Impact Factor ISSN: 1449-1907
Int J Med Sci 2022; 19(10):1615-1627. doi:10.7150/ijms.73800 This issue Cite
Research Paper
Clustering of Chromatin Remodeling Enzymes Predicts Prognosis and Clinical Benefit of Therapeutic Strategy in Pancreatic Cancer
1. Graduate Institute of Clinical Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung 807, Taiwan.
2. Department of Internal Medicine, Division of Hematology and Oncology, Kaohsiung Medical University Hospital, Kaohsiung Medical University, Kaohsiung 807, Taiwan.
3. Faculty of Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung 807, Taiwan.
4. Department of Internal Medicine, Kaohsiung Medical University Hospital, Kaohsiung, Taiwan.
5. Department of Pathology, Kaohsiung Medical University Hospital, Kaohsiung Medical University, Kaohsiung 807, Taiwan.
6. National Institute of Cancer Research, National Health Research Institutes, Tainan, Taiwan.
7. Division of Breast Oncology and Surgery, Department of Surgery, Kaohsiung Medical University Hospital, Kaohsiung 807, Taiwan.
8. Specialist Nurse and Surgical Nurse Practitioner Office, Kaohsiung Medical University Chung-Ho Memorial Hospital.
9. Translational Research Center, Kaohsiung Medical University Hospital, Kaohsiung, Taiwan.
10. Center of Cancer Program Development, E-Da Cancer Hospital, I-Shou University, Kaohsiung 807, Taiwan.
11. Drug Development and Value Creation Research Center, Kaohsiung Medical University, Kaohsiung, Taiwan.
12. Department of Medical Research, Kaohsiung Medical University Hospital.
* These authors contributed equally to this work.
Abstract
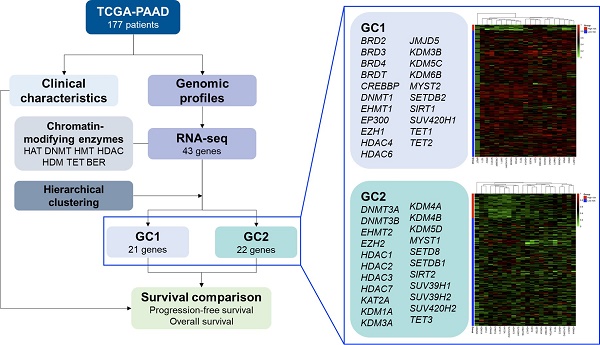
In recent years, translational research and pharmacological targeting of epigenetic modifications have become the focus of personalized therapy for patients with pancreatic cancer. Preclinical and clinical trials targeting post-translational modifications have been evaluated as monotherapy or in combination with standard chemotherapy. In this study, we selected 43 genes from seven families of chromatin-modifying enzymes and investigated the influences of epigenetic modifications and their interactions on pancreatic ductal adenocarcinoma (PDAC) using hierarchical clustering analysis. Our analysis also evaluated their effects on treatment modalities and regimens of chemotherapy for PDAC. RNA-seq data for a total of 177 patients with pancreatic cancer, obtained from The Cancer Genome Atlas database, were analyzed. Our results suggested that high-risk patients of survival significant chromatin remodeling-associated gene cluster (gene cluster 2), composed of histone methyltransferases, histone acetyltransferases, histone deacetylases, histone demethylases, and 10-11 translocation family, demonstrated inferior progression-free survival and overall survival in patients with PDAC, especially in men. Our novel biomarker, survival significant chromatin remodeling-associated gene cluster, showed superior prediction performance compared with the conventional TNM system. Overall, these findings suggest that epigenetic modifications and interactions play an important role in the prognosis and therapeutic response of patients with PDAC.
Keywords: Epigenetic modification, Pancreatic Cancer, Hierarchical clustering analysis

 Global reach, higher impact
Global reach, higher impact