Impact Factor ISSN: 1449-1907
Int J Med Sci 2026; 23(4):1444-1455. doi:10.7150/ijms.128956 This issue Cite
Research Paper
Association of Glucagon-like Peptide-1 Receptor Agonists with Mortality and Aspiration Pneumonia in Patients with Type 2 Diabetes After Gastrostomy: A Target Trial Emulation Study
1. Department of Public Health, College of Medicine, National Cheng Kung University, No. 1, University Road, Tainan City 701, Taiwan.
2. Department of Medical Research, Tainan Municipal Hospital (Managed by Show Chwan Medical Care Corporation), No. 670, Chongde Road, East District, Tainan City 701, Taiwan.
3. Division of Gastroenterology, Department of Internal Medicine, Chang Bing Show Chwan Memorial Hospital, No. 6, Lugong Road, Lugang Township, Changhua County 505, Taiwan.
4. Nursing Department, Nursing College, Hung Kuang University, No. 1018, Section 6, Taiwan Boulevard, Shalu District, Taichung City 433304, Taiwan.
5. Division of Cardiology, Department of Medicine, Tainan Sin-Lau Hospital, No. 57, Section 1, Dongmen Road, East District, Tainan City 701001, Taiwan.
6. Department of Health Care Administration, Chang Jung Christian University, No. 1, Changda Road, Gueiren District, Tainan City 711301, Taiwan.
7. Department of Nursing, Jenteh Junior College of Medicine, Nursing and Management, No. 79-9, Sha-Luen Hu, Xi-Zhou Li, Houlong Township, Miaoli County 356, Taiwan.
8. Department of Public Health, College of Public Health, China Medical University, Taichung, Taiwan, No. 91, Hsueh-Shih Road, Taichung 40402, Taiwan.
9. Department of Healthcare Administration, College of Medical and Health Science, Asia University, No. 500, Lioufeng Rd., Wufeng, Taichung 41354, Taiwan.
* These authors contributed equally to this work.
Received 2025-11-25; Accepted 2026-3-4; Published 2026-3-17
Abstract
Background: This study aimed to compare the safety and effectiveness of glucagon-like peptide-1 receptor agonists (GLP-1 RA) versus dipeptidyl peptidase-4 inhibitors (DPP-4i) in patients with type 2 diabetes following gastrostomy.
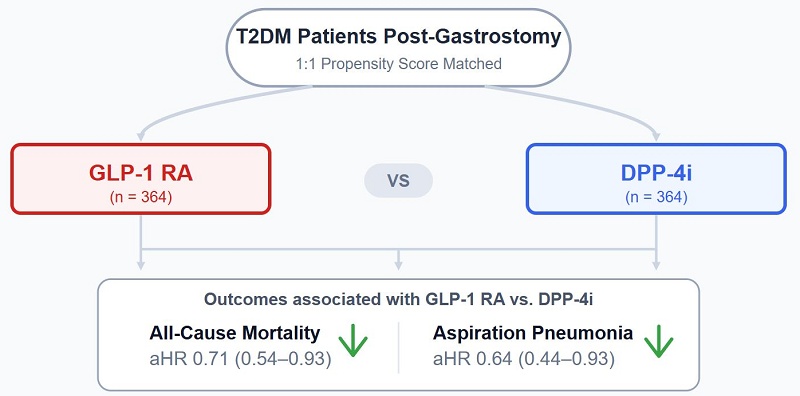
Methods: We conducted a target trial emulation using real-world data. From January 1, 2015, to December 31, 2024, we identified 728 patients who initiated either GLP-1 RA or DPP-4i after gastrostomy. After 1:1 propensity score matching to balance baseline covariates, 364 patients were included in each group. The primary analysis followed an intention-to-treat principle, and follow-up continued until June 30, 2025. Several sensitivity analyses, including landmark analysis and E-value calculation, were performed to assess the robustness of the findings.
Results: Compared with DPP-4i initiation, GLP-1 RA initiation was associated with lower all-cause mortality (adjusted hazard ratio [aHR], 0.71; 95% confidence interval [CI], 0.54-0.93) and aspiration pneumonia (aHR, 0.64; 95% CI, 0.44-0.93).
Conclusions: Our findings indicate that initiation of GLP-1 RA, compared with initiation of DPP-4i, was associated with lower all-cause mortality and a lower risk of aspiration pneumonia in patients with type 2 diabetes following gastrostomy. Further prospective studies are warranted to confirm these findings in this vulnerable population.
Keywords: glucagon-like peptide-1 receptor agonists, gastrostomy, all-cause mortality, target trial emulation
1. Introduction
Type 2 diabetes mellitus (T2DM) is a globally prevalent chronic condition characterized by significant morbidity and mortality, primarily from cardiovascular complications [1]. Contemporary diabetes management has evolved to incorporate GLP-1 RA and DPP-4i as foundational treatment options, reflecting advances in glucose-lowering pharmacotherapy [2, 3]. Large-scale cardiovascular outcome trials (CVOTs) have demonstrated that GLP-1 RA improve glycemic control and reduce both major adverse cardiovascular events (MACE) and all-cause mortality in T2DM populations [4-6]. Conversely, DPP-4i have shown neutral cardiovascular effects [7].
Despite robust trial data in general diabetes populations, significant uncertainties remain regarding treatment selection in high-risk subgroups who were not represented in landmark cardiovascular outcome studies. Type 2 diabetes patients requiring gastrostomy tubes represent a medically complex population facing substantial mortality risks. These individuals typically present with numerous concurrent health conditions, including malignancies, neurological disorders, and nutritional deficiencies, that contribute to elevated morbidity beyond traditional cardiovascular complications [8, 9].
Choosing between GLP-1 RA and DPP-4i in post-gastrostomy patients presents a clinical dilemma. While GLP-1 RA offer established benefits, their delayed gastric emptying raises concerns about aspiration pneumonia [10, 11], particularly in this frail population already predisposed to aspiration [12]. Given the clinical importance of aspiration complications in patients with severe dysphagia, evidence to guide optimal therapeutic decisions is lacking.
To address this gap, we utilized target trial emulation to compare risks and benefits of GLP-1 RA versus DPP-4i in real-world T2DM patients following gastrostomy. We evaluated all-cause mortality (primary), plus MACE and aspiration pneumonia (secondary outcomes) in this complex population.
2. Methods
2.1 Study Design and Data Source
This analysis employed a retrospective cohort approach with target trial emulation methodology [13, 14]. We utilized the TriNetX global health network, which aggregates de-identified patient records from healthcare institutions across multiple countries. The platform provides access to detailed clinical information encompassing patient demographics, diagnostic codes (ICD-10-CM), procedural data, medication histories, and laboratory findings [15]. Data de-identification has been validated by certified experts following HIPAA Privacy Rule requirements (Section §164.514(b)(1)), ensuring patient confidentiality and regulatory compliance. Because the dataset is fully de-identified, this study was granted approval by the Institutional Review Board of Show Chwan Memorial Hospital (IRB No: 1141007) with a waiver of informed consent. [16-18]
2.2 Study Population and Cohort Construction
Our study cohort comprised 59,043 individuals with type 2 diabetes who received gastrostomy tubes during the period from January 2015 through December 2024 (Figure 1). We excluded patients <18 years (n=1,480) and those without GLP-1 RA or DPP-4i use (n=49,064), yielding 8,499 eligible patients.
