among patients treated with percutaneous drainage compared with those not treated (aOR 0.48, 95% CI 0.37-0.63) (Figure 16). Nonetheless, the pooled estimate from two studies enrolling 18,105 patients on surgical drainage indicated no significant association (aOR 0.92, 95% CI 0.74-1.15; moderate GRADE certainty) (Figure S25).
Forest plot of the association between percutaneous drainage and short-term mortality in pyogenic liver abscess.
Prognostic factors for secondary outcomes
A total of five eligible studies described prognostic factors for the secondary outcomes, but meta-analysis could not be conducted because of differences in timeframes, statistical methods, or factors analyzed.
Discussion
This comprehensive systematic review of prognostic studies, conducted in accordance with the PRISMA and CHARMS-PF guidelines, enrolled 71 observational studies examining prognostic factors for mortality in patients with PLA identified through eight databases, two trial registries, reference lists, and conference abstracts. With risk of bias assessed using the QUIPS tool, 57 articles involving 126,056 subjects were included in meta-analysis of predictors for short-term mortality, validated by subgroup analyses, sensitivity analyses according to adjustment status, meta-regression, and GRADE evaluation. Demographic characteristics, comorbidities, clinical presentation, abscess patterns, laboratory findings, and microbiological variables were identified as prognostic factors, whereas complications and treatment modalities were identified as factors associated with mortality.
One review article demonstrated the higher mortality rate in patients with PLA of biliary origin than that of PLA of nonbiliary origin, inconsistent with our findings [17]. Many studies have emphasized the importance of specific etiologic factors for clinical outcomes in patients with PLA [18-20]. However, according to methodological guidance for systematic reviews of prognostic factor studies, prognostic associations should ideally be derived from multivariable regression models, such as logistic regression or Cox proportional hazards regression, which allow estimation of the independent prognostic contribution of a factor after adjustment for other established prognostic variables [12]. Under these methodological considerations, the number of eligible studies addressing specific etiological factors was limited. Therefore, the above-mentioned literature on etiologic factors was not included in our study because it did not meet our methodological criteria. During our systematic search, most included studies reported demographic, clinical, laboratory, or microbiological prognostic factors, but did not consistently stratify analyses according to detailed etiological subgroups, particularly hepatic artery injury/ischemia or post-transplant ischemic complications. Consequently, a quantitative meta-analysis focusing on these etiological categories was not feasible in this study. Therefore, this evidence gap and methodologic considerations were acknowledged in this prognostic systematic review and highlight the need for further high-quality studies to address this issue.
In this study, short-term mortality was defined to include closely related early mortality endpoints, including in-hospital mortality, ICU mortality, 28- or 30-day mortality, and post-discharge 30-day mortality. These outcomes are commonly considered comparable short-term outcomes in studies of infectious disease. Research on septic shock disclosed highly comparable pooled estimates for ICU mortality (37.3%), in-hospital mortality (39.0%), and 28/30-day mortality (36.7%), suggesting that these endpoints reflect closely related measures of early mortality [21]. In systematic reviews of sepsis and liver abscess, mortality outcomes are frequently reported using different but closely related short-term endpoints, including in-hospital mortality, 28-day mortality, 30-day mortality, 6-week mortality, and post-discharge mortality within 30 or 90 days [22-24]. Although the exact follow-up duration varies across studies, these endpoints primarily capture deaths occurring within the early clinical course of infection and are therefore regarded as comparable measures of short-term prognosis. This approach follows established methodological guidance for systematic reviews of prognostic factors, which recognizes that outcome definitions may vary across studies and may require harmonization when they represent the same clinical concept [12]. Additionally, subgroup analyses stratified by outcome timeframe were performed (Table S5) for several factors, including female sex, diabetes mellitus, higher APACHE II score, abscess size, and azotemia; most showed no significant differences between subgroups in this study.
For demographic characteristics, older age and female sex were significant prognostic factors for mortality in this research. Irrespective of being treated as a categorical or continuous variable, age was shown to have a strong association with prognosis in both unadjusted and adjusted models. This finding was consistent with previous literature indicating that older age is associated with poor prognosis in sepsis and infectious diseases [25]. Generally speaking, males may exhibit higher mortality and worse prognoses than females in various diseases, potentially because of lower healthcare utilization, inadequate treatment adherence, the effects of female sex hormones, and lifestyle determinants [26]. In contrast, some research still demonstrated more unfavorable outcomes in females than in males, consistent with our findings [27]. Although statistical heterogeneity was noted in our forest plot (Figure S4), the point estimates of the included studies consistently lay in the same direction relative to the threshold of the null effect. Hence, the plausible mechanisms through which female sex serves as a prognostic factor merit more biological and epidemiological exploration.
