Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(5):1588-1594. doi:10.7150/ijms.130315 This issue Cite
Research Paper
Changes of the QRS-T angle in Patients Undergoing Transcatheter Aortic Valve Implantation
1. Department of Cardiology and Angiology, Contilia Heart and Vascular Center, Elisabeth-Krankenhaus Essen, Essen, Germany.
2. Ruhr University Bochum, Bochum, Germany.
3. B. Braun Ambulantes Herzzentrum Kassel, Kassel, Germany.
4. Department of Cardiology and Pulmonology, University Medical Center Goettingen, Goettingen, Germany.
Received 2025-12-21; Accepted 2026-3-4; Published 2026-3-17
Abstract
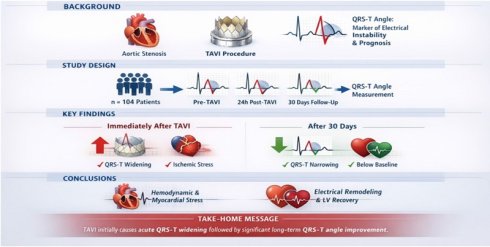
Objective: This study aimed to investigate changes in the frontal QRST angle in patients who underwent transcatheter aortic valve implantation (TAVI).
Background: The QRS-T angle is a prognostic marker in several clinical settings. The impact of TAVI on the QRS-T angle, especially in the long term, has not been investigated thus far.
Methods: A total of 104 patients undergoing transcatheter aortic valve replacement due to severe aortic stenosis underwent a standardized digital 12-lead ECG for the calculation of the QRS-T angle before and 24 h after the intervention, as well as 30 days after the intervention.
Results: There was a significant and dynamic change in the QRS-T angle during the TAVI procedure. An initial increase in the number of patients with a wider QRS-T angle (>90°) was notable immediately after valve replacement. After 30 days, there was a significant decrease in the number of patients with a wide QRS T-angle.
Conclusion: The QRS-T angle widens early after transcatheter aortic valve replacement and improves below the baseline during a 3 months follow-up. This could be related to the cardiac
ischemic stress during valve intervention. In contrast, QRS-T angle narrowing during follow-up might reflect the normalization of left ventricular hemodynamics and prognostic improvement.
Keywords: QRS-T angle, aortic valve stenosis, transcatheter aortic valve replacement
Introduction
Aortic valve stenosis (AVS) is the most common valvular heart disease in Europe and North America that requires surgical or catheter intervention [1, 2, 3]. Prognosis is poor in untreated severe aortic stenosis as soon as symptoms (angina, dyspnea, syncope) develop [4,5,6]. Surgical aortic valve replacement (SAVR) has been shown to improve symptoms and survival and restore life expectancy to that of the general population [7,9]. However, advanced age and comorbidities leave many patients untreated [8]. During the last decade, transcatheter aortic valve implantation (TAVI) has become an established alternative to surgery in elderly patients with high or intermediate surgical risks [10,11].
Individual risk assessment and joint decision-making (heart team approach) are crucial for adequate treatment in patients with AVS. Apart from imaging, a simple derived parameter from routine ECG would be extremely helpful in risk stratification of patients following TAVI. The frontal QRS-T angle, which represents the balance of depolarization to repolarization and, thus, electrical instability, is a promising risk marker [12]. The QRS-T angle has a strong prognostic impact in various clinical settings [14-17], and a poor prognosis in patients with a wide QRS-T angle undergoing SAVR has been recently demonstrated [13]. In a previous study, we identified a correlation between the extent of myocardial damage caused by ST-elevation myocardial infarction (STEMI), as quantified by late gadolinium enhancement (LGE) on cardiac magnetic resonance imaging (MRI), and a larger QRS-T angle (≥ 90°) [18]. In addition, the extent of myocardial fibrosis in patients with hypertrophic cardiomyopathy is associated with a wide QRS-T angle and can therefore be considered a prognostic predictor of mortality [19].
The serial changes in the QRS-T angle and the impact of valve intervention on the QRS-T angle have not yet been investigated.
This study aimed to examine the long-term changes in the frontal QRST-angle in patients undergoing transcatheter aortic valve replacement.
Methods
In this retrospective trial, patients who underwent transcatheter aortic valve replacement (from January 2014 to December 2014) for symptomatic severe aortic valve stenosis at the Department of Cardiology and Angiology of the Elisabeth Hospital Essen were enrolled. In general, the indications for TAVI followed the current guidelines, and individual decisions were based on the heart team approach for every patient [20]. Demographic, imaging, and procedural data were recorded. Routine 12-lead digital ECG was obtained 24 h before, 24 h after TAVI, and at 1-month follow-up. The 12-lead digital ECGs using a Schiller Cardiovit AT 102 plus® were recorded at a speed of 50 mm/s with 10 mm/mV for the limb and precordial leads. The computerized values of the QRS and T-wave axes were automatically provided by the Schiller AT 102 plus® software. The frontal QRS-T angle was calculated as the absolute difference between the frontal QRS- and frontal T-wave axes and expressed as absolute values (Oehler). Due to the retrospective nature of this study informed consent was waived by the ethics committee.
