Impact Factor ISSN: 1449-1907
Int J Med Sci 2018; 15(10):986-991. doi:10.7150/ijms.24715 This issue Cite
Short Research Communication
Regulation of TLR4 in silica-induced inflammation: An underlying mechanism of silicosis
1. Nethersole School of Nursing, The Chinese University of Hong Kong, Shatin, Hong Kong
2. Department of Orthopaedics and Traumatology, The Chinese University of Hong Kong, Shatin, Hong Kong
3. School of Life Sciences, The Chinese University of Hong Kong, Shatin, Hong Kong
4. Grantham Hospital, Aberdeen, Hong Kong
5. School of Biomedical Sciences, The Chinese University of Hong Kong, Shatin, Hong Kong
Received 2018-1-3; Accepted 2018-4-12; Published 2018-6-14
Abstract
Silicosis is an incurable lung disease affecting millions of workers in hazardous occupations. It is caused by chronic exposure to the dust that contains free crystalline silica. Silica-induced lung damage occurs by several main mechanisms including cell death by apoptosis, fibrosis and production of cytokines. However, the signal pathways involved in these mechanisms are not fully characterized. In this study, the toll-like receptor 4 (TLR4)-related signal pathway was examined in silica-treated U937-differentiated macrophages. The expression level of TLR4 was measured by both quantitative PCR and Western blot. Confirmation of the involvement of MyD88/TIRAP and NFκB p65 cascade was performed by Western blot. The secretion of cytokines IL-1β, IL-6, IL-10 and TNFα was measured by enzyme-linked immunosorbent assay. Our results showed that TLR4 and related MyD88/TIRAP pathway was associated with silica-exposure in U937-differentiated macrophages. Protein expression of TLR4, MyD88 and TIRAP was upregulated when the U937-differentiated macrophages were exposed to silica. However, the upregulation was attenuated when TLR4 inhibitor, TAK-242 was present. At different incubation times of silica exposure, it was found that NFκB p65 cascade was activated at 10-60 minutes. Release of cytokines IL-1β, IL-6, IL-10 and TNFα was induced by silica exposure and the induction of IL-1β, IL-6 and TNFα was suppressed by the addition of TAK-242. In conclusion, our study demonstrated that TLR4 and related MyD88/TIRAP pathway was involved in silica-induced inflammation in U937-differentiated macrophages. Downstream NFκB p65 cascade was activated within 1 hour when the U937-differentiated macrophages were exposed to silica. The better understanding of early stage of silica-induced inflammatory process may help to develop earlier diagnosis of silicosis.
Keywords: Silica, Macrophage, Toll-like receptor, Inflammation
Introduction
Silicosis is an irreversible and incurable lung disease. It is a worldwide occupational disease problem affecting more than 35 million workers in the world [1,2]. Crystalline silica exposure is found to be instrumental for the development of silicosis. Crystalline silica in the form of quartz is the most commonly occurring polymorph. It is the major component of the concrete and constitutes 10 - 40 % components in granite [2], a rock found commonly in the landscape and construction sites.
Studies show that upon chronic and acute exposures to crystalline silica, these foreign mineral will be phagocytized by alveolar macrophages and trigger pro-inflammatory events such as cytokine modulation. Followed by the inflammatory events, the fibrotic processes such as production of growth factors will be initiated, which will finally determine the development of silicosis [3]. The mechanism of crystalline silica induced silicosis is not yet fully understood. Studies on apoptosis, however, reveal that the crystalline silica also capable of inducing apoptosis and generation of reactive oxygen species (ROS) and reactive nitrogen species (RNS) [4,5]. The generation of oxidants by silica particles and by silica-activated cells results in cell and lung damage; increased expression of inflammatory cytokines, including TNF-alpha, IL 1 beta, and TGF-beta; activation of cell signaling pathways, including the MAP kinase pathways; and phosphorylation and activation of specific transcription factors (e.g., NFκB) [6,7]. Among these various studies, one key pro-inflammatory event is the formation of the NLR family, pyrin domain containing 3 inflammasome upon crystalline silica exposure. Inflammasome activation and caspase-1 mediated events had been also shown in myeloid cells [8], silicotic rat lung and non-tumorigenic human bronchial epithelial cells [9]. The crystalline silica-induced activation of NLRP3 inflammasome is proved to be important in the caspase-1 (CASP1) mediated activation of pro-inflammatory cytokines (such as IL-1β and IL-18) and the release of growth factors as well as alarmins including basic fibroblast growth factor (bFGF) and high mobility group protein B1 (HMGB1) respectively [9,10]. Using a MyD88 knockout mice model, it was demonstrated that the MyD88-signaling pathway mobilized during the lung responses to silica particles organizes granuloma formation, Th17-associated inflammatory responses and neutrophil accumulation; three major features of silicosis [11].