The significant comorbidities related to patients' fatal outcomes detected in our study were malignancy and CKD, but not diabetes mellitus. Previous literature also listed advanced malignancy as a prognostic factor for worse survival in sepsis [28]. Geng et al. stated that PLA in pancreatic cancer patients with biliary stents requires prolonged antibiotic regimens due to a high rate of polymicrobial infections, immunocompromised status, and delayed abscess resolution [29]. Our meta-regression analysis indicated that studies with low risk of bias and populations of younger mean age may show a decreased predictive effect on mortality. CKD has also been described as an independent predictor of adverse outcomes in literature [30]. Furthermore, our subgroup analysis found that participants with uremia undergoing renal replacement therapy had higher pooled estimates of short-term mortality than those with any stage of CKD (aOR 6.27 vs. 1.95), demonstrating a dose-response gradient that led to an upgraded GRADE level. However, our study did not observe a significant effect of diabetes mellitus on mortality, even with very low certainty of evidence according to GRADE. This finding contrasts with longstanding perspectives and prior research, which have linked diabetes to adverse outcomes among adult sepsis survivors [31]. Emerging evidence highlights that diabetes mellitus status alone does not impair the prognosis of septic patient [24]. Instead, metrics of glycemic control, such as HbA1c, glycemic variability, and antidiabetic medication use, may be more strongly associated with mortality than diabetes status itself [22]. Potential confounding by diabetes treatment effects or glycemic control was not uniformly adjusted for in the included studies; therefore, further research with adequate adjustment is needed in the future.
Potential heterogeneity arising from variations among different regions was also an important consideration in this investigation. We conducted subgroup analyses of prognostic factors according to study country (Asian vs. non-Asian), and no significant subgroup differences were observed (Table S5). Within this prognostic systematic review, the three studies investigating Klebsiella pneumoniae infection were exclusively performed in Taiwan, demonstrating no heterogeneity (I² = 0%; p = 0.54) [32-34]. Therefore, concerns about region-related heterogeneity in this analysis may be reduced. In Southeast Asia, PLA caused by community-acquired, drug-sensitive Klebsiella pneumoniae is associated with a lower fatality rate, and our findings are consistent with this observation [35].
In this study, patients with attenuated febrile responses, higher APACHE II scores, ICU admission, or septic shock were associated with increased mortality. An impaired immune response may lead to delayed recognition of infection by clinicians and subsequent delays in antibiotic therapy; accordingly, afebrile patients have been associated with worse outcomes in an infection cohort [36]. The APACHE II scoring system was found to be closely correlated with subsequent in-hospital death among ICU patients and has also been validated as a good outcome predictor in infectious diseases [37, 38]. In addition, septic shock, characterized by both cellular dysfunction and cardiovascular compromise, contributes to a notably high 30-day mortality of nearly 35% [39]. Furthermore, these identified factors—the APACHE II scoring system, septic shock, and ICU admission—primarily function as markers of illness severity rather than independent prognostic determinants.
In accordance with methodological guidelines, adjusted estimates of prognostic factors associated with mortality from each included study were pooled as the primary results in these meta-analyses. When available, unadjusted effect estimates were reported separately in sensitivity analyses. For example, the sensitivity analysis for fever (Table S6) revealed a discrepancy between adjusted and unadjusted estimates: the pooled result based on unadjusted estimates was statistically significant (OR 0.29, 95% CI 0.13-0.68), whereas the pooled result based on adjusted estimates was not statistically significant (aOR 0.53, 95% CI 0.12-2.29). Discrepancies between adjusted and unadjusted estimates probably indicate the presence of confounding, as prognostic factors are often correlated with other predictors. Therefore, adjusted estimates are generally considered more reliable when evaluating the independent prognostic effect [14]. In addition, according to the QUIPS tool for assessing risk of bias in prognostic factor studies, inadequate adjustment for important confounders may introduce bias due to confounding. Therefore, in the GRADE assessment of the certainty of evidence, the risk-of-bias domain may be downgraded when studies lack adequate adjustment for key confounders [12, 40].
With respect to radiologic features, gas formation, rather than abscess size or multiple abscesses, emerged as a significant prognostic marker in this systematic review. Gas-forming abscesses result from mixed acid fermentation within the abscess cavity, catalyzed by formic hydrogenlyase. Gas formation in liver abscess has been reported to be associated with poorly controlled DM, which appears to provide a favorable environment for the growth of gas-forming microorganisms and is generally linked to a remarkably high mortality rate. Gas formation usually implies sufficient liquefaction of the abscess for drainage, and thus early abscess drainage, in addition to antibiotic administration, remains the primary principle of infection control [41, 42].
In our study, bacteremia was associated with increased mortality. This association likely reflects greater systemic disease severity rather than a direct causal effect of bloodstream infection per se [43]. Additionally, Klebsiella pneumoniae infection was inversely associated with mortality, whereas Escherichia coli and anaerobic infections were associated with increased mortality in this investigation. These pathogen-specific findings should be interpreted with caution. Differences in underlying host factors, abscess etiology (e.g., biliary versus cryptogenic origin), healthcare exposure, regional practice patterns, and detection methods may confound these associations [44, 45]. The extremely large effect estimate observed for anaerobic infection was derived from a limited number of studies and may reflect sparse data bias or residual confounding. Infection with multidrug-resistant organisms was also associated with higher mortality, which may be related to delays in effective antimicrobial therapy and greater underlying comorbidity [46]. Overall, microbiological factors appear to be associated with mortality; however, whether they represent independent causal determinants remains uncertain.