Patient Selection Flowchart for the Intention-to-Treat (ITT) Cohorts. The study cohort was selected from an initial population of 59,043 patients with type 2 diabetes who underwent gastrostomy. An active-comparator design was implemented, defining eligible patients as those who initiated either a GLP-1 RA or a DPP-4i within six months post-procedure. A 12-month washout period was applied, requiring patients to be naive to the comparator drug class in the year prior to gastrostomy. Baseline covariates were captured during a 5-year lookback period before the index date. Subsequently, 1:1 propensity score matching was performed using these covariates (detailed in Table 1), resulting in 364 patients per group. Following the ITT principle, all matched patients were analyzed according to their initial treatment assignment, regardless of subsequent medication changes during follow-up. As a sensitivity analysis, the PP analysis excluded patients who switched between GLP-1 RA and DPP-4i during the follow-up period to evaluate treatment effects among patients who remained on their initial therapy. Abbreviations: DPP-4i, dipeptidyl peptidase-4 inhibitors; GLP-1 RA, glucagon-like peptide-1 receptor agonists; ITT, Intention-to-Treat; PP: per-protocol.
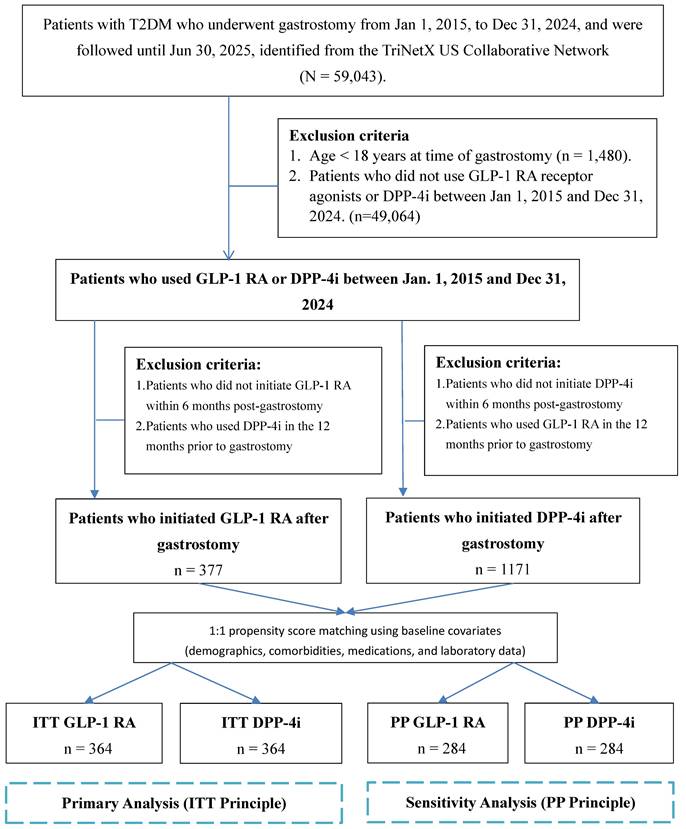
Following an active comparator design for the overall cohort, we identified patients who initiated either GLP-1 RA or DPP-4i within 6 months after gastrostomy, regardless of whether they had prior use of the index drug before the procedure (i.e., the primary analysis included both new initiators and prevalent users who continued therapy post-gastrostomy). To minimize carryover confounding from the comparator drug class, a 12-month washout period was applied, requiring patients to have no prescription records of the comparator drug in the 12 months preceding gastrostomy. Specifically, the GLP-1 RA group initiated treatment within 6 months post-gastrostomy without DPP-4i use in the 12 months prior to gastrostomy. The DPP-4i group initiated treatment within 6 months post-gastrostomy without GLP-1 RA use in the 12 months prior to gastrostomy.
We performed 1:1 propensity score matching to balance baseline differences, creating final intention-to-treat (ITT) cohorts of 364 patients each. The index date was the first GLP-1 RA or DPP-4i prescription post-gastrostomy [19, 20].
2.3 Outcome Measures
Patients were followed from the index date until the occurrence of all-cause mortality, loss to follow-up, or the end of the study observation period. The primary outcome was all-cause mortality. Secondary outcomes included MACE and the incidence of pneumonia. MACE was defined as a composite endpoint of acute myocardial infarction, stroke (ischemic or hemorrhagic), and cardiac arrest. Aspiration pneumonia was identified using diagnosis codes; the database does not allow reliable adjudication of whether events were peri-procedural/anesthesia-related versus feeding-related. Follow-up ended at the first outcome event, death, the last recorded encounter (loss to follow-up), or the end of data availability; apart from recorded death, reasons for study termination cannot be fully distinguished.
2.4 Covariates
Baseline patient characteristics were captured during a 5-year lookback period prior to the index date. Covariates included for propensity score matching (PSM) were demographics (age, sex, race), anthropometrics (body mass index, BMI), social and behavioral factors (nicotine dependence, alcohol abuse), major comorbidities (e.g., hypertension, hyperlipidemia, chronic kidney disease, heart failure), concomitant medications (e.g., insulin, metformin, statins, ACE inhibitors/angiotensin II receptor blockers, ARBs), and baseline laboratory data (e.g., hemoglobin A1c; HbA1c, estimated glomerular filtration rate; eGFR, albumin, C-reactive protein). For the purpose of subgroup analysis, a composite variable for atherosclerotic cardiovascular disease (ASCVD) was created, defined as a baseline diagnosis of either ischemic heart disease or cerebrovascular disease (see Supplementary Table 1 for detailed definitions and codes).
2.5 Statistical Analysis
Initial cohorts included 377 GLP-1 RA and 1,171 DPP-4i users. 1:1 PSM used nearest neighbor matching with 0.2 SD caliper to balance covariates, yielding 364 patients per group [21]. Primary analysis followed ITT principles. We used Kaplan-Meier curves with log-rank tests and Cox proportional hazards models for adjusted hazard ratios (aHRs) with 95% confidence intervals (CIs).
To assess how the hazard ratios changed over time, landmark analyses were conducted at 30, 60, and 180 days post-index date; for each landmark time point, the analysis included only those patients who were still alive and had not experienced the outcome of interest [22]. To evaluate the robustness of our findings against potential unmeasured confounding, an E-value was calculated for the primary outcome [23]. A two-sided p-value < 0.05 was considered statistically significant. Primary analyses were performed using the integrated analytical tools of the TriNetX platform. All data visualization and subsequent advanced analyses were conducted using R software (version 4.3.2; R Foundation for Statistical Computing) through the RStudio interface (version 2023.12.0; Posit PBC).
2.6 Subgroup and Sensitivity Analyses
To assess the consistency of the associations with the primary outcome, we conducted several prespecified subgroup analyses stratified by the following baseline characteristics: GLP-1 RA user type (long-term vs. new user; defined by whether patients had any prescription record of the index drug at any time prior to gastrostomy), history of ASCVD, history of heart failure, baseline eGFR (< 60 vs. ≥ 60 mL/min/1.73m²), age (< 65 vs. ≥ 65 years), BMI (< 30 vs. ≥ 30 kg/m²), and history of cancer. The potential for effect modification was assessed by including an interaction term in the Cox model.