Statistical analysis
Continuous variables are presented as mean ± standard deviation and were compared using two-way ANOVA. Categorical variables are presented as numbers and percentages and were compared using the chi-square test. Differences between groups were analyzed using a two-way ANOVA. All variables with a univariate p-value < 0.05 were included in the final multivariate model. The results of the binary logistic regression model are presented in the same way as the variables in the univariate analysis. All reported p-values were two-sided, and a p-value <0.05 was considered statistically significant. All statistical analyses were performed using IBM SPSS Statistics version 26.0 (IBM Corp., Armonk, N.Y., USA).
Results
A total of 104 patients (82.5±6.19 years, 57,69% female) with symptomatic high-degree aortic valve stenosis (mean maximum velocity 4.19±0.68, mean transvalvular gradient 44.21±13.64, mean aortic valve area 0.74±0.20) underwent transcatheter aortic valve replacement. The median ejection fraction was 52.27 ± 11.73. The rate of syncope prior to TAVI was 14%. The mean (± SD) log. EuroSCORE was 22.38±11.29. (Table 1).
Demographic, imaging, angiographic and QRS-T angle data at baseline
| n = 104 | |
|---|---|
| Gender w (%) | 57.7 |
| Age (years) | 82.5 + 6.2 |
| BMI (kg/m²) | 26.8 + 4.8 |
| CAD (%) | 52.9 |
| 1 vessel | 11.5 |
| 2 vessels | 7.7 |
| 3 vessels | 33.7 |
| In % | |
| prev MI | 5.8 |
| prev CABG | 22.1 |
| prev PCI | 26.9 |
| In % | |
| Diabetes | 31.7 |
| Hypertension | 98.1 |
| Hypercholesterolemia | 68.3 |
| Smoker | 6.7 |
| Family history | 5.8 |
| Ckd | 28.9 |
| Carotid disease | 14.4 |
| Periph. Disease | 18.3 |
| Af | 9.6 |
| Pulmonary disease | 24.1 |
| Cancer | 9.6 |
| Log Euroscore-I in % | 22.4 + 11.3 |
| Syncope in % | 14.4 |
| CCS (I-IV) | 1.1 + 0.9 |
| NYHA (I-IV) | 3.0 + 0.6 |
| QRS-T angle in degree ° | |
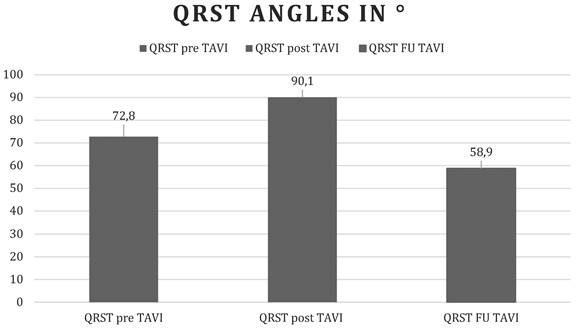
| pre TAVI | 72.8 + 49.5 |
| post TAVI | 90.1 + 49.6 |
| FU TAVI | 58.9 + 47.4 |
| Lab results | |
| Creatinine (mg/dl) | 1.2 + 0.7 |
| Hb (g/dl) | 12.0 + 1.6 |
| TnT (ng/l) | 0.1 + 0.1 |
| Creatinine post (mg/dl) | 1.3 + 0.8 |
| Hb post (g/dl) | 9.8 + 1.5 |
| TnT max post (ng/l) | 0.2 + 0.2 |
| EF pre in % | 52.2 + 11.7 |
| Left atrial diameter (mm) | 43.3 + 6.0 |
| Left ventricular enddiastolic diameter (mm) | 48.1 + 8.0 |
| Max V (m/s) | 4.2 + 0.7 |
| Max grad (mmHg) | 71.0 + 24.0 |
| Pmean pre (mmHg) | 44.2 + 13.6 |
| AVA pre (cm²) | 0.7 + 0.20 |
| EF post in % | 53.0 + 9.4 |
| Velocity (m/s) | 2.2 + 0.6 |
| Pmean post (mmHg) | 12.3 + 5.3 |
| Pmax post (mmHg) | 23.3 + 9.1 |
| Valve dimensions (mm) | 26.5 + 2.1 |
| Access transfemoral (%) | 99.0 |
| Balloon size in mm | 22.1 + 4.8 |
| P-P Gradient pre (mmHg) | 45.5 + 19.6 |
| P-P Gradient post (mmHg) | 6.2 + 5.2 |
a.) QRS T angle pre TAVI:
Patients with a QRS-T angle ≥ 90° (n = 36; mean angle 130.44° ± 28.70°) at baseline experienced episodes of syncope with significantly greater frequency (p = 0.0022) than those with a QRS-T angle < 90° (n = 68). Furthermore, patients with a QRS-T angle < 90° showed a higher rate of pulmonary disease (p = 0.02) and cancer (p = 0.04). (Table 2).