In our investigation, percutaneous drainage was associated with a lower mortality rate. Apart from antibiotic administration, drainage was the primary effective therapeutic intervention for PLA [47]. The published meta-analyses of randomized controlled trials documented a significantly higher treatment success rate with percutaneous catheter drainage compared with percutaneous needle aspiration, particularly for large abscesses, but no difference in mortality [3, 48]. However, in our systematic review, only one of the included studies specified pigtail drainage as the percutaneous drainage modality [49], whereas the other three studies did not clarify the type of percutaneous drainage used. Consequently, percutaneous drainage appears to confer survival benefits based on this meta-analysis of real-world evidence, although the true treatment effect of each specific drainage technique remains uncertain. Additionally, in patients with infected hepatic necrosis due to hepatic artery thrombosis, surgical intervention or liver transplantation may be required, and percutaneous drainage alone may not be sufficient [19, 50]. Huang et al. also emphasized that surgical drainage should be regarded as a complementary rather than competing approach, particularly patients in with unsuccessful percutaneous drainage or those requiring operative management of underlying conditions, including multiple large abscesses, steroid use, or ascites [51]. Therefore, early source control and pathogen identification through appropriate percutaneous drainage may play an important role in the management of many patients with PLA, although treatment should be individualized according to the underlying etiology and clinical context.
This systematic review has several limitations. First, as these meta-analyses focused on prognostic factors for short-term mortality, further investigation of predictors of long-term prognosis may be necessary. Second, many significant factors were described in this research, but the potential inflation of type I error should be taken into consideration. Third, some factors were recognized as having low to very low certainty of evidence, regardless of whether they were significant or not. The presence of substantial heterogeneity, moderate-to-high risk of bias, and incomplete adjustment for key confounders across studies may compromise the reliability of pooled results for several factors. Because the adjusted estimates were derived from multivariable models with different covariate selections across studies, residual confounding cannot be completely excluded. Fourth, PLA represents a heterogeneous disease with diverse etiologies, including biliary obstruction, hepatic artery injury/ischemia, hematogenous dissemination, and cryptogenic infection. Because most included studies did not stratify outcomes according to detailed etiological categories—particularly hepatic artery injury/ischemia—our pooled analyses may reflect averaged associations across heterogeneous subgroups. This limitation may reduce the ability to identify etiology-specific prognostic determinants and should be considered when interpreting the results. Given the above limitations, future high-quality prospective studies with adequate adjustment for key confounders and detailed etiological classification of PLA are warranted to clarify the prognostic impact of different etiologies, strengthen the evidence base, identify etiology-specific prognostic factors, guide individualized interventions, and improve patient outcomes.
Conclusion
In this prognostic systematic review and meta-analysis of observational studies, multiple demographic, clinical, laboratory, radiographic, microbiological, and treatment-related factors were associated with short-term mortality in PLA. With high to moderate certainty of evidence, older age, female sex, malignancy, chronic kidney disease, high APACHE II score, septic shock, gas-forming abscess, thrombocytopenia, hyperbilirubinemia, elevated ALT, impaired renal function, bacteremia, Escherichia coli infection, multidrug-resistant organism infection, metastatic infection, pneumonia, and ICU admission were consistently associated with increased mortality. In contrast, Klebsiella pneumoniae infection and percutaneous drainage were associated with lower mortality. Additional factors — including anemia, anaerobic infection, hypoalbuminemia, and absence of fever — were suggestively associated with increased mortality, although the certainty of evidence was low to very low. These findings should be interpreted as prognostic associations rather than causal relationships, as most included studies were observational and varied in their adjustment for confounding. Several identified factors—particularly disease severity markers and ICU admission—likely reflect underlying disease severity rather than independent, modifiable determinants. Future high-quality prospective studies with standardized adjustment for key confounders are warranted to clarify independent etiology-specific prognostic factors and identify actionable targets for improving clinical outcomes in PLA.
Abbreviations
PLA: pyogenic liver abscess; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; CHARMS-PF: Checklist for Critical Appraisal and Data Extraction for Systematic Reviews of Prognostic Factor Studies; ICTRP: International Clinical Trials Registry Platform; ICU: intensive care unit; QUIPS: Quality in Prognostic Studies; CKD: chronic kidney disease; OR: odds ratio; aOR: adjusted odds ratio; HR: hazard ratio; CI: confidence interval; GRADE: Grading of Recommendations, Assessment, Development and Evaluations; APACHE II: Acute Physiology and Chronic Health Evaluation II; ALT: alanine aminotransferase.
Supplementary Material
Supplementary figures and tables.
Acknowledgements
We express our gratitude to the Department of Emergency Medicine and the Division of Evidence-based Medicine, Department of Medical Education, at Taipei Veterans General Hospital, Taiwan, for their generous support in conducting this study. We also extend our sincere gratitude to Miss Chi-Jung Huang for her dedicated and valuable assistance with this research.
Declaration of generative AI and AI-assisted technologies in the writing process
In preparing this work, the authors used ChatGPT (OpenAI) to assist with readability and language editing. After using this tool, the authors thoroughly reviewed and edited the content and take full responsibility for the final manuscript.
Data availability statement
The authors stated that all data supporting the findings of this research are provided within the article and its supplementary materials.