Furthermore, a series of sensitivity analyses were performed to evaluate the robustness of our findings. First, to account for all-cause mortality as a competing risk for non-fatal outcomes, we analyzed composite outcomes of (1) MACE or all-cause mortality and (2) pneumonia or all-cause mortality using a Cox model [24, 25]. Second, to evaluate the stability of our matching strategy, we constructed five different propensity score models with varying levels of covariate adjustment and compared the resulting aHRs. Third, we conducted a per-protocol (PP) analysis that excluded patients who switched from their initial treatment group to the comparator's drug class during follow-up.
2.7 Validation Analyses
To evaluate potential residual confounding and selection bias, we conducted several validation analyses. For positive outcome controls, we assessed the risk of biliary-related diseases and gastrointestinal adverse events [26]. For negative outcome controls, we assessed the risk of appendicitis, urinary tract infection, and acute pancreatitis [27]. Furthermore, for validation, we conducted a positive exposure control comparing statin users to non-users, and a negative exposure control comparing vitamin C users to non-users [28].
3. Results
3.1 Study Population Characteristics
Our study initially identified 59,043 patients with type 2 diabetes who underwent a gastrostomy procedure from the TriNetX network. After applying exclusion criteria, we established a pre-matched cohort of 377 GLP-1 RA users and 1,171 DPP-4i users. Before matching, significant baseline differences were observed between the groups; for instance, the GLP-1 RA group was younger (mean age 62.9 vs. 67.2 years) and had a higher BMI (30.5 vs. 27.3 kg/m²) compared to the DPP-4i group. Following a 1:1 PSM procedure, two analytical cohorts of 364 patients each were created. PSM was performed using the nearest neighbor approach with a 0.2 SD caliper to achieve the best possible covariate balance (the patient selection process is detailed in Figure 1, and baseline characteristics are compared in Table 1).
Baseline Characteristics of Patients Treated with GLP-1 RA or DPP-4i Before and After PSM
| Before PSM | After PSM | |||||
|---|---|---|---|---|---|---|
| Characteristics | GLP-1 RA (N= 377) | DPP-4i (N= 1,171) | SMD | GLP-1 RA (N= 364) | DPP-4i (N= 364) | SMD |
| Demographics | ||||||
| Age at Index | 62.9±11.9 | 67.2±11.5 | 0.3699 | 63.2±11.7 | 63.5±11.7 | 0.0221 |
| Sex | ||||||
| Male | 217(57.6%) | 679(58.0%) | 0.0086 | 212(58.2%) | 224(61.5%) | 0.0673 |
| Female | 155(41.1%) | 474(40.5%) | 0.0129 | 147(40.4%) | 136(37.4%) | 0.0620 |
| Race | ||||||
| White | 242(64.2%) | 594(50.7%) | 0.2749 | 235(64.6%) | 232(63.7%) | 0.0172 |
| Black | 63(16.7%) | 284(24.3%) | 0.1877 | 63(17.3%) | 58(15.9%) | 0.0369 |
| Asian | 13(3.4%) | 119(10.2%) | 0.2690 | 13(3.6%) | 16(4.4%) | 0.0422 |
| Unknown Race | 36(9.5%) | 89(7.6%) | 0.0696 | 31(8.5%) | 32(8.8%) | 0.0098 |
| Other Race | 16(4.2%) | 48(4.1%) | 0.0073 | 15(4.1%) | 19(5.2%) | 0.0521 |
| Lifestyles | ||||||
| Nicotine dependence | 84(22.3%) | 225(19.2%) | 0.0757 | 80(22.0%) | 82(22.5%) | 0.0132 |
| Alcohol abuse | 21(5.6%) | 67(5.7%) | 0.0066 | 20(5.5%) | 22(6.0%) | 0.0236 |
| BMI | 30.5±7.8 | 27.3±7.5 | 0.4077 | 30.5±7.7 | 28.3±7.4 | 0.0342 |
| Social economic status | ||||||
| Socioeconomic Hazards | 30(8.0%) | 74(6.3%) | 0.0637 | 25(6.9%) | 27(7.4%) | 0.0213 |
| Comorbidities | ||||||
| Hypertension | 339(89.9%) | 1068(91.2%) | 0.0439 | 329(90.4%) | 331(90.9%) | 0.0189 |
| Hyperlipidemia | 270(71.6%) | 809(69.1%) | 0.0555 | 259(71.2%) | 265(72.8%) | 0.0367 |
| Cerebrovascular diseases | 184(48.8%) | 632(54.0%) | 0.1035 | 178(48.9%) | 189(51.9%) | 0.0605 |
| Ischemic heart diseases | 175(46.4%) | 544(46.5%) | 0.0007 | 171(47.0%) | 184(50.5%) | 0.0715 |
| GERD | 173(45.9%) | 474(40.5%) | 0.1094 | 166(45.6%) | 174(47.8%) | 0.0441 |
| Cancer | 159(42.2%) | 497(42.4%) | 0.0054 | 156(42.9%) | 149(40.9%) | 0.0390 |
| Malnutrition | 145(38.5%) | 525(44.8%) | 0.1295 | 144(39.6%) | 144(39.6%) | 0.0000 |
| CKD | 134(35.5%) | 490(41.8%) | 0.1296 | 130(35.7%) | 134(36.8%) | 0.0229 |
| Pneumonitis | 103(27.3%) | 353(30.1%) | 0.0624 | 97(26.6%) | 102(28.0%) | 0.0308 |
| COPD | 76(20.2%) | 236(20.2%) | 0.0001 | 75(20.6%) | 79(21.7%) | 0.0269 |
| Inflammatory arthritis | 56(14.9%) | 179(15.3%) | 0.0121 | 53(14.6%) | 55(15.1%) | 0.0155 |
| Osteoporosis | 49(13.0%) | 151(12.9%) | 0.0030 | 46(12.6%) | 47(12.9%) | 0.0082 |
| IBD | 41(10.9%) | 139(11.9%) | 0.0313 | 39(10.7%) | 32(8.8%) | 0.0649 |
| Intracranial injury | 39(10.3%) | 123(10.5%) | 0.0052 | 34(9.3%) | 41(11.3%) | 0.0633 |
| Dementia | 20(5.3%) | 160(13.7%) | 0.2882 | 20(5.5%) | 24(6.6%) | 0.0461 |
| Liver cirrhosis | 16(4.2%) | 50(4.3%) | 0.0013 | 16(4.4%) | 15(4.1%) | 0.0136 |
| Spine fracture | 13(3.4%) | 54(4.6%) | 0.0592 | 13(3.6%) | 17(4.7%) | 0.0553 |
| Parkinson's disease | 10(2.7%) | 43(3.7%) | 0.0583 | 10(2.7%) | 10(2.7%) | 0.0000 |
| SLE | 10(2.7%) | 10(0.9%) | 0.1374 | 10(2.7%) | 10(2.7%) | 0.0000 |
| Medications | ||||||
| Analgesics | 363(96.3%) | 1133(96.8%) | 0.0256 | 350(96.2%) | 347(95.3%) | 0.0408 |