QSR-T angle and clinical data at baseline stratified by QRS-T angle.
| QRS-T ≥ 90 pre TAVI | QRST < 90 pre TAVI | p value | |
|---|---|---|---|
| (n = 36) | (n = 68) | ||
| Gender w (%) | 64.0 | 55.9 | 0.28 |
| Age (years) | 83.2 ± 5.4 | 82.4 ± 6,5 | 0.22 |
| BMI (kg/m²) | 27.1 ± 4.12 | 26.5 ± 5.0 | 0.25 |
| CAD (%) | 47.22 | 55.9 | 0.20 |
| 1 vessel | 5.6 | 14.7 | |
| 2 vessels | 11.1 | 5.9 | |
| 3 vessels | 30.6 | 35.3 | |
| In % | |||
| prev MI | 8.3 | 4.4 | 0.21 |
| prev CABG | 19.4 | 23.5 | 0.32 |
| prev PCI | 22.2 | 27.9 | 0.27 |
| In % | |||
| Diabetes | 41.7 | 26.5 | 0.13 |
| Hypertension | 100.0 | 97.1 | 0.15 |
| Hypercholesterolemia | 77.8 | 63.2 | 0.06 |
| Smoker | 2.8 | 8.8 | 0.12 |
| Family history | 5.6 | 5.9 | 0.47 |
| Ckd | 27.8 | 27.9 | 0.49 |
| Carotid disease | 11.1 | 16.1 | 0.24 |
| Periph disease | 16.7 | 20.6 | 0.31 |
| Af | 5.6 | 10.3 | 0.13 |
| Pulmonary disease | 11.1 | 29.4 | 0.0269 |
| Cancer | 2.8 | 13.2 | |
| Log Euroscore in % | 22.5 ± 13.6 | 22.4 ± 9.8 | 0.48 |
| Syncope in % | 27.8 | 7.4 | 0.0022 |
| CCS (I-IV) | 1.3 ± 0.8 | 1.0 ± 1.0 | 0.16 |
| NYHA (I-IV) | 3.0 ± 0.7 | 2.9 ± 0.6 | 0.50 |
| QRS-T angle ° | 130.4 ± 28.7 | 41.7 ± 23.8 | < 0.001 |
| Crea base (mg/dl) | 1.2 ± 0.7 | 1.2 ± 0.8 | 0.50 |
| Hb base (g/dl) | 12.0 ± 1.4 | 12.1 ± 1.7 | 0.36 |
| TnT base (ng/l) | 0.02 ± 0.02 | 0.1 ± 0.2 | 0.38 |
| crea post (mg/dl) | 1.3 ± 1.0 | 1.2 ± 0.8 | 0.28 |
| Hb post (g/dl) | 9.7 ± 1.3 | 9.9 ± 1.6 | 0.31 |
| TnT max post (ng/l) | 0.3 ± 0.2 | 0.2 ± 0.1 | 0.0178 |
| EF base in % | 51.4 ± 11.6 | 52.4 ± 9.9 | 0.38 |
| Left atrial diameter (mm) | 42.4 ± 5.4 | 42.8 ± 6.2 | 0.28 |
| Left enddiastolic diameter (mm) | 43.1 ± 5.8 | 47.6 ± 7.8 | 0.41 |
| Max V (m/s) | 4.3 ± 0.6 | 4.2 ± 0.7 | 0.32 |
| Max grad (mmHg) | 71.1 ± 25.8 | 71.4 ± 23.0 | 0.48 |
| Pmean mmHg base (mmHg) | 45.1 ± 10.7 | 43.9 ± 15.0 | 0.35 |
| AVA base (cm²) | 0.8 ± 0.2 | 0.7 ± 0.2 | 0.32 |
| EF post (%) | 52.4 ± 9.9 | 52.4 ± 9.1 | 0.45 |
| Velocity (m/s) | 2.3 ± 0.5 | 2.3 ± 0.5 | 0.19 |
| Pmean post (mmHg) | 10.9 ± 5.1 | 13.2 ± 4.9 | 0.15 |
| Pmax post (mmHg) | 20.9 ± 9.9 | 23.7 ± 8.5 | 0.16 |
| Valve dimensions (mm) | 26.9 ± 1.9 | 25.4 ± 2.0 | 0.0531 |
| Access in % | 100 | 100 | |
| Balloon size (mm) | 22.4 ± 1.6 | 21.8 ± 6.0 | 0.44 |
| P-P Gradient pre (mmHg) | 44.5 ± 14.5 | 44.5 ± 21.6 | 0.34 |
| P-P Gradient post (mmHg) | 7.5 ± 4.1 | 5.6 ± 5.4 | 0.14 |
b.) QRS T angle post TAVI
Immediate results post TAVI showed a higher number of patients with a wide QRS-T angle ≥ 90° (n = 48; mean angle 133.81° ± 30.95°) and significantly lower max grades of 64.85 ± 24.03 in comparison to 56 patients with a lower QRS T angle < 90° (max grad 75.47 ± 22.97; p = 0,0256). The left ventricular mass (Devereux) was significantly lower in patients with a QRS-T angle ≥ 90° (219.90 ± 53.01; p = 0.0498). (Table 3).
QSR-T angle and clinical data post-intervention stratified by QRS-T angle.
| QRS-T ≥ 90 post TAVI | QRST < 90 post TAVI | p value | |
|---|---|---|---|
| (n = 48) | (n = 56) | ||
| Gender w (%) | 60.4 | 54.4 | 0.30 |
| Age (years) | 83.3 ± 6.2 | 81.8 ± 6.1 | 0.11 |
| BMI (kg/m²) | 26.8 ± 4.7 | 26.5 ± 5.0 | 0.47 |
| CAD (%) | 49.1 | 39.7 | 0.26 |
| 1 vessel | 5.9 | 11.8 | |
| 2 vessels | 7.4 | 4.4 | |
| 3 vessels | 26.5 | 25.0 | |
| In % | |||
| prev MI | 8.3 | 3.5 | 0.15 |
| prev CABG | 25.0 | 19.3 | 0.26 |
| prev PCI | 27.9 | 29.8 | 0.20 |
| In % | |||
| Diabetes | 27.1 | 35.1 | 0.31 |
| Hypertension | 97.9 | 96.5 | 0.46 |
| Hypercholesterolemia | 77.1 | 59.7 | 0.04 |
| Smoker | 6.3 | 7.1 | 0.42 |
| Family history | 2.1 | 8.8 | 0.07 |
| Ckd | 31.3 | 26.3 | 0.31 |
| Carotid disease | 18.8 | 10.5 | 0.12 |
| Periph disease | 14.6 | 21.1 | 0.19 |
| Af | 10.4 | 8.8 | 0.30 |
| Pulmonary disease | 12.5 | 33.3 | 0.08 |
| Cancer | 8.3 | 10.5 | 0.34 |
| Log euroscore | 21.8 ± 11.6 | 22.9 ± 11.0 | 0.33 |
| Syncope in % | 18.8 | 10.5 | 0.12 |
| Ccs (i-iv) | 1.2 ± 1.0 | 1.1 ± 0.9 | 0.22 |
| Nyha (i-iv) | 2.9 ± 0.7 | 3.1 ± 0.6 | 0.11 |
| QRS-T angle ° | 133.8 ± 31.0 | 52.5 ± 26.3 | |
| Lab results | |||
| Creatinine pre (mg/dl) | 1.3 ± 0.7 | 1.2 ± 0.8 | 0.39 |
| Hb pre (g/dl) | 12.1 ± 1.5 | 12.0 ± 1.7 | 0.08 |
| Tnt pre (ng/l) | 0.1 ± 1.6 | 0.04 ± 0.02 | 0.18 |
| Creatinine post (mg/dl) | 1.2 ± 1.5 | 1.2 ± 0.9 | 0.39 |
| Hb post (g/dl) | 9.9 ± 1.5 | 9.8 ± 1.5 | 0.41 |
| Tnt max post (ng/l) | 0.2 ± 0.2 | 0.2 ± 0.1 | 0.08 |
| EF pre in % | 52.6 ± 11.6 | 51.7 ± 11.8 | 0.39 |
| Left atrial diameter (mm) | 43.0 ± 5.1 | 43.6 ± 6.7 | 0.32 |
| Left ventricular enddiastolic diameter (mm) | 47.6 ± 8.6 | 48.5 ± 7.5 | 0.29 |
| Max V (m/s) | 4.1 ± 0.6 | 4.3 ± 0.7 | 0.06 |
| Max grad (mmhg) | 64.9 ± 24.0 | 75.5 ± 23.0 | 0.0256 |
| Pmean mmhg pre (mmhg) | 43.2 ± 11.5 | 44.9 ± 15.2 | 0.29 |
| AVA pre (cm²) | 0.7 ± 0.2 | 0.8 ± 0.2 | 0.32 |
| EF post (%) | 54.7 ± 8.9 | 51.9 ± 9.5 | 0.15 |
| Velocity (m/s) | 2.2 ± 0.6 | 2.2 ± 0.5 | 0.49 |
| Pmean post (mmhg) | 11.8 ± 5.1 | 12.5 ± 5.3 | 0.34 |
| Pmax post (mmhg) | 22.1 ± 10.6 | 24.0 ± 7.8 | 0.25 |
| Valve dimensions (mm) | 26.5 ± 1.9 | 26.4 ± 2.2 | 0.41 |
| Access TF in % | 100 | 100 | |
| Balloon size (mm) | 20.2 ± 7.2 | 23.1 ± 1.8 | 0.0182 |
| P-P Gradient pre (mmhg) | 46.4 ± 15.8 | 45.1 ± 21.6 | 0.40 |
| P-P Gradient post (mmHg) | 6.0 ± 4.7 | 6.3 ± 5.5 | 0.42 |
c.) One-month follow-up data:
The one-month follow-up (30 days) data indicated a decrease in the patient rate with a high QRS-T angle (n = 22) compared to pre-TAVI and a 24-hour analysis post TAVI. The incidence of cancer and pulmonary diseases remained significantly higher in the group of patients with a lower QRS-T angle (< 90°; 0.0097 pulmonary disease; cancer 0.0432). (Table 4).
QSR-T angle and clinical data at follow-up stratified by QRS-T angle.