Author contributions
JWL: conception and study design, protocol development and registration, literature search, study selection, data extraction, risk of bias assessment, evaluation of GRADE evidence certainty, data analysis and interpretation, manuscript drafting and revision. THO: conception and study design, protocol development and registration, literature search, study selection, data extraction, risk of bias assessment, data analysis and interpretation, manuscript drafting and revision. MCS: conception and study design, literature search, study selection, evaluation of GRADE evidence certainty. HMC: data analysis and interpretation. YZL: result validation, manuscript drafting and revision. CKH: data extraction, evaluation of GRADE evidence certainty. TFH: conception and study design, protocol development and registration, risk of bias assessment, data analysis and interpretation, manuscript drafting and revision. All authors approved the final version to be published and confirmed their accountability for the study.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Kang SC, Hwang SJ. Impact of advanced age on inpatients with pyogenic liver abscess in Taiwan: a nationwide claim-based analysis. J Chin Med Assoc. 2011;74:539-43
2. Yoo JJ, Lee TK, Kyoung DS, Park MA, Kim SG, Kim YS. A population-based study of pyogenic liver abscess in Korea: Incidence, mortality and temporal trends during 2007-2017. Liver Int. 2021;41:2747-58
3. Lin JW, Chen CT, Hsieh MS, Lee IH, Yen DH, Cheng HM. et al. Percutaneous catheter drainage versus percutaneous needle aspiration for liver abscess: a systematic review, meta-analysis and trial sequential analysis. BMJ Open. 2023;13:e072736
4. Molle I, Thulstrup AM, Vilstrup H, Sorensen HT. Increased risk and case fatality rate of pyogenic liver abscess in patients with liver cirrhosis: a nationwide study in Denmark. Gut. 2001;48:260-3
5. Ruiz-Hernandez JJ, Conde-Martel A, Serrano-Fuentes M, Hernandez-Meneses M, Merlan-Hermida A, Rodriguez-Perez A. et al. Pyogenic liver abscesses due to Escherichia coli are still related to worse outcomes. Ir J Med Sci. 2020;189:155-61
6. Wong WM, Wong BC, Hui CK, Ng M, Lai KC, Tso WK. et al. Pyogenic liver abscess: retrospective analysis of 80 cases over a 10-year period. J Gastroenterol Hepatol. 2002;17:1001-7
7. Thomsen RW, Jepsen P, Sorensen HT. Diabetes mellitus and pyogenic liver abscess: risk and prognosis. Clin Infect Dis. 2007;44:1194-201
8. Kuo SH, Lee YT, Li CR, Tseng CJ, Chao WN, Wang PH. et al. Mortality in Emergency Department Sepsis score as a prognostic indicator in patients with pyogenic liver abscess. Am J Emerg Med. 2013;31:916-21
9. Tian LT, Yao K, Zhang XY, Zhang ZD, Liang YJ, Yin DL. et al. Liver abscesses in adult patients with and without diabetes mellitus: an analysis of the clinical characteristics, features of the causative pathogens, outcomes and predictors of fatality: a report based on a large population, retrospective study in China. Clin Microbiol Infect. 2012;18:E314-30
10. Law ST, Li KK. Is hepatic neoplasm-related pyogenic liver abscess a distinct clinical entity? World J Gastroenterol. 2012;18:1110-6
11. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Bmj. 2021;372:n71
12. Riley RD, Moons KGM, Snell KIE, Ensor J, Hooft L, Altman DG. et al. A guide to systematic review and meta-analysis of prognostic factor studies. Bmj. 2019;364:k4597
13. Hayden JA, Côté P, Bombardier C. Evaluation of the quality of prognosis studies in systematic reviews. Ann Intern Med. 2006;144:427-37
14. Foroutan F, Guyatt G, Zuk V, Vandvik PO, Alba AC, Mustafa R. et al. GRADE Guidelines 28: Use of GRADE for the assessment of evidence about prognostic factors: rating certainty in identification of groups of patients with different absolute risks. J Clin Epidemiol. 2020;121:62-70
15. Guyatt G, Agoritsas T, Brignardello-Petersen R, Mustafa RA, Rylance J, Foroutan F. et al. Core GRADE 1: overview of the Core GRADE approach. Bmj. 2025;389:e081903
16. Dai H, Xu J. The AST/ALT (De Ritis) Ratio Independently Predicts Adverse Outcomes in Patients with Pyogenic Liver Abscess. Research Square. 2020
17. Shi SH, Zhai ZL, Zheng SS. Pyogenic Liver Abscess of Biliary Origin: The Existing Problems and Their Strategies. Semin Liver Dis. 2018;38:270-83