| Antithrombotic agents | 362(96.0%) | 1107(94.5%) | 0.0701 | 349(95.9%) | 349(95.9%) | 0.0000 |
| Insulin | 354(93.9%) | 1085(92.7%) | 0.0497 | 342(94.0%) | 342(94.0%) | 0.0000 |
| Statin | 308(81.7%) | 900(76.9%) | 0.1196 | 299(82.1%) | 296(81.3%) | 0.0213 |
| Beta blocking agents | 308(81.7%) | 958(81.8%) | 0.0029 | 298(81.9%) | 304(83.5%) | 0.0436 |
| Proton pump inhibitors | 296(78.5%) | 856(73.1%) | 0.1267 | 286(78.6%) | 289(79.4%) | 0.0202 |
| Diuretics | 294(78.0%) | 840(71.7%) | 0.1445 | 282(77.5%) | 284(78.0%) | 0.0132 |
| Corticosteroids | 281(74.5%) | 769(65.7%) | 0.1946 | 271(74.5%) | 264(72.5%) | 0.0436 |
| Benzodiazepine derivatives | 281(74.5%) | 837(71.5%) | 0.0689 | 270(74.2%) | 272(74.7%) | 0.0126 |
| Dihydropyridine derivatives | 218(57.8%) | 657(56.1%) | 0.0347 | 211(58.0%) | 210(57.7%) | 0.0056 |
| Metformin | 220(58.4%) | 532(45.4%) | 0.2609 | 208(57.1%) | 209(57.4%) | 0.0056 |
| ACEI | 167(44.3%) | 515(44.0%) | 0.0064 | 165(45.3%) | 158(43.4%) | 0.0387 |
| metoclopramide | 132(35.0%) | 366(31.3%) | 0.0799 | 129(35.4%) | 130(35.7%) | 0.0057 |
| Laboratory Tests | ||||||
| Platelets | 285.0±110.5 | 271.8±117.3 | 0.1156 | 282.5±109.6 | 282.4±121.2 | 0.0408 |
| Albumin | 3.3±0.7 | 3.1±0.7 | 0.2346 | 3.2±0.7 | 3.1±0.7 | 0.0000 |
| eGFR | 80.8±29.8 | 77.8±31.8 | 0.0968 | 79.8±29.8 | 82.4±32.6 | 0.0289 |
| HbA1c | 7.8±2.0 | 7.3±1.9 | 0.2198 | 7.8±2.0 | 7.3±2.0 | 0.0128 |
| CRP | 51.7±68.6 | 56.9±70.9 | 0.0746 | 53.8±69.8 | 48.9±59.5 | 0.0449 |
3.1 Primary and Secondary Outcomes
Median follow-up was 1.93 years (GLP-1 RA) and 1.73 years (DPP-4i). Kaplan-Meier analysis showed a lower mortality risk with GLP-1 RA (log-rank P = 0.015) (Figure 2A).
Kaplan-Meier Curves for the Cumulative Risk of All-Cause Mortality and Aspiration Pneumonia in Patients Using GLP-1 RA Versus DPP-4i. Kaplan-Meier curves illustrate the cumulative risk of (A) All-Cause Mortality and (B) Aspiration Pneumonia in propensity score-matched cohorts of patients treated with GLP-1 RA (red line) and DPP-4i (blue line). P-values were derived from the log-rank test to compare the curves. The shaded areas represent the 95% confidence intervals. The number of patients at risk at various time points is provided below each panel. Abbreviations: DPP-4i, dipeptidyl peptidase-4 inhibitors; GLP-1 RA, glucagon-like peptide-1 receptor agonists.
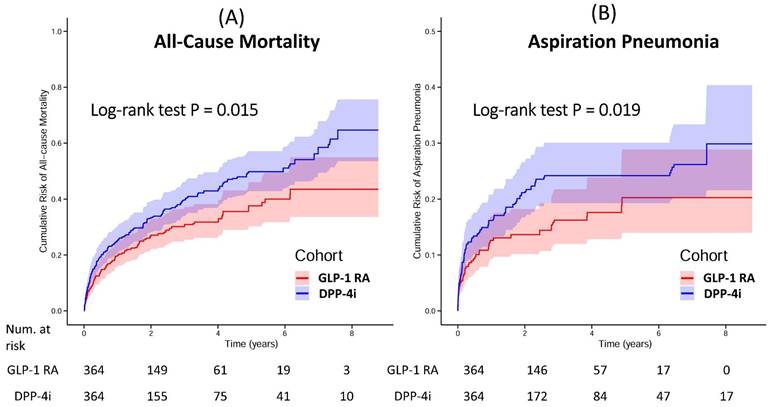
GLP-1 RA use was associated with lower aspiration pneumonia incidence (aHR: 0.64, 95% CI: 0.44-0.93; log-rank P = 0.019) (Figure 2B). No significant difference emerged for MACE (aHR: 1.01, 95% CI: 0.79-1.28).
Overall, the use of GLP-1 RA was associated with a lower risk of the primary outcome, all-cause mortality, compared to the use of DPP-4i (aHR: 0.71, 95% Confidence Interval [CI]: 0.54-0.93).
Landmark analyses revealed temporal variation in the association with mortality, with the strongest association observed after 30 days of follow-up (aHR: 0.63, 95% CI: 0.44-0.89), while associations at 60 and 180 days were not statistically significant. The E-value for the all-cause mortality point estimate was 2.17 (confidence interval limit: 3.11), suggesting robustness against potential unmeasured confounding (Table 2).
Overall and Landmark Hazard Ratios for MACE and All-Cause Mortality in the Matched Cohort
| Post-matching Analysis | Landmark analyses c | |||||||
|---|---|---|---|---|---|---|---|---|
| Outcomes | GLP-1 RA (n=364) | DPP-4i (n=364) | Overall HR (95%CI) a | Overall E-value b | >30 days HR (95% CI) | >60 days HR (95% CI) | >180 days HR (95% CI) | |
| Point value | 95% CI Limit | |||||||
| All-cause Mortality | 91 | 131 | 0.71 (0.54 -0.93) | 2.17 | 3.11 | 0.63 (0.44-0.89) | 0.79 (0.52-1.21) | 0.99 (0.62-1.56) |
| MACE | 135 | 136 | 1.01(0.79 -1.28) | 1.11 | 1.00 | 0.96 (0.68-1.36) | 0.70 (0.45-1.08) | 1.13 (0.67-1.91) |
| Aspiration Pneumonia | 47 | 71 | 0.64(0.44 -0.93) | 2.50 | 3.97 | 0.64 (0.40-1.02) | 0.81 (0.48-1.37) | 0.77 (0.45-1.32) |
a Propensity score matching was performed on all listed characteristics.
b The E-value represents the minimum strength of association that an unmeasured confounder would need to have with both the exposure and the outcome to fully explain the observed hazard ratio. In this analysis, the E-value was 2.17 for all-cause mortality, 1.11 for major adverse cardiovascular events (MACE), and 2.50 for aspiration pneumonia, suggesting that the findings are relatively robust to potential unmeasured confounding.
c A landmark approach was used to analyze risk at different time points. The >30 days analysis included only patients who were alive and event-free at day 31 to assess their subsequent risk. This was similarly applied to the >60 days and >180 days analyses, which respectively included event-free survivors at day 61 and day 181.
* The mean (±SD) follow-up duration was 3.53 (±3.63) years in the GLP-1 RA group and 2.85 (±3.06) years in the DPP-4i group. The median follow-up times were 1.93 and 1.73 years, respectively.
Abbreviations: CI, confidence interval; DPP-4i, dipeptidyl peptidase-4 inhibitors; GLP-1 RA, glucagon-like peptide-1 receptor agonists; HR, hazard ratio; MACE, major adverse cardiovascular events.