| QRS-T ≥ 90 FU TAVI | QRST < 90 FU TAVI | p value | |
|---|---|---|---|
| (n = 22) | (n = 82) | ||
| Gender w (%) | 45.46 | 61.0 | 0.10 |
| Age (years) | 83.0 ± 5.8 | 82.4 ± 6.23 | 0.34 |
| BMI (kg/m²) | 26.3 ± 4.0 | 26.9 ± 5.0 | 0.27 |
| CAD in % | 66.6 | 50.0 | 0.13 |
| 1 vessel | 2.9 | 14.7 | |
| 2 vessels | 2.9 | 8.8 | |
| 3 vessels | 14.7 | 36.8 | |
| In % | |||
| Prev MI | 9.1 | 4.9 | 0.23 |
| Prev CABG | 27.3 | 20.7 | 0.26 |
| Prev PCI | 40.9 | 29.3 | 0.15 |
| In % | |||
| Diabetes | 40.9 | 29.3 | 0.41 |
| Hypertension | 100 | 97.6 | 0.23 |
| Hypercholesterolemia | 77.3 | 65.9 | 0.16 |
| Smoker | 13.6 | 4.9 | 0.07 |
| Family history | 0 | 7.3 | 0.10 |
| Ckd | 13.6 | 32.9 | 0.04 |
| Carotid disease | 4.6 | 17.1 | 0.07 |
| Periph disease | 18.2 | 18.3 | 0.50 |
| Af | 0 | 12.2 | 0.40 |
| Pulmonary disease | 9.1 | 28.1 | 0.0097 |
| Cancer | 0 | 12.2 | 0.0432 |
| Log euroscore (%) | 21.1 ± 12.4 | 22.7 ± 11.0 | 0.28 |
| Syncope in % | 13.6 | 14.6 | 0.45 |
| Ccs (i-iv) | 1.1 ± 0.8 | 1.2 ± 0.9 | 0.46 |
| Nyha (i-iv) | 3.1 ± 0.2 | 3.0 ± 0.7 | 0.30 |
| QRS-T angle ° | 133.2 ± 27.4 | 38.7 ± 27.4 | < 0.001 |
| Lab results | |||
| Creatinine pre (mg/dl) | 1.1 ± 0.3 | 1.3 ± 0.8 | 0.16 |
| Hb pre (g/dl) | 12.5 ± 1.3 | 11.9 ± 1.7 | 0.10 |
| Tnt pre (ng/l) | 0.03 ± 0.01 | 0.1 ± 0.1 | 0.27 |
| Crea post (mg/dl) | 1.1 ± 0.4 | 58.5 ± 31.5 | 0.15 |
| Hb post (g/dl) | 10.1 ± 1.8 | 9.8 ± 1.4 | 0.29 |
| Tnt max post (ng/l) | 0.2 ± 0.2 | 0.2 ± 0.2 | 0.39 |
| EF pre in % | 53.0 ± 11.3 | 52.1 ± 11.9 | 0.38 |
| Left atrial diameter (mm) | 43.3 ± 7.4 | 43.3 ± 5.5 | 0.50 |
| Left ventricular enddiastolic diameter (mm) | 47.7 ± 7.0 | 48.2 ± 8.3 | 0.40 |
| Max V (m/s) | 4.2 ± 0.7 | 4.0 ± 0.7 | 0.48 |
| Max grad (mmhg) | 71.6 ± 21.2 | 70.8 ± 24.7 | 0.45 |
| Pmean pre (mmhg) | 44.0 ± 9.0 | 44.3 ± 14.5 | 0.47 |
| AVA pre (cm²) | 0.8 ± 0.2 | 0.7 ± 0.2 | 0.48 |
| EF post (%) | 53.0 ± 7.2 | 52.9 ± 9.7 | 0.49 |
| Velocity (m/s) | 2.2 ± 0.6 | 2.2 ± 0.6 | 0.46 |
| Pmean post (mmhg) | 13.6 ± 5.9 | 12.0 ± 5.1 | 0.22 |
| Pmax post (mmhg) | 24.2 ± 7.6 | 23.1 ± 9.4 | 0.38 |
| Valve dimensions (mm) | 27.1 ± 2.2 | 22.0 ± 5.1 | 0.0404 |
| Access TF in % | 100 | 100 | |
| Balloon size (mm) | 22.7 ± 1.5 | 22.0 ± 5.1 | 0.37 |
| P-P Gradient Pre (mmHg) | 43.1 ± 16.6 | 46.0 ± 20.1 | 0.35 |
| P-P Gradient Post (mmHg) | 7.6 ± 6.9 | 6.0 ± 4.8 | 0.21 |
Discussion
Our study shows that the QRS-T angle, an easily accessible risk marker computed with every routine surface ECG, widens early after transcatheter aortic valve replacement, reflecting global ischemic stress during valve intervention, and improves below baseline during follow-up, which in turn reflects the long-term effects of normalization of left ventricular hemodynamics and prognostic improvement (Figure 1).
Changes in the QRS-T angle over time. Before, immediately, and 1 month after TAVI. Values are displayed as mean QRST angles in degrees.
The QRS-T angle is a marker of electrical instability and poor prognosis. However, evidence of its potential role in patients with aortic valve stenosis is limited. In 34 patients with hemodynamically assessed aortic valve stenosis, the spatial QRS-T angle was correlated with the peak systolic and mean transvalvular gradients. A QRS-T angle above 90°was diagnostic of severe aortic valve stenosis (mean transvalvular gradient of ≥ 50 mmHg) [20]. In 372 patients undergoing SAVR, Erturk et al. found that in-hospital and long-term mortality were significantly higher in patients with a wide QRS-T angle above 90° [13]. An electrical risk score, including the frontal QRS-T angle, was associated with poor prognosis in patients with severe aortic valve stenosis following TAVI [21].
Only a few studies have focused on changes in the QRS-T angle. Widening of the QRS-T angle during follow-up was independently associated with mortality in 2,929 patients with heart failure [22]. In 152 of 455 patients from the Defibrillators in Nonischemic Cardiomyopathy Treatment Evaluation (DEFINITE) trial with serial ECG follow-ups, changes in the QRS-T angle over time were associated with changes in left ventricular function [23]. Perez-Alday et al. demonstrated dynamic changes of the prediction of sudden cardiac death by a combination of ECG markers (global electrical heterogeneity index) including the QRS-T angle from 15,716 patients of the Atherosclerosis Risk In Community study [17].