18. Fernandez H, Gupta A, Anthony T, Kim P, Onaca N, Ruiz R. et al. Impact of hepatic artery thrombosis on patient and allograft survival: Indication for re-transplantation. Transplant International. 2015;28:471
19. Justo I, Jiménez-Romero C, Manrique A, Caso O, Calvo J, Cambra F. et al. Management and Outcome of Liver Abscesses After Liver Transplantation. World J Surg. 2018;42:3341-9
20. Lafont E, Roux O, de Lastours V, Dokmak S, Leflon V, Fantin B. et al. Pyogenic liver abscess in liver transplant recipient: A warning signal for the risk of recurrence and retransplantation. Transpl Infect Dis. 2020;22:e13360
21. Vincent JL, Jones G, David S, Olariu E, Cadwell KK. Frequency and mortality of septic shock in Europe and North America: a systematic review and meta-analysis. Crit Care. 2019;23:196
22. Liang H, Ding X, Li L, Wang T, Kan Q, Wang L. et al. Association of preadmission metformin use and mortality in patients with sepsis and diabetes mellitus: a systematic review and meta-analysis of cohort studies. Crit Care. 2019;23:50
23. Chan KS, Chia CTW, Shelat VG. Demographics, Radiological Findings, and Clinical Outcomes of Klebsiella pneumonia vs. Non-Klebsiella pneumoniae Pyogenic Liver Abscess: A Systematic Review and Meta-Analysis with Trial Sequential Analysis. Pathogens. 2022 11
24. Jiang L, Cheng M. Impact of diabetes mellitus on outcomes of patients with sepsis: an updated systematic review and meta-analysis. Diabetol Metab Syndr. 2022;14:39
25. Ning C, Ouyang H, Shen D, Sun Z, Liu B, Hong X. et al. Prediction of survival in patients with infected pancreatic necrosis: a prospective cohort study. Int J Surg. 2024;110:777-87
26. Espejo-Paeres C, Espliguero RA, Uribarri A, Antón-Huguet B, Romero R, Fernández-Rozas I. et al. Predictors of poor prognosis in healthy, young, individuals with SARS-CoV-2 infections. Clin Microbiol Infect. 2022;28:273-8
27. Chang RK, Chen AY, Klitzner TS. Female sex as a risk factor for in-hospital mortality among children undergoing cardiac surgery. Circulation. 2002;106:1514-22
28. Cuenca JA, Manjappachar NK, Ramírez CM, Hernandez M, Martin P, Gutierrez C. et al. Outcomes and Predictors of 28-Day Mortality in Patients With Solid Tumors and Septic Shock Defined by Third International Consensus Definitions for Sepsis and Septic Shock Criteria. Chest. 2022;162:1063-73
29. Geng D, Lv N, Miao Y. Pyogenic liver abscess following biliary stent placement in pancreatic cancer patients: a retrospective case series. BMC Cancer. 2025;25:965
30. Ravid JD, Kamel MH, Chitalia VC. Uraemic solutes as therapeutic targets in CKD-associated cardiovascular disease. Nat Rev Nephrol. 2021;17:402-16
31. Angriman F, Rosella LC, Lawler PR, Ko DT, Martin CM, Wunsch H. et al. Risk Factors for Major Cardiovascular Events in Adult Sepsis Survivors: A Population-Based Cohort Study. Crit Care Med. 2023;51:471-83
32. Chen SC, Lee YT, Yen CH, Lai KC, Jeng LB, Lin DB. et al. Pyogenic liver abscess in the elderly: clinical features, outcomes and prognostic factors. Age Ageing. 2009;38:271-6 discussion
33. Tsai FC, Huang YT, Chang LY, Wang JT. Pyogenic liver abscess as endemic disease, Taiwan. Emerg Infect Dis. 2008;14:1592-600
34. Chou K-CS, Maw-Soan; Lin, Kwo-Chuan; Soon, Anny C. Hypoalbuminemia is Better Than Hypocholesterolemia for Prognostic Prediction in Pyogenic Liver Abscess. Gastroenterol J Taiwan. 2009;26:13-22
35. Yang CC, Yen CH, Ho MW, Wang JH. Comparison of pyogenic liver abscess caused by non-Klebsiella pneumoniae and Klebsiella pneumoniae. J Microbiol Immunol Infect. 2004;37:176-84
36. Kim W, Kim SM, Yu H, Jang M, Baek SD, Kim SB. Association between afebrile status and in-hospital mortality among adult chronic hemodialysis patients with bacteremia. Hemodial Int. 2018;22:119-25
37. Williams JM, Greenslade JH, Chu K, Brown AF, Lipman J. Severity Scores in Emergency Department Patients with Presumed Infection: A Prospective Validation Study. Crit Care Med. 2016;44:539-47
38. Kuraria V, Paruthy SB, Singh AK, Hameed T, Khera D, Dhawaria M. et al. Evaluation of APACHE II score as predictor of outcome in patients with ruptured liver abscess: prospective study in a tertiary care hospital. Sci Rep. 2025;15:27291
39. Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M. et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). Jama. 2016;315:801-10
40. Hayden JA, van der Windt DA, Cartwright JL, Côté P, Bombardier C. Assessing bias in studies of prognostic factors. Ann Intern Med. 2013;158:280-6