3.2 Subgroup Analyses
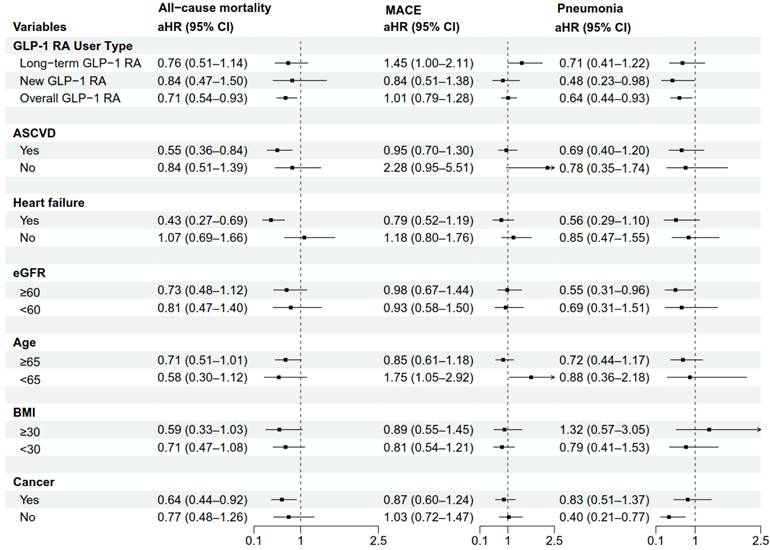
To assess the consistency of the associations, we conducted prespecified subgroup analyses for all-cause mortality, MACE, and pneumonia (Figure 3). For all-cause mortality, the association with lower mortality for GLP-1 RA was consistent across multiple subgroups, with a more pronounced trend observed in patients with a history of heart failure (aHR: 0.43, 95% CI: 0.27-0.69), a BMI ≥ 30 kg/m² (aHR: 0.59, 95% CI: 0.33-1.03), and a history of cancer (aHR: 0.64, 95% CI: 0.44-0.92). However, the results for MACE and pneumonia risk varied considerably across subgroups, with most not reaching statistical significance. Notably, among patients younger than 65 years, GLP-1 RA use was associated with an increased risk of MACE (aHR: 1.75, 95% CI: 1.05-2.92). This finding should be interpreted with caution, as it may be a chance occurrence due to the multiple comparisons performed.
Subgroup Analyses of Mortality, MACE, and Pneumonia for GLP-1 RA Versus DPP-4i. The forest plot displays the adjusted hazard ratios (aHRs) for the use of GLP-1 RA compared with DPP-4i (reference group) on the outcomes of all-cause mortality, MACE, and pneumonia across various prespecified subgroups. The squares represent the point estimates of the aHRs, and the horizontal lines represent the 95% confidence intervals (CIs). A 'New GLP-1 RA user' was defined as a patient with no prior prescription record for GLP-1 RA at any time before gastrostomy, whereas a 'Long-term GLP-1 RA user' was defined as a patient who had an existing GLP-1 RA prescription before gastrostomy and continued use within 6 months post-procedure. Abbreviations: aHR, adjusted hazard ratio; ASCVD, atherosclerotic cardiovascular disease; BMI, body mass index; CI, confidence interval; DPP-4i, dipeptidyl peptidase-4 inhibitors; eGFR, estimated glomerular filtration rate; GLP-1 RA, glucagon-like peptide-1 receptor agonists; MACE, major adverse cardiovascular events.
3.3 Sensitivity and Validation Analyses
To verify the robustness of our findings, we performed a series of sensitivity and validation analyses. For positive outcome controls, we assessed the risk of biliary-related diseases and gastrointestinal adverse events. The results showed no statistically significant difference (aHR 0.96 and 0.81, respectively). We interpret the absence of the expected signal in these positive outcome controls cautiously, as symptom-driven adverse events may be incompletely ascertained in routinely collected EHR data; mild-to-moderate gastrointestinal symptoms are often managed conservatively and may not be consistently captured as diagnosis-coded events unless they lead to distinct medical encounters or interventions, which can reduce sensitivity to detect an expected association. In addition, the sample size (n=364 per group) may be insufficient to detect modest differences in these adverse event rates. In the analysis of negative outcome controls (e.g., appendicitis, urinary tract infection), there was no significant difference in risk between the groups, suggesting the absence of major systemic bias. Crucially, however, the positive exposure control analysis successfully replicated the known mortality benefit of statins versus non-users (aHR: 0.75, 95% CI: 0.69-0.82), while the negative exposure control (Vitamin C) showed no association with mortality (Supplementary Figure 1). This suggests that our propensity score matching strategy and data source are valid and sufficiently sensitive to detect true differences in the primary outcome (mortality), despite limited sensitivity for secondary, symptom-driven adverse event signals.
Sensitivity analyses using five different propensity score models with varying levels of covariate adjustment showed that the aHRs for all-cause mortality and aspiration pneumonia remained relatively stable (all-cause mortality range: 0.63-0.76; aspiration pneumonia range: 0.57-0.69) (Supplementary Table 2), supporting the stability of our matching strategy. Finally, the results of the per-protocol analysis were directionally consistent with the primary ITT analysis and suggested a stronger trend of risk reduction for all-cause mortality and aspiration pneumonia (aHR for all-cause mortality: 0.64 vs. 0.71; aHR for aspiration pneumonia: 0.52 vs. 0.64) (Supplementary Table 3), which support the reliability of the study conclusions.
4. Discussion
In a unique and vulnerable population of patients with T2DM post-gastrostomy, this study employed a target trial emulation framework to compare the clinical associations and risks of GLP-1 RA versus DPP-4i. The findings delineate a nuanced association profile, providing real-world evidence for this population frequently excluded from large-scale clinical trials.
4.1 Summary of Main Findings
Three key findings emerged: First, after PSM, GLP-1 RA was associated with 29% lower all-cause mortality versus DPP-4i (aHR: 0.71, 95% CI: 0.54-0.93). Second, GLP-1 RA was associated with a lower aspiration pneumonia risk (aHR: 0.64, 95% CI: 0.44-0.93), contradicting expectations from delayed gastric emptying. Third, no MACE difference was observed (aHR: 1.01, 95% CI: 0.79-1.28), unlike broader T2DM populations. A subgroup analysis identified a potential signal for increased MACE risk in patients < 65 years (aHR: 1.75, 95% CI: 1.05-2.92); however, as discussed in the Limitations section, this finding requires prospective validation and should not be interpreted as established evidence of differential safety.
4.2 Mortality Association in a High-Risk Gastrostomy Cohort
This study demonstrates an association between GLP-1 RA and reduced mortality risk compared to DPP-4i. The direction of this association is consistent with meta-analyses of multiple cardiovascular outcome trials (CVOTs), which have uniformly shown that GLP-1 RA reduce all-cause mortality relative to placebo and are superior to DPP-4i in head-to-head comparisons [29, 30]. For instance, a recent meta-analysis reported a pooled HR of 0.82 for all-cause mortality with GLP-1 RA, while another indicated a 12% risk reduction [31].
The association with lower all-cause mortality was consistent across most subgroups, including older adults (Age ≥ 65). Unlike CVOT participants, our post-gastrostomy cohort faces high risks of non-cardiovascular death (e.g., sepsis, cachexia) due to severe comorbidities. The observed lower mortality without a corresponding MACE reduction suggests GLP-1 RA use may be associated with lower mortality through non-atherosclerotic pathways, possibly involving anti-inflammatory effects [32] or enhanced resilience [33]. However, this association was most pronounced early after initiation and attenuated over time, a pattern that may reflect treatment channeling toward clinically stable patients. Accordingly, we interpret these temporal findings cautiously as observational associations rather than definitive causal effects. The temporal attenuation of the mortality association in landmark analyses warrants cautious interpretation. While such a pattern might reflect a diminishing treatment effect over time, it could also arise from survivor-related or informative censoring bias, treatment channeling toward more clinically stable patients at initiation, or reduced statistical power due to smaller sample sizes at later time points.
4.3 Association with Aspiration Pneumonia Risk
We observed a lower incidence of aspiration pneumonia among GLP-1 RA initiators compared with DPP-4i initiators. Because aspiration pneumonia was identified using diagnosis codes in a frail population, outcome misclassification and differential ascertainment (e.g., variation in diagnostic workup, coding practices, and care intensity) cannot be excluded. In addition, the available data do not allow reliable classification of whether aspiration events were peri-procedural/anesthesia-related versus feeding-related. Therefore, mechanistic interpretations should be considered speculative. Recent peri-procedural studies reported no statistically significant increase in aspiration events associated with GLP-1 RA use; however, these studies examined associations in populations where full stomach precautions were commonly applied [34-37]. The same caveat applies to our findings: because our database lacks granular data on peri-procedural management, the observed lower incidence of aspiration pneumonia among GLP-1 RA initiators may partly reflect clinical preventive measures rather than a pharmacological effect of GLP-1 RA [38].
However, our results provide observational evidence suggesting that delayed gastric emptying does not necessarily translate to an increased risk of clinical aspiration pneumonia in this population. This finding is consistent with the conclusions of several recent systematic reviews and meta-analyses, which, despite confirming that GLP-1 RA delay gastric emptying, did not demonstrate a statistically significant association between their use and the clinical incidence of aspiration pneumonia in patients undergoing elective surgery or endoscopic procedures. Our study extends this observation from the short-term perioperative setting to long-term outpatient follow-up in a high-risk population, further contributing this perspective to the existing body of observational evidence [37].
Several hypotheses could potentially explain this unexpected association, though these remain speculative. Pneumonia is fundamentally an inflammatory lung injury, and its risk and severity depend not only on the aspiration event itself but also on the host's immune response. GLP-1 RA possess systemic anti-inflammatory properties, capable of modulating the immune response, suppressing the production of pro-inflammatory cytokines (e.g., TNF-α and IL-6), and inhibiting the NF-κB inflammatory pathway [39]. One hypothesis is that while GLP-1 RA may not reduce micro-aspiration events, their well-documented anti-inflammatory properties might theoretically reduce the likelihood of such events progressing to clinically significant pneumonia. However, this remains highly speculative, and the exact mechanisms underlying our observed association require further investigation through dedicated mechanistic studies. This observation suggests that the theoretical concerns regarding GLP-1 RA-related aspiration risk may not translate into clinical harm in this population, though this interpretation requires validation in future studies.
4.4 Interpretation of the Neutral MACE Outcome
Our neutral MACE findings contrast with meta-analyses showing 12-14% MACE reduction with GLP-1 RA versus placebo and superiority over DPP-4i [29, 31]. Three factors explain this: First, using active comparator versus placebo reduces effect size. DPP-4i, though neutral in CVOTs [39], increase endogenous GLP-1 levels [40, 41] and outperform sulfonylureas. Second, high competing non-cardiovascular mortality (131 deaths vs. 136 MACE in DPP-4i group) limits MACE detection in frail populations. Third, insufficient power with 364 patients per group and < 2 years follow-up challenged detecting modest differences. The wide confidence interval (0.79-1.28) supports this.
4.5 Subgroup Analyses and Mechanistic Plausibility
The association with lower all-cause mortality was consistent across most pre-specified subgroups, supporting the consistency of the primary finding. Notably, this association appeared more pronounced in patients with a history of heart failure (aHR: 0.43, 95% CI: 0.27-0.69).
This finding has an important clinical context. Previously, CVOTs of DPP-4i raised concerns about a potential increased risk of hospitalization for heart failure (HHF), particularly for saxagliptin, as reported in meta-analyses of DPP-4i trials [42], although subsequent trials with other agents yielded neutral results. In contrast, meta-analyses of GLP-1 RA have shown a modest but significant reduction in HHF risk [43]. Our observation of a notable association with reduced mortality in patients with a history of heart failure suggests that in a head-to-head comparison, the properties of GLP-1 RA on pathways relevant to heart failure (e.g., weight loss, blood pressure reduction, and potential direct myocardial effects) may be associated with more differential outcomes compared to the neutral effects of DPP-4i in this high-risk subgroup.
4.6 Exploration of MACE Outcomes in Younger Patients
Our exploratory analysis identified a potential signal for increased MACE risk among patients <65 years. This finding requires careful interpretation within the specific context of our post-gastrostomy population. While GLP-1 RA have established cardiovascular associations in the general adult population with type 2 diabetes [44], the interaction between age and underlying gastrointestinal pathology in determining cardiovascular risk remains poorly understood. The theoretical concerns about delayed gastric emptying and potential complications in patients with altered gastrointestinal anatomy [45, 46] may be particularly relevant in this subgroup. However, given the exploratory nature of this analysis, this finding should be considered hypothesis-generating, revealing an important knowledge gap that requires confirmation through targeted prospective research.
4.7 Strengths
This study addresses an important clinical question in a population systematically excluded from randomized controlled trials, using a target trial emulation framework with an active comparator design that reflects real-world treatment decisions. The extensive propensity score matching on multiple covariates represents a comprehensive attempt to address measured confounding. Several methodological approaches enhance the robustness of the analysis, including landmark analyses to assess temporal patterns, E-value calculations to quantify sensitivity to unmeasured confounding, and multiple sensitivity analyses including per-protocol approaches. The validation strategy using positive and negative controls (e.g., replicating known statin mortality benefits while showing no association with vitamin C) suggests the basic validity of the analytical platform and reduces concerns about systematic database bias.
4.8 Limitations
Critical limitations include: First, despite extensive PSM, unmeasured confounding remains likely. Unavailable variables include gastrostomy indication, functional status, nutritional parameters, frailty scores, and disease severity (e.g., cancer staging). These factors are fundamental determinants of outcomes in post-gastrostomy patients and may be strongly associated with treatment selection. Specifically, the database lacks structured information on the indication for gastrostomy, neurologic disease severity, functional/frailty status, swallowing function assessments, cancer staging, and goals of care, all of which may strongly influence both treatment selection and outcomes. Clinicians may preferentially prescribe GLP-1 RA to more clinically stable patients, introducing confounding by indication that propensity score matching cannot fully address. Although the E-value (2.17 for the point estimate; 3.11 for the confidence interval limit) suggests that a moderately strong unmeasured confounder would be needed to nullify the observed association, this threshold could plausibly be exceeded by unmeasured factors in this complex population.
Second, our study design limitations significantly impact the interpretation of secondary endpoints. With 364 patients per group and median follow-up of less than 2 years, the study was designed primarily for the mortality endpoint. The wide confidence intervals for MACE outcomes (0.79-1.28 for overall analysis) reflect substantial uncertainty and limit our ability to draw definitive conclusions about cardiovascular safety, particularly given the active comparator design where smaller effect sizes would be expected.