Our study highlights the dynamic changes in the QRS-T angle during the catheter-based treatment of severe aortic stenosis. Changes in the QRS-T angle (narrowing) following successful therapeutic interventions have been demonstrated in patients undergoing thrombolysis for pulmonary artery embolism [25], thrombolysis for ST-segment elevation myocardial infarction [26], percutaneous coronary intervention in patients with chronic total occlusion [27], and improvements in left ventricular function following renal transplantation [28]. These findings underscore the sensitivity of the QRS-T angle to alterations in myocardial electrical activity and structural remodeling after intervention.
During TAVI, patients experience extreme hemodynamic fluctuations that impose significant stress on the myocardium and cardiovascular system. These hemodynamic changes trigger a cascade of physiological responses, including myocardial remodeling, characterized by alterations in myocardial fibrosis, cellular architecture, and electrophysiological properties. The left ventricle, which is directly impacted by the relief of valvular obstruction, undergoes substantial electrical remodeling, as reflected in the shifting QRS-T angle values. This remodeling encompasses changes in depolarization and repolarization patterns due to modifications of myocardial conduction pathways and repolarization heterogeneity. Consequently, the QRS-T angle serves not only as a marker of electrical instability but also reflects the underlying structural and functional myocardial recovery. Importantly, the observed QRS-T angle dynamics during and after catheter-based aortic valve implantation suggest that this parameter can be leveraged as a model to investigate the complex interplay between myocardial structural changes, such as fibrosis progression or regression, and electrical remodeling in severe aortic stenosis. Understanding this interplay is crucial because myocardial fibrosis influences both mechanical function and arrhythmogenic potential.
This research has several limitations that need to be recognized. Firstly, the sample size was quite small, which might restrict the applicability of the results to a wider population. Furthermore, the study's retrospective nature limits the ability to determine causal links and could introduce selection bias. The follow-up period was also relatively brief, which hampers the evaluation of long-term changes in QRS-T angle dynamics and their clinical significance. Moreover, the lack of prognostic data, such as patient outcomes related to arrhythmia occurrence or survival, limits the capacity to associate QRS-T angle changes with significant clinical endpoints. These limitations underscore the necessity for prospective studies with longer follow-up durations and thorough outcome monitoring to better understand the prognostic importance of QRS-T angle variations after catheter-based aortic valve implantation.
Given the unique pathophysiological environment created by TAVI, with its abrupt hemodynamic shifts and subsequent myocardial adaptation, this patient population provides a valuable opportunity to study disease progression and recovery using noninvasive ECG markers, such as the QRS-T angle. Future research focusing on correlating QRS-T angle changes with advanced imaging modalities that assess myocardial fibrosis and function could yield deeper insights into the mechanisms of electrical and structural remodeling.
Moreover, integrating cutting-edge computational approaches, such as deep learning algorithms applied to large-scale patient data, could enhance the predictive accuracy and clinical utility of QRS-T angle monitoring [29]. Such advancements may facilitate personalized risk stratification and optimize post-TAVI management by identifying patients at a higher risk for adverse remodeling or arrhythmic events.
In summary, the QRS-T angle emerges as a promising, easily accessible biomarker that reflects the dynamic myocardial and electrical remodeling processes occurring during and after catheter-based aortic valve replacement, offering significant potential for both clinical monitoring and mechanistic research in patients with severe aortic stenosis.
AI Usage Statement
Generative AI was used in the creation of the graphical abstract figure.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Iung B, Baron G, Butchart EG, Delahaye F, Gohlke-Bärwolf C, Levang OW, Tornos P, Vanoverschelde JL, Vermeer F, Boersma E, Ravaud P, Vahanian A. A prospective survey of patients with valvular heart disease in Europe: The Euro Heart Survey on Valvular Heart Disease. Eur Heart J. 2003Jul;24(13):1231-43 doi: 10.1016/s0195-668x(03)00201-x. PMID: 12831818