41. Yang CC, Chen CY, Lin XZ, Chang TT, Shin JS, Lin CY. Pyogenic liver abscess in Taiwan: emphasis on gas-forming liver abscess in diabetics. Am J Gastroenterol. 1993;88:1911-5
42. Chen SC, Huang CC, Tsai SJ, Yen CH, Lin DB, Wang PH. et al. Severity of disease as main predictor for mortality in patients with pyogenic liver abscess. Am J Surg. 2009;198:164-72
43. Meddings L, Myers RP, Hubbard J, Shaheen AA, Laupland KB, Dixon E. et al. A population-based study of pyogenic liver abscesses in the United States: incidence, mortality, and temporal trends. Am J Gastroenterol. 2010;105:117-24
44. Olivera MA, Kershenobich D. Pyogenic Liver Abscess. Curr Treat Options Gastroenterol. 1999;2:86-90
45. Chen SC, Tsai SJ, Lee YT, Yen CH, Huang CC, Lin DB. et al. Predictors of mortality in patients with pyogenic liver abscess. Netherlands Journal of Medicine. 2008;66:196-203
46. Park JW, Kim JH, Jung JH, Kim SE, Kim HS, Jeong H. et al. A Multicenter Retrospective Study on Clinical Characteristics and Outcome of Pyogenic Liver Abscess Focusing Multidrug-Resistant Organisms. J Clin Med. 2022;11:1114
47. Siu LK, Yeh KM, Lin JC, Fung CP, Chang FY. Klebsiella pneumoniae liver abscess: a new invasive syndrome. Lancet Infect Dis. 2012;12:881-7
48. Mahmoud A, Abuelazm M, Ahmed AAS, Elshinawy M, Abdelwahab OA, Abdalshafy H. et al. Percutaneous catheter drainage versus needle aspiration for liver abscess management: an updated systematic review, meta-analysis, and meta-regression of randomized controlled trials. Ann Transl Med. 2023;11:190
49. Lee SS, Chen YS, Tsai HC, Wann SR, Lin HH, Huang CK. et al. Predictors of septic metastatic infection and mortality among patients with Klebsiella pneumoniae liver abscess. Clin Infect Dis. 2008;47:642-50
50. Zanus G, Romano M, Finotti M, Dalla Bona E, Sgarabotto D, Bassi D. et al. Liver Retransplantation for Hepatic Abscess Due to Hepatic Artery Thrombosis: A Case Report. Transplant Proc. 2017;49:736-9
51. Huang CJ, Pitt HA, Lipsett PA, Osterman FA Jr, Lillemoe KD, Cameron JL. et al. Pyogenic hepatic abscess. Changing trends over 42 years. Ann Surg. 1996;223:600-7 discussion 7-9
52. Lee KT, Sheen PC, Chen JS, Ker CG. Pyogenic liver abscess: multivariate analysis of risk factors. World J Surg. 1991;15:372-6 discussion 6-7
53. Mischinger HJ, Hauser H, Rabl H, Quehenberger F, Werkgartner G, Rubin R. et al. Pyogenic liver abscess: studies of therapy and analysis of risk factors. World J Surg. 1994;18:852-7 discussion 8
54. Chu KM, Fan ST, Lai EC, Lo CM, Wong J. Pyogenic liver abscess. An audit of experience over the past decade. Arch Surg. 1996;131:148-52
55. Barakate MS, Stephen MS, Waugh RC, Gallagher PJ, Solomon MJ, Storey DW. et al. Pyogenic liver abscess: a review of 10 years' experience in management. Aust N Z J Surg. 1999;69:205-9
56. Lee KT, Wong SR, Sheen PC. Pyogenic liver abscess: an audit of 10 years' experience and analysis of risk factors. Dig Surg. 2001;18:459-65 discussion 65-6
57. Alvarez Perez JA, Gonzalez JJ, Baldonedo RF, Sanz L, Carreno G, Junco A. et al. Clinical course, treatment, and multivariate analysis of risk factors for pyogenic liver abscess. Am J Surg. 2001;181:177-86
58. Ng FH, Wong WM, Wong BC, Kng C, Wong SY, Lai KC. et al. Sequential intravenous/oral antibiotic vs. continuous intravenous antibiotic in the treatment of pyogenic liver abscess. Aliment Pharmacol Ther. 2002;16:1083-90
59. Chen SC, Yen CH, Lai KC, Tsao SM, Cheng KS, Chen CC. et al. Pyogenic liver abscesses with Escherichia coli: etiology, clinical course, outcome, and prognostic factors. Wien Klin Wochenschr. 2005;117:809-15
60. Chen SC, Yen CH, Tsao SM, Huang CC, Chen CC, Lee MC. et al. Comparison of pyogenic liver abscesses of biliary and cryptogenic origin. An eight-year analysis in a University Hospital. Swiss Med Wkly. 2005;135:344-51
61. Jepsen P, Vilstrup H, Schonheyder HC, Sorensen HT. A nationwide study of the incidence and 30-day mortality rate of pyogenic liver abscess in Denmark, 1977-2002. Aliment Pharmacol Ther. 2005;21:1185-8
62. Chen SC, Lee YT, Lai KC, Cheng KS, Jeng LB, Wu WY. et al. Risk factors for developing metastatic infection from pyogenic liver abscesses. Swiss Med Wkly. 2006;136:119-26
63. Hsieh CB, Tzao C, Yu CY, Chen CJ, Chang WK, Chu CH. et al. APACHE II score and primary liver cancer history had risk of hospital mortality in patients with pyogenic liver abscess. Dig Liver Dis. 2006;38:498-502