Third, our subgroup analyses were exploratory and should be considered hypothesis-generating rather than confirmatory. The potential MACE signal in patients < 65 years (aHR: 1.75, 95% CI: 1.05-2.92) occurred in the context of multiple subgroup comparisons, increasing the likelihood that this represents a chance finding rather than a true effect.
Fourth, the high competing mortality risk in our population (131 deaths vs. 136 MACE events in the DPP-4i group) may mask true associations with non-fatal outcomes. Although composite endpoints were used to partially address this bias, traditional competing-risk analyses (e.g., Fine-Gray subdistribution hazard models) were not feasible due to platform constraints described in the Methods section.
Fifth, outcome misclassification is an inherent limitation of database studies. Our reliance on ICD-10 coding for complex outcomes such as aspiration pneumonia and MACE lacks clinical validation or adjudication. The counterintuitive finding of lower pneumonia risk with GLP-1 RA use may partly reflect coding inconsistencies rather than true clinical benefit. For aspiration pneumonia in particular, reliance on diagnostic codes and the inability to adjudicate peri-procedural versus feeding-related timing may contribute to misclassification and differential ascertainment.
Finally, the generalizability of findings from this highly selected population to broader clinical practice remains uncertain. Post-gastrostomy patients represent a unique subset of the diabetes population, and our findings may not apply to other clinical contexts.
5. Conclusion
This target trial emulation found that GLP-1 RA initiation, compared with DPP-4i, was associated with lower all-cause mortality and aspiration pneumonia risk. However, given the limited sample size, these results warrant confirmation in prospective studies.
Supplementary Material
Supplementary figure and tables.
Acknowledgements
Funding
This study received funding support from Chang Bing Show Chwan Memorial Hospital (Grant No. BRD-114003).
Ethics Approval and Consent to Participate
The Institutional Review Board of Show Chwan Memorial Hospital granted approval for this study protocol (IRB No: 1141007) and waived the requirement for informed consent. This determination was made because the research was conducted using the TriNetX global collaborative health research network, which contains a large, federated database of de-identified electronic health records (EHRs). The data set's fully de-identified nature, attested to by a qualified expert as defined in the HIPAA Privacy Rule (§164.514(b)(1)), means the research is not considered human subject research.
Data Availability
The data source for this study was the TriNetX global collaborative health research network. This is a federated database of de-identified electronic health records that provides longitudinal clinical data, including patient demographics, diagnoses, procedures, laboratory results, and prescribed medications.
Declaration of Generative AI and AI-assisted Technologies in the Writing Process
During the preparation of this work the author(s) used Google's Gemini in order to improve the clarity, grammar, and readability of the manuscript text.
Author Contributions
Conceptualization: Y.-T. T., C.-H. C.; Data curation: Y.-T. T., C.-Y. L.; Formal analysis: Y.-T. T., C.-Y. L.; Funding acquisition: Y.-T. T., L.-P. C.; Investigation: Y.-T. T., C.-H. C., H.-Y. H., J.-W. W., S.-Y. C.; Methodology: Y.-T. T., C.-Y. L.; Resources: C.-Y. L.; Software: C.-Y. L.; Supervision: C.-Y. L.; Visualization: Y.-T. T.; Writing - original draft: Y.-T. T.; Writing - review & editing: C.-H. C., C.-Y. L.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Ma C, Ma X, Guan C, Li Y, Mauricio D, Fu S. Cardiovascular disease in type 2 diabetes mellitus: progress toward personalized management. Cardiovascular Diabetology. 2022;21:74
2. Tan X, Liang Y, Rajpura J, Yedigarova L, Noone J, Xie L. et al. Once-weekly glucagon-like peptide-1 receptor agonists vs dipeptidyl peptidase-4 inhibitors: cardiovascular effects in people with diabetes and cardiovascular disease. Cardiovascular Diabetology. 2023 22
3. Sachinidis A, Nikolić D, Stoian A, Papanas N, Tarar O, Rizvi A. et al. Cardiovascular outcomes trials with incretin-based medications: a critical review of data available on GLP-1 receptor agonists and DPP-4 inhibitors. Metabolism: clinical and experimental. 2020: 154343.
4. Giugliano D, Scappaticcio L, Longo M, Caruso P, Maiorino M, Bellastella G. et al. GLP-1 receptor agonists and cardiorenal outcomes in type 2 diabetes: an updated meta-analysis of eight CVOTs. Cardiovascular Diabetology. 2021 20
5. Chen X, Zhang X, Xiang X, Fang X, Feng S. Effects of glucagon-like peptide-1 receptor agonists on cardiovascular outcomes in high-risk type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabetology & Metabolic Syndrome. 2024 16
6. Sattar N, Lee M, Kristensen S, Branch K, Del Prato S, Khurmi N. et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. The lancet Diabetes & endocrinology. 2021
7. Khera R, Aminorroaya A, Dhingra L, Thangaraj P, Camargos AP, Bu F. et al. Comparative Effectiveness of Second-Line Antihyperglycemic Agents for Cardiovascular Outcomes: A Multinational, Federated Analysis of LEGEND-T2DM. Journal of the American College of Cardiology. 2024 84 10: 904-17
8. Huang D, He D, Gong L, Wang W, Yang L, Zhang Z. et al. Clinical characteristics and risk factors associated with mortality in patients with severe community-acquired pneumonia and type 2 diabetes mellitus. Critical Care. 2021 25
9. Bellary S, Kyrou I, Brown J, Bailey C. Type 2 diabetes mellitus in older adults: clinical considerations and management. Nature Reviews Endocrinology. 2021;17:534-48
10. Jalleh R, Plummer M, Marathe C, Umapathysivam M, Quast D, Rayner C. et al. Clinical Consequences of Delayed Gastric Emptying With GLP-1 Receptor Agonists and Tirzepatide. The Journal of Clinical Endocrinology and Metabolism. 2024;110:1-15
11. Van Zuylen M, Siegelaar S, Plummer M, Deane A, Hermanides J, Hulst A. Perioperative management of long-acting glucagon-like peptide-1 (GLP-1) receptor agonists: concerns for delayed gastric emptying and pulmonary aspiration. British journal of anaesthesia. 2024
12. Chen CH, Yan SL, Wang CH, Yeh YH, Chen CH, Lee YT. et al. Long-term risk associations of percutaneous endoscopic gastrostomy and nasogastric tubes: A population-based cohort study. Advances in Digestive Medicine. 2025: e13425.