2. Iung B, Delgado V, Rosenhek R, Price S, Prendergast B, Wendler O, De Bonis M, Tribouilloy C, Evangelista A, Bogachev-Prokophiev A, Apor A, Ince H, Laroche C, Popescu BA, Piérard L, Haude M, Hindricks G, Ruschitzka F, Windecker S, Bax JJ, Maggioni A, Vahanian A; EORP VHD II Investigators. Contemporary Presentation and Management of Valvular Heart Disease: The EURObservational Research Programme Valvular Heart Disease II Survey. Circulation. 2019Oct;140(14):1156-1169 doi: 10.1161/CIRCULATIONAHA.119.041080. Epub 2019 Sep 12. PMID: 31510787
3. Nkomo VT, Gardin JM, Skelton TN, Gottdiener JS, Scott CG, Enriquez-Sarano M. Burden of valvular heart diseases: a population-based study. Lancet. 2006Sep16;368(9540):1005-11 doi: 10.1016/S0140-6736(06)69208-8. PMID: 16980116
4. Ross J Jr, Braunwald E. Aortic stenosis. Circulation. 1968Jul;38(1 Suppl):61-7 doi: 10.1161/01.cir.38.1s5.v-61. PMID: 4894151
5. Bach DS, Siao D, Girard SE, Duvernoy C, McCallister BD Jr, Gualano SK. Evaluation of patients with severe symptomatic aortic stenosis who do not undergo aortic valve replacement: the role of subjectively overestimated operative risk. Circ Cardio- vasc Qual Outcomes. 2009;2:533-9
6. D. Horstkotte, F. Loogen, The natural history of aortic valve stenosis, European Heart Journal, Volume 9, Issue suppl_E, Apr. 1988 Pages 57-64, https://doi.org/10.1093/eurheartj/9.suppl_E.57
7. Schwarz F, Baumann P, Manthey J, Hoffmann M, Schuler G, Mehmel HC, Schmitz W, Kübler W. The effect of aortic valve replacement on survival. Circulation. 1982Nov;66(5):1105-10 doi: 10.1161/01.cir.66.5.1105. PMID: 7127696
8. Iung B, Cachier A, Baron G, Messika-Zeitoun D, Delahaye F, Tornos P, Gohlke-Bärwolf C, Boersma E, Ravaud P, Vahanian A. Decision-making in elderly patients with severe aortic stenosis: why are so many denied surgery? Eur Heart J. 2005Dec;26(24):2714-20 doi: 10.1093/eurheartj/ehi471. Epub 2005 Sep 1. PMID: 16141261
9. Elayda MA, Hall RJ, Reul RM, Alonzo DM, Gillette N, Reul GJ Jr, Cooley DA. Aortic valve replacement in patients 80 years and older. Operative risks and long-term results. Circulation. 1993Nov;88(5 Pt 2):II11-6 PMID: 8222145
10. Smith CR, Leon MB, Mack MJ, Miller DC, Moses JW, Svensson LG, Tuzcu EM, Webb JG, Fontana GP, Makkar RR, Williams M, Dewey T, Kapadia S, Babaliaros V, Thourani VH, Corso P, Pichard AD, Bavaria JE, Herrmann HC, Akin JJ, Anderson WN, Wang D, Pocock SJ; PARTNER Trial Investigators. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011Jun9;364(23):2187-98 doi: 10.1056/NEJMoa1103510. Epub 2011 Jun 5. PMID: 21639811
11. Leon MB, Smith CR, Mack MJ, Makkar RR, Svensson LG, Kodali SK, Thourani VH, Tuzcu EM, Miller DC, Herrmann HC, Doshi D, Cohen DJ, Pichard AD, Kapadia S, Dewey T, Babaliaros V, Szeto WY, Williams MR, Kereiakes D, Zajarias A, Greason KL, Whisenant BK, Hodson RW, Moses JW, Trento A, Brown DL, Fearon WF, Pibarot P, Hahn RT, Jaber WA, Anderson WN, Alu MC, Webb JG; PARTNER 2 Investigators. Transcatheter or Surgical Aortic-Valve Replacement in Intermediate-Risk Patients. N Engl J Med. 2016Apr28;374(17):1609-20 doi: 10.1056/NEJMoa1514616. Epub 2016 Apr 2. PMID: 27040324
12. Oehler A, Feldman T, Henrikson CA, Tereshchenko LG. QRS-T angle: a review. Ann Noninvasive Electrocardiol. 2014Nov;19(6):534-42 doi: 10.1111/anec.12206. Epub 2014 Sep 9. PMID: 25201032; PMCID: PMC4237708
13. Erturk M, Avci Y, Agus HZ, Guner A, Demir AR, Tasbulak O, Aslan S, Yildirim C, Can C, Oz K. The prognostic value of fQRSTa in patients with aortic stenosis undergoing surgical aortic valve replacement. J Card Surg. 2020Oct;35(10):2627-2632 doi: 10.1111/jocs.14858. Epub 2020 Jul 27. PMID: 32720442
14. Sweda R, Sabti Z, Strebel I, Kozhuharov N, Wussler D, Shrestha S, Flores D, Badertscher P, Lopez-Ayala P, Zimmermann T, Michou E, Gualandro DM, Häberlin A, Tanner H, Keller DI, Nowak A, Pfister O, Breidthardt T, Mueller C, Reichlin T. Diagnostic and prognostic values of the QRS-T angle in patients with suspected acute decompensated heart failure. ESC Heart Fail. 2020Aug;7(4):1817-1829 doi: 10.1002/ehf2.12746. Epub 2020 May 26. PMID: 32452635; PMCID: PMC7373892
15. Medvedovsky AT, Pollak A, Shuvy M, Gotsman I. Prognostic significance of the frontal QRS-T angle in patients with AL cardiac amyloidosis. J Electrocardiol. 2020 Mar-Apr;59:122-125. doi: 10.1016/j.jelectrocard.2020.02.001. Epub. 2020 Feb 6. PMID: 32062381