64. Chen SC, Wu WY, Yeh CH, Lai KC, Cheng KS, Jeng LB. et al. Comparison of Escherichia coli and Klebsiella pneumoniae liver abscesses. Am J Med Sci. 2007;334:97-105
65. Ruiz-Hernandez JJ, Leon-Mazorra M, Conde-Martel A, Marchena-Gomez J, Hemmersbach-Miller M, Betancor-Leon P. Pyogenic liver abscesses: mortality-related factors. Eur J Gastroenterol Hepatol. 2007;19:853-8
66. Chen SC, Tsai SJ, Chen CH, Huang CC, Lin DB, Wang PH. et al. Predictors of mortality in patients with pyogenic liver abscess. Neth J Med. 2008;66:196-203
67. Chen W, Chen CH, Chiu KL, Lai HC, Liao KF, Ho YJ. et al. Clinical outcome and prognostic factors of patients with pyogenic liver abscess requiring intensive care. Crit Care Med. 2008;36:1184-8
68. Ng W-C, Li W-H, Cheung M-T. Audit of management of pyogenic liver abscess in a tertiary referral hospital. Surgical Practice. 2008;12:7-10
69. Foo NP, Chen KT, Lin HJ, Guo HR. Characteristics of pyogenic liver abscess patients with and without diabetes mellitus. Am J Gastroenterol. 2010;105:328-35
70. Kuo M-J, Mo L-R, Chou C-L, Chang K-K, Wang C-H, Lin R-C. et al. Pyogenic Liver Abscess-Comparison between Diabetic Patients and Non-Diabetic Patients. Gastroenterological Journal of Taiwan. 2010;27(4):403-9
71. Lou H-YL, Hong-Ji, Hsieh, Ching-Ruey; Cheng Tiong; Cheng, Yang-Chih; Fang, Sheng-Uei; Liu Jean-Dean; Pan, Shiann; Chang, Chun-Chao. Predictors of Mortality in Patients with Pyogenic Liver Abscess Requiring Intensive Care. Gastroenterol J Taiwan. 2010;27:416-23
72. Chen SC, Lee YT, Tsai SJ, Lai KC, Huang CC, Wang PH. et al. Clinical outcomes and prognostic factors of cancer patients with pyogenic liver abscess. J Gastrointest Surg. 2011;15:2036-43
73. Law ST, Li KK. Is pyogenic liver abscess associated with recurrent pyogenic cholangitis a distinct clinical entity? A retrospective analysis over a 10-year period in a regional hospital. Eur J Gastroenterol Hepatol. 2011;23:770-7
74. Law ST, Li KK. Older age as a poor prognostic sign in patients with pyogenic liver abscess. Int J Infect Dis. 2013;17:e177-84
75. Law ST, Li KK. Role of C-reactive protein in response-guided therapy of pyogenic liver abscess. Eur J Gastroenterol Hepatol. 2014;26:179-86
76. Yoon JH, Kim YJ, Jun YH, Kim SI, Kang JY, Suk KT. et al. Liver abscess due to Klebsiella pneumoniae: risk factors for metastatic infection. Scand J Infect Dis. 2014;46:21-6
77. Chen CH, Wu SS, Chang HC, Chang YJ. Initial presentations and final outcomes of primary pyogenic liver abscess: a cross-sectional study. BMC Gastroenterol. 2014;14:133
78. Hong CS, Chung KM, Huang PC, Wang JJ, Yang CM, Chu CC. et al. Epidemiology and mortality of liver abscess in end-stage renal disease dialysis patients: Taiwan national cohort study. PLoS One. 2014;9:e88078
79. Li MF, Liu GD, Zhao GJ, Hong GL, Qiu QM, Wu B. et al. Clinical characteristics and prognostic factors of pyogenic liver abscess in patients with diabetes mellitus. Chin J Crit Care Med. 2015;8:96-9
80. Czerwonko ME, Huespe P, Bertone S, Pellegrini P, Mazza O, Pekolj J. et al. Pyogenic liver abscess: current status and predictive factors for recurrence and mortality of first episodes. HPB (Oxford). 2016;18:1023-30
81. Shelat VG, Wang Q, Chia CL, Wang Z, Low JK, Woon WW. Patients with culture negative pyogenic liver abscess have the same outcomes compared to those with Klebsiella pneumoniae pyogenic liver abscess. Hepatobiliary Pancreat Dis Int. 2016;15:504-11
82. Sohn SH, Kim KH, Park JH, Kim TN. Predictors of Mortality in Korean Patients with Pyogenic Liver Abscess: A Single Center, Retrospective Study. Korean J Gastroenterol. 2016;67:238-44
83. Mucke MM, Kessel J, Mucke VT, Schwarzkopf K, Hogardt M, Stephan C. et al. The role of Enterococcus spp. and multidrug-resistant bacteria causing pyogenic liver abscesses. BMC Infect Dis. 2017;17:450
84. Bettinger D, Martin D, Rieg S, Schultheiss M, Buettner N, Thimme R. et al. Treatment with proton pump inhibitors is associated with increased mortality in patients with pyogenic liver abscess. Aliment Pharmacol Ther. 2018;47:801-8
85. Chen J, Zhang M, Chen J, Ning Y, Cai X, Zhang L. et al. Cryptogenic and non-cryptogenic liver abscess: A retrospective analysis of 178 cases revealed distinct characteristics. J Int Med Res. 2018;46:3824-36