13. Matthews A, Danaei G, Islam N, Kurth T. Target trial emulation: applying principles of randomised trials to observational studies. BMJ. 2022 378
14. Fu E. Target Trial Emulation to Improve Causal Inference from Observational Data: What, Why, and How? Journal of the American Society of Nephrology: JASN. 2023;34:1305-14
15. Tseng Y-T, Wang C-H, Wang J-D, Chen K-T, Li C-Y. Nonlinear associations of serum vitamin D levels with advanced liver disease and mortality: a US Cohort Study. Therapeutic Advances in Gastroenterology. 2025;18:17562848251338669
16. Kappenstein M, Von Bubnoff N. Real-World Electronic Medical Records Data Identify Risk Factors for Myelofibrosis and Can Be Used to Validate Established Prognostic Scores. Cancers. 2024 16
17. Gau S-Y, Lo S-W, Tu Y-F, Liao W-C, Su Y-J, Chang H-C. et al. Human Papillomavirus Infection Increases Risk of New-Onset Prurigo Nodularis: A Multi-Center Retrospective Cohort Study Using Global and US Electronic Medical Records of TriNetX network. Journal of the American Academy of Dermatology. 2025
18. Olbrich H, Kridin K, Hernández G, Zirpel H, Sadik CD, Terheyden P. et al. Increased cardiovascular risks and mortality in prurigo nodularis: a global cohort study. EBioMedicine. 2024 103
19. He Q, Gao F, Dukes O, Delany-Moretlwe S, Zhang B. Generalizing the Intention-to-Treat Effect of an Active Control from Historical Placebo-Controlled Trials: A Case Study of the Efficacy of Daily Oral TDF/FTC in the HPTN 084 Study. Journal of the American Statistical Association. 2024;119:2478-92
20. Romero-Gomez M, Lawitz E, Shankar R, Chaudhri E, Liu J, Lam R. et al. A Phase 2a active-comparator-controlled study to evaluate the efficacy and safety of efinopegdutide in patients with nonalcoholic fatty liver disease. Journal of hepatology. 2023
21. Wan F. Propensity Score Matching: should we use it in designing observational studies? BMC Medical Research Methodology. 2024 25
22. Pickett K, Suresh K, Campbell K, Davis S, Juarez-Colunga E. Random survival forests for dynamic predictions of a time-to-event outcome using a longitudinal biomarker. BMC Medical Research Methodology. 2021 21
23. Chung W, Chung K. Key concepts in clinical epidemiology: the use of the E-value for sensitivity analysis. Journal of clinical epidemiology. 2023
24. Lin H-T, Tsai Y-F, Liao P-L, Wei JC-C. Neurodegeneration and stroke after semaglutide and tirzepatide in patients with diabetes and obesity. JAMA Network Open. 2025;8:e2521016
25. Pan H-C, Chen J-Y, Chen H-Y, Yeh F-Y, Huang TT-M, Sun C-Y. et al. Sodium-glucose cotransport protein 2 inhibitors in patients with type 2 diabetes and acute kidney disease. JAMA Network Open. 2024;7:e2350050
26. Kelly A, Carlson K, Wallace Z, Putman M. Positive and negative controls in rheumatology research. Rheumatology. 2024
27. Horner M, Ogdie A, Orroth K, Kent ST, Tran K, Deignan C. et al. Implementing Negative Control Outcomes to Assess Comparability of Treatments for Psoriasis and Psoriatic Arthritis. Pharmacoepidemiology and Drug Safety. 2025 34
28. De Vries BP, Groenwold R. Negative controls: Concepts and caveats. Statistical Methods in Medical Research. 2023;32:1576-87
29. Sattar N, Lee MM, Kristensen SL, Branch KR, Del Prato S, Khurmi NS. et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. The lancet Diabetes & endocrinology. 2021;9:653-62
30. Giugliano D, Longo M, Signoriello S, Maiorino MI, Solerte B, Chiodini P. et al. The effect of DPP-4 inhibitors, GLP-1 receptor agonists and SGLT-2 inhibitors on cardiorenal outcomes: a network meta-analysis of 23 CVOTs. Cardiovascular diabetology. 2022;21:42
31. Kristensen SL, Rørth R, Jhund PS, Docherty KF, Sattar N, Preiss D. et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. The lancet Diabetes & endocrinology. 2019;7:776-85
32. Drucker DJ. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell metabolism. 2018;27:740-56
33. Yang X, Sun H, Hou S, Zhang W, Meng H. The Impact of Nutritional and Inflammatory Status on Mortality in Stroke Patients: Results from NHANES 2005-2018. Journal of stroke and cerebrovascular diseases: the official journal of National Stroke Association. 2025: 108334.
34. Kindel TL, Wang AY, Wadhwa A, Schulman AR, Sharaiha RZ, Kroh M. et al. Multisociety Clinical Practice Guidance for the Safe Use of Glucagon-like Peptide-1 Receptor Agonists in the Perioperative Period. Clin Gastroenterol Hepatol. 2024
35. Ushakumari DS, Sladen RN. ASA consensus-based guidance on preoperative management of patients on glucagon-like peptide-1 receptor agonists. Anesthesiology. 2023
36. Alkabbani W, Suissa K, Gu KD, Cromer SJ, Paik JM, Bykov K. et al. Glucagon-like peptide-1 receptor agonists before upper gastrointestinal endoscopy and risk of pulmonary aspiration or discontinuation of procedure: cohort study. bmj. 2024 387
37. Baig MU, Piazza A, Lahooti A, Johnson KE, Rangwani S, Gouda Z. et al. Glucagon-like peptide-1 receptor agonist use and the risk of residual gastric contents and aspiration in patients undergoing GI endoscopy: a systematic review and a meta-analysis. Gastrointestinal Endoscopy. 2025;101:762-71 e13
38. Joshi GP, Abdelmalak BB, Weigel WA, Soriano SG, Harbell M, Kuo CI. et al. American Society of Anesthesiologists consensus-based guidance on preoperative management of patients (adults and children) on glucagon-like peptide-1 (GLP-1) receptor agonists. Last updated: June. 2023 29
39. Razavi M, Wei Y-Y, Rao X-Q, Zhong J-X. DPP-4 inhibitors and GLP-1RAs: cardiovascular safety and benefits. Military Medical Research. 2022;9:45
40. Smits M, Galsgaard K, Jepsen S, Albrechtsen N, Hartmann B, Holst J. In vivo inhibition of dipeptidyl peptidase 4 allows measurement of GLP-1 secretion in mice. Diabetes. 2024
41. Gaggini M, Sabatino L, Suman AF, Chatzianagnostou K, Vassalle C. Insights into the Roles of GLP-1, DPP-4, and SGLT2 at the Crossroads of Cardiovascular, Renal, and Metabolic Pathophysiology. Cells. 2025 14
42. Mannucci E, Nreu B, Montereggi C, Ragghianti B, Gallo M, Giaccari A. et al. Cardiovascular events and all-cause mortality in patients with type 2 diabetes treated with dipeptidyl peptidase-4 inhibitors: An extensive meta-analysis of randomized controlled trials. Nutrition, metabolism, and cardiovascular diseases: NMCD. 2021
43. Banerjee M, Maisnam I, Mukhopadhyay S. Impact of Heart Failure History at Baseline on Cardiovascular Effects of GLP-1 Receptor Agonists in Type 2 Diabetes: a Meta-analysis. Cardiovascular Drugs and Therapy. 2023:1-8
44. Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P. et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. The Lancet. 2019;394:121-30
45. Kotecha P, Huang W, Yeh Y-Y, Narvaez VM, Adirika D, Tang H. et al. Efficacy and Safety of GLP-1 RAs in Children and Adolescents with Obesity or Type 2 Diabetes: A Systematic Review and Meta-Analysis. JAMA pediatrics. 2025
46. Tarar Z, Farooq U, Chaudhry A, Gandhi M, Alayli AE, Ayoub M. et al. Evidence Report on the Safety of Gastrointestinal Endoscopy in Patients on Glucagon-like Peptide-1 Receptor Agonists: A Systematic Review and Meta-Analysis. Diagnostics. 2025 15
Author contact
Corresponding author: Chung-Yi Li, Department of Public Health, College of Medicine, National Cheng Kung University, No.1, University Rd., Tainan, Taiwan, 701. Phone: 886-6-2353535 ext.5862. Email: cyli99ncku.edu.tw.

 Global reach, higher impact
Global reach, higher impact