16. Dogan A, Kahraman S. Frontal QRS-T angle predicts coronary atherosclerotic burden in patients with ST segment elevation myocardial infarction. J Electrocardiol. 2020 Jan-Feb;58:155-159. doi: 10.1016/j.jelectrocard.2019.11.042. Epub. 2019 Nov 25. PMID: 31895991
17. Perez-Alday EA, Bender A, German D, Mukundan SV, Hamilton C, Thomas JA, Li-Pershing Y, Tereshchenko LG. Dynamic predictive accuracy of electrocardiographic biomarkers of sudden cardiac death within a survival framework: the Atherosclerosis Risk in Communities (ARIC) study. BMC Cardiovasc Disord. 2019Nov14;19(1):255 doi: 10.1186/s12872-019-1234-9. PMID: 31726979; PMCID: PMC6854807
18. Zadeh B, Wambach JM, Lambers M, Nassenstein K, Jensen CJ, Bruder O. QRS-T-angle in Patients with ST-Segment Elevation Myocardial Infarction (STEMI) - a Comparison with Cardiac Magnetic Resonance Imaging. Int J Med Sci. 2020;17(15):2264-2268
19. Jensen CJ, Lambers M, Zadeh B, Wambach JM, Nassenstein K, Bruder O. QRS-T angle in patients with Hypertrophic Cardiomyopathy - A comparison with Cardiac Magnetic Resonance Imaging. Int J Med Sci. 2021Jan1;18(3):821-825
20. Baumgartner H, Falk V, Bax JJ, De Bonis M, Hamm C, Holm PJ, Iung B, Lancellotti P, Lansac E, Rodriguez Muñoz D, Rosenhek R, Sjögren J, Tornos Mas P, Vahanian A, Walther T, Wendler O, Windecker S, Zamorano JL; ESC Scientific Document Group. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2017Sep21;38(36):2739-2791 doi: 10.1093/eurheartj/ehx391. PMID: 28886619
21. Talwar KK, Mohan JC, Narula J, Kaul U, Bhatia ML. Spatial quantitative vectorcardiography in aortic stenosis: correlation with hemodynamic findings. Int J Cardiol. 1988Feb;18(2):151-61 doi: 10.1016/0167-5273(88)90160-x. PMID: 3343071
22. Piccirillo G, Moscucci F, Mastropietri F, Di Iorio C, Mariani MV, Fabietti M, Stricchiola GM, Parrotta I, Sardella G, Mancone M, Magrì D. Possible predictive role of electrical risk score on transcatheter aortic valve replacement outcomes in older patients: preliminary data. Clin Interv Aging. 2018Sep11;13:1657-1667 doi: 10.2147/CIA.S170226. PMID: 30237702; PMCID: PMC6138964
23. Gotsman I, Shauer A, Elizur Y, Zwas DR, Lotan C, Keren A. Temporal changes in electrocardiographic frontal QRS-T angle and survival in patients with heart failure. PLoS One. 2018Mar26;13(3):e0194520 doi: 10.1371/journal.pone.0194520. PMID: 29579123; PMCID: PMC5868814
24. Pavri BB, Hillis MB, Subacius H, Brumberg GE, Schaechter A, Levine JH, Kadish A; Defibrillators in Nonischemic Cardiomyopathy Treatment Evaluation (DEFINITE) Investigators. Prognostic value and temporal behavior of the planar QRS-T angle in patients with nonischemic cardiomyopathy. Circulation. 2008Jun24;117(25):3181-6 doi: 10.1161/CIRCULATIONAHA.107.733451. PMID: 18574059
25. Şahan E, Aydemir S. Evaluation of thrombolytic treatment effect on frontal plane QRS-T angle in patients with acute pulmonary embolism: A novel marker of successful thrombolysis. Herz. 2020 Sep 25. English. doi: 10.1007/s00059-020-04978-0. Epub ahead of print. PMID: 32975629
26. Colluoglu T, Tanriverdi Z, Unal B, Ozcan EE, Dursun H, Kaya D. The role of baseline and post-procedural frontal plane QRS-T angles for cardiac risk assessment in patients with acute STEMI. Ann Noninvasive Electrocardiol. 2018Sep;23(5):e12558 doi: 10.1111/anec.12558. Epub 2018 Jun 6. PMID: 29873439; PMCID: PMC6931431
27. Uzun F, Güner A, Demir AR, Can A, Yalçin AA, Aguş HZ, Kalkan AK. Improvement of the frontal QRS-T angle after successful percutaneous coronary revascularization in patients with chronic total occlusion. Coron Artery Dis. 2020Dec;31(8):716-721 doi: 10.1097/MCA.0000000000000935. PMID: 32804782
28. Jaroszyński A, Furmaga J, Zapolski T, Zaborowski T, Rudzki S, Dąbrowski W. The improvement of QRS-T angle as a manifestation of reverse electrical remodeling following renal transplantation in end-stage kidney disease patients on haemodialysis. BMC Nephrol. 2019Dec2;20(1):441 doi: 10.1186/s12882-019-1624-3. PMID: 31791259; PMCID: PMC6889434
29. Sbrollini A, De Jongh MC, Ter Haar CC, Treskes RW, Man S, Burattini L, Swenne CA. Serial electrocardiography to detect newly emerging or aggravating cardiac pathology: a deep-learning approach. Biomed Eng Online. 2019Feb12;18(1):15 doi: 10.1186/s12938-019-0630-9. PMID: 30755195; PMCID: PMC6371549
Author contact
Corresponding author: Christoph Jensen, MD, Associate Professor of Medicine, Department of Cardiology and Pneumology, University Medical Center Göttingen, Robert Koch Strasse 40, Göttingen, Germany; Email: Chris.jensenuni-goettingen.de.

 Global reach, higher impact
Global reach, higher impact