86. Sharma A, Mukewar S, Mara KC, Dierkhising RA, Kamath PS, Cummins N. Epidemiologic Factors, Clinical Presentation, Causes, and Outcomes of Liver Abscess: A 35-Year Olmsted County Study. Mayo Clin Proc Innov Qual Outcomes. 2018;2:16-25
87. Park KS, Lee SH, Yun SJ, Ryu S, Kim K. Neutrophil-to-lymphocyte ratio as a feasible prognostic marker for pyogenic liver abscess in the emergency department. Eur J Trauma Emerg Surg. 2019;45:343-51
88. Xu J, Wang L. Low T3 Syndrome as a Predictor of Poor Prognosis in Patients with Pyogenic Liver Abscess. Front Endocrinol (Lausanne). 2019;10:541
89. Xu J, Zhou X, Zheng C. The geriatric nutritional risk index independently predicts adverse outcomes in patients with pyogenic liver abscess. BMC Geriatr. 2019;19:14
90. Lee JH, Jang YR, Ahn SJ, Choi SJ, Kim HS. A retrospective study of pyogenic liver abscess caused primarily by Klebsiella pneumoniae vs. non-Klebsiella pneumoniae: CT and clinical differentiation. Abdom Radiol (NY). 2020;45:2669-79
91. Du Z, Zhou X, Zhao J, Bi J, Ren Y, Zhang J. et al. Effect of diabetes mellitus on short-term prognosis of 227 pyogenic liver abscess patients after hospitalization. BMC Infect Dis. 2020;20:145
92. Faridi SA, Harris SH, Alvi Y. Evaluation of predictors of mortality in patients of ruptured liver abscess: a prospective study. International Surgery Journal. 2021 8
93. Lee CH, Jo HG, Cho EY, Song JS, Jung GM, Cho YK. et al. Maximal diameter of liver abscess independently predicts prolonged hospitalization and poor prognosis in patients with pyogenic liver abscess. BMC Infect Dis. 2021;21:171
94. Lee YT, Wang CC, Li CF, Chen HY, Liao HH, Lin CC. Utility of Acute Physiology and Chronic Health Evaluation (APACHE II) in Predicting Mortality in Patients with Pyogenic Liver Abscess: A Retrospective Study. J Clin Med. 2021 10
95. Losie JA, Lam JC, Gregson DB, Parkins MD. Epidemiology and risk factors for pyogenic liver abscess in the Calgary Health Zone revisited: a population-based study. BMC Infect Dis. 2021;21:939
96. Yu J, Ma J, Wang H, Shi Y, He S, Chen Y. et al. Clinical comparison of febrile and afebrile patients with pyogenic liver abscess: A two-centre retrospective study. Saudi J Gastroenterol. 2021;27:370-5
97. Große K, Ohm D, Würstle S, Brozat JF, Schmid RM, Trautwein C. et al. Clinical characteristics and outcome of patients with enterococcal liver abscess. Sci Rep. 2021;11:22265
98. Chan KS, Junnarkar SP, Low JK, Huey CWT, Shelat VG. Aging is Associated with Prolonged Hospitalisation Stay in Pyogenic Liver Abscess-A 1:1 Propensity Score Matched Study in Elderly Versus Non-Elderly Patients. Malays J Med Sci. 2022;29:59-73
99. Myeong JH, Kyoung DS, Park MA, Kim SG, Kim YS, Yoo JJ. et al. Anaerobe coverage is important for the prognosis of pyogenic liver abscess: A population-based study in Korea. J Infect Public Health. 2022;15:425-32
100. Wu ZZ, Chen Y, Lin HN, Sun HY, Pan JY. Risk factors and prognosis analysis of sepsis caused by pyogenic liver abscess. Chin J Crit Care Med. 2022;42:957-62
101. Meister P, Irmer H, Paul A, Hoyer DP. Therapy of pyogenic liver abscess with a primarily unknown cause. Langenbecks Arch Surg. 2022;407:2415-22
102. Rossi G, Nguyen Y, Lafont E, Rossi B, Canouï E, Roux O. et al. Large retrospective study analysing predictive factors of primary treatment failure, recurrence and death in pyogenic liver abscesses. Infection. 2022;50:1205-15
103. Jimenez-Romero C, Marcacuzco A, Caso O, Lechuga I, Manrique A, Garcia-Sesma A. et al. Pyogenic liver abscesses in liver transplant recipients versus non-transplant population. Outcome and risk factors of patient survival. Clin Transplant. 2023;37:e14966
104. Li S-Z, Liu S-H, Hao M, Yu T, Hu S, Liu L. et al. Thrombocytopenia as an important determinant of poor prognosis in patients with pyogenic liver abscess: a retrospective case series. Frontiers in surgery. 2023 p. 1192523
105. Liu YM, Ren YQ, Song SL, Zheng CS. Pyogenic liver abscess in non-liver cancer patients and liver cancer patients treated with TACE: Etiological characteristics, treatment, and outcome analysis. Kaohsiung J Med Sci. 2023;39:87-94
Author contact
Corresponding author: Teh-Fu Hsu, Department of Emergency Medicine, Taipei Veterans General Hospital. No. 201, Sec. 2, Shipai Rd., Beitou Dist., Taipei City 112201, Taiwan. E-mail: tfhsugov.tw. Telephone: +886-2-2875-7628. Fax: +886-2-2873-8013.

 Global reach, higher impact
Global reach, higher impact