Impact Factor ISSN: 1449-1907
Int J Med Sci 2024; 21(3):492-495. doi:10.7150/ijms.84590 This issue Cite
Research Paper
Prophylactic effect of pilocarpine on acute sialadenitis following radioactive iodine therapy in thyroid cancer patients
1. Department of Radiology, Incheon St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea.
2. Department of Surgery, Incheon St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea.
3. Division of Endocrinology & Metabolism, Department of Internal Medicine, Incheon St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Republic of Korea.
Abstract
Purpose: Our aim was to evaluate the effect of prophylactic pilocarpine on acute salivary symptoms after radioactive iodine (RAI) therapy in patients with differentiated thyroid cancer.
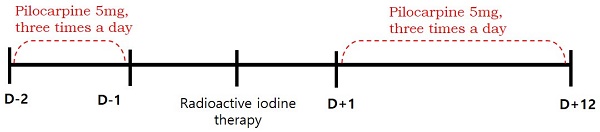
Methods: We enrolled 88 patients (76 women and 12 men; mean age: 47 years; range: 20-74 years) with differentiated thyroid cancer who received RAI. Patients were divided into pilocarpine (51 patients) and control (37 patients) groups. Pilocarpine was given orally, at a dose of 5 mg three times a day, from 2 days before and 12 days after RAI therapy. Symptoms and signs of acute sialadenitis within 3 months of RAI therapy were recorded.
Results: During the 3 months after RAI therapy, 13 of the 88 patients (14.7%) developed acute symptomatic sialadenitis (swelling or pain of salivary glands). Acute salivary symptoms were reported by 4 (7.8%) and 9 (24.3%) patients in the pilocarpine and control groups, respectively. Acute salivary symptoms were less frequent in the pilocarpine than control group (p = 0.04), but did not differ by age, sex, or RAI dose (p = 0.3357, p = 0.428, and p = 0.2792).
Conclusions: Pilocarpine reduced the likelihood of acute sialadenitis after RAI therapy in patients with differentiated thyroid cancer.
Keywords: Thyroid Neoplasms, Pilocarpine, Sialadenitis, Pre-Exposure Prophylaxis, Radioactive Iodine

 Global reach, higher impact
Global reach, higher impact