Silica dust can provoke inflammatory events and cellular damage in alveolar macrophages, however, the molecular events behind the first site of attack are complex and not completely elucidated. In this study, we aim to demonstrate the regulation of toll-like receptor 4 (TLR4) and related signal pathway in the silica-induced inflammatory responses.
Materials and Methods
Cell culture
U937 cell line was purchased from American Type Culture Collection. It is a pro-monocytic, human myeloid leukaemia cell line that can be induced to differentiate into macrophages. U937 cells were maintained in suspension culture in RPMI-1640 (Invitrogen Corporation, California, USA) supplemented with 1% Penicillin and Streptomycin solution and 10% FBS (Invitrogen Corporation, California, USA). Culture was maintained at 37°C in 5% CO2 incubator and sub-culture was done 2-3 times per week. The cells were induced to differentiate into macrophages by exposing the cells to 10 ng/ml of phorbol 12-myristate 13-acetate (PMA) (Sigma-Aldrich, Missouri, USA) for 48 h. After that, the medium was replaced with fresh medium and incubated for further 24 h to complete the differentiation.
Silica exposure
Quartz silica powder was purchased from Sigma-Aldrich Corporation (Missouri, USA). The size is 0.5 - 10 µm (~80% between 1 - 5 µm) with purity ~99%. Silica powder was sterilized by heating in 160°C oven for 2 h. Silica powder was suspended in RPMI-1640 medium with 1.25% FBS and sonicated for 10 minutes before added into the U937-differentiated macrophage at concentration 2.5 µg/cm2 and incubated for different durations for different experiments [12].
Inhibition of TLR4
TLR4 expression in U937-differentiated macrophages was inhibited by adding 5 µM TAK-242 solution into the medium and incubated the cells for 1 h before exposed to silica.
Quantitative PCR
Quantitative PCR (qPCR) to detect the expression level of TLR4 gene was performed using SuperScript first-strand cDNA synthesis and RT-PCR Kit with SYBR Green (Thermo Fisher, USA) on Vii 7 Real-Time PCR Detection System (Applied Biosystem, USA) according to previous study [13]. Total RNA amount in each sample was normalized by expression level of β-actin. The primer sequence for qPCR against TLR4 and β-actin were TLR4: 5'TGGGAGCCTTTTCTGGACTAT3' (forward) 5'TTGATGTAGAACCCGCAAGTC3' (reverse) and β-actin: 5'ctcttccagccttccttcct3' (forward) 5'AGCACTGTGTTGGCGTACAG3' (reverse).
Western blot analysis
Protein expression level of TLR4 and related pathway regulators was examined by Western blot analysis [14]. Total protein was collected from the U937-differentiated macrophages with appropriate silica treatment and incubation time. Then, 25 µg of protein was electrophoresed in 12% SDS-polyacrylamide gel and the resolved protein was transferred onto PVDF membranes. After that, the membrane was blocked using 10% non-fat milk and incubated with primary antibody of TLR4, MyD88, TIRAP, p-NFκB p65, p-IκBα, p-IKKα/β and β-actin respectively. The antigen-antibody complex was detected with horseradish peroxidase (HRP)-conjugated secondary antibodies using an enhanced chemiluminescence detection system. The image was finally captured onto an X-ray film.
Measurement of cytokines
The U937-differentiated macrophages were treated appropriately with silica in the presence or absence of TAK-242. After incubation for 24 h, the medium was collected and centrifugation was proceeded for 5 min at 14,000 x g to remove the cell debris. For cytokine measurement, enzyme-linked immunosorbent assay (ELISA) was performed to measure the amount of IL-1β, IL-6, IL-10 and TNFα in the culture supernatant. Briefly, ELISA plate was pre-coated with 100 μL of capture antibody. The plate was then blocked with 10% fetal bovine serum for 1 h before the addition of cytokine standard or sample supernatants. After further incubation for 3 h, the wells were washed and detection reagent (detection antibody + avidin-HRP reagent) was added. After 1 h incubation, the wells were washed again and substrate was added. Stop solution was used to terminate the reaction when appropriate signal was developed. Finally, absorbance at 450 nm was recorded using microplate reader (R&D Systems, Inc.).
Statistical analysis
One Way ANOVA with Dunnett's Multiple Comparison Test was used to analyze the statistical significance of the results. All experimental results were expressed as mean ± standard deviation (SD). The difference was statistically significant when * p < 0.05 or ** p < 0.01.
Results
Effects of silica exposure on TLR4 expression
Silica (2.5 mg/cm2) was added to the U937-differentiated macrophages and incubated for various time intervals (0, 0.5, 2, 8, 16, 24 h) before RNA was harvested and real-time PCR was performed. The results in Fig. 1 showed that relative mRNA expression level of TLR4 was significantly upregulated at 16 (1.96 fold) and 24 h (3.79 fold) when compared to 0 h.
Relative mRNA expression level of TLR4 was detected by qPCR. Fold of change of TLR4 mRNA expression level was measured at different time intervals after silica exposed to U937-differentiated macrophages when compared to time 0 (which is set as 1). Total RNA amount of each sample was normalized by expression level of β-actin. Data was expressed as mean ± standard deviation (S.D.) of 3 replicates.
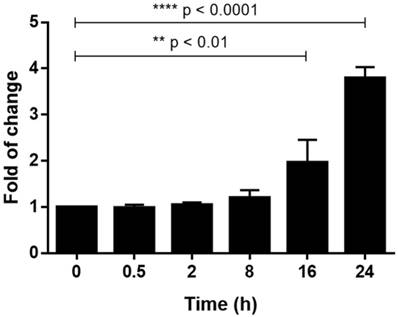
Effects of silica on TLR4 signal pathway in U937-differentiated macrophages
After the U937-differentiated macrophages were exposed to silica for 24 h with or without pre-incubation of TAK-242, total protein was harvested and the expression level of TLR4, MyD88 and TIRAP was detected by Western blot. The results in Fig. 2 showed that silica exposure apparently upregulated the expression of TLR4 and its related pathway regulators MyD88 and TIRAP. When TLR4 inhibitor TAK-242 was added before the addition of silica, upregulation of TLR4, MyD88 and TIRAP were attenuated.
Detection of expression level of TLR4, MyD88 and TIRAP by Western blot analysis. U937-differentiated macrophages were exposed to silica and incubated for 24 h in the presence or absence of pre-incubation of TAK-242 before total protein was harvested. Then, the protein expression level of TLR4, MyD88 and TIRAP was detected by primary antibodies and respective secondary antibodies followed by enhanced chemiluminescence detection and image capture by X-ray film. Total protein amount of each sample was normalized by expression of β-actin. The image was a representative of three independent trials.
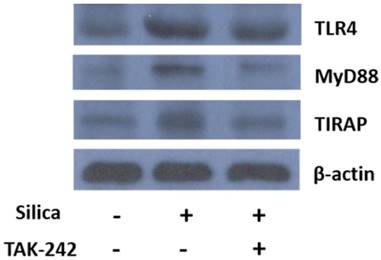
Effects of silica on NFκB p65 inflammatory pathway
Detection of protein level of p-NFκB p65 and its related regulators p-IκBα and p-IKKα/β at different time intervals after silica exposure was done by Western blot. The results in Fig. 3 showed that when silica was added to the U937-differentiated macrophages for 10 minutes, the expression of phosphorylated form of NFκB p65, IκBα and IKKα/β were apparently increased and the upregulation was lasted up to 60 minutes.
Detection of expression level of p-NFκB p65, p-IκBα and p-IKKα/β by Western blot analysis. U937-differentiated macrophages were exposed to silica and incubated for 5, 10, 30 and 60 min, respectively, before total protein was harvested. Then, the protein expression level of phosphorylated form of NFκB p65, IκBα and IKKα/β was detected by primary antibodies and respective secondary antibodies followed by enhanced chemiluminescence detection and image captured by X-ray film. Total protein amount of each sample was normalized by expression of β-actin. The image was a representative of three independent trials.

Effects of TLR4 inhibition on cytokine secretion
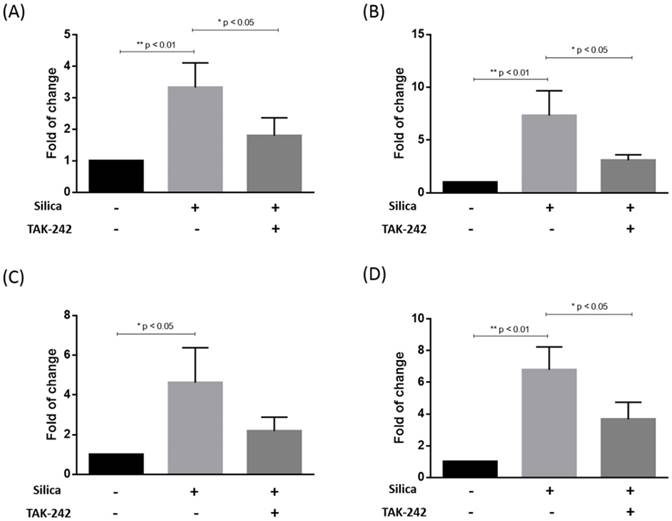
The release of IL-1β, IL-6, IL-10 and TNFα into the medium by the U937-differentiated macrophages was detected by ELISA after silica exposure for 24 h. From the results in Fig. 4, it could be seen that the secretion of all of four cytokines were significantly increased when silica was added to the U937-differentiated macrophages for 24 h. However, when the cells were pre-treated with TAK-242 before silica exposure, the production of IL-1β, IL-6 and TNFα were significantly inhibited when compared to silica treatment alone. The amount of both IL-10 was also apparently decreased although the difference was not statistically significant.
Release of IL-1β, IL-6, IL-10 and TNFα was measured by ELISA. After the U937-differentiated macrophages were exposed to silica for 24 h in the presence or absence of TAK-242 pre-treatment, the release of IL-1β (A), IL-6 (B), IL-10 (C) and TNFα (D) was measured by ELISA. Data was expressed as mean ± standard deviation (S.D.) of 3 replicates. Significant difference was indicated by * p < 0.05 and ** p < 0.01.
Discussion
Excessive exposure to crystalline silica results in adverse health effects for millions of workers. Silicosis, an irreversible and incurable disease, is the most consequential adverse health effect associated with silica exposure. To understand the underlying mechanisms of silicosis, global gene expression profiling by employing microarray, or newer sequencing platforms, and bioinformatic analysis of gene expression data were utilized [15,16,17]. However, there are still some missing gaps in this aspect.
Our present study revealed that silica-induced inflammation was regulated via TLR4 signal pathway. Toll-like receptor (TLR) family were located on the plasma membrane or endosomal vesicles. They are mainly expressed in immune cells such as macrophages, monocytes, dendritic cells, etc. and play important roles in sensing and responding to pathogens [18]. In this study, TLR4, and the related myeloid differentiation factor 88 (MyD88) and toll-interleukin 1 receptor domain containing adaptor protein (TIRAP) was activated by exposing U937-differentiated macrophages to silica for 24 h. Upregulation of TLR4 was detected both by real-time PCR and Western blot (Figure 1 & 2). The MyD88-dependent pathway is regulated by two adaptor-associated proteins: MyD88 and TIRAP. From the results shown in Fig. 2, silica exposure activated both MyD88 and TIRAP. The activation was inhibited when TLR4 inhibitor, TAK-242, was added together. MyD88/TIRAP cascade regulates NFκB p65 activation and finally to cytokine production. By Western blot analysis, the activation of NFκB p65 cascade by silica exposure was shown in Fig. 3. Phosphorylated form of NFκB p65, IκBα and IKKα/β were all upregulated when U937-differentiated macrophages exposed to silica for 10-60 minutes. TAK-242, also called resatorvid, is a small molecule specifically inhibiting TLR4 signaling by binding to the intracellular domain of TLR4. It interferes with protein-protein interactions between TLR4 and its adaptor molecules [19]. In the current study, TAK-242 inhibited TLR4 and the associated MyD88/TIRAP dependent pathway. This further confirms the involvement of TLR4-related signaling pathway in U937-differentiated macrophages when exposed to silica.
In this study, U937-differentiated macrophages were used as cell model. Indeed, preliminary experiments were done on two well-established human macrophage cell lines: differentiated-U937 and -THP-1. It was found that the cytotoxicity of silica on both differentiated U937 and THP-1 were similar, i.e. 20% cytotoxicity was induced by 2.5 µg/cm2 silica at 24 h incubation [12]. In addition, mRNA expression of TLR4 was upregulated in differentiated THP-1 when the cells were exposed to silica at 2.5 µg/cm2 for 24 h (data not shown). After that, U937-differentiated macrophage was selected to further examine the underlying pathway because they are easier to use, virtually unlimited number of cells can prepared, and they are relatively uniform [20].
Our present study reported the association of TLR4 with silica-induced inflammation in U937-differentiated macrophage. In other studies, TLR4 was also reported to be involved in other kinds of inflammatory responses. For example, TLR4 was activated by the agonists, such as LPS in Leydig cells, and subsequently induce the production of inflammatory factors. LPS binds to the Toll-like receptor 4 (TLR-4) located on the basolateral membranes of enterocytes and plasma membrane of circulating immune cells. [21,22]. The activation of TLR-4 receptor complex sets forth a diverse array of intracellular signaling pathways that determine the magnitude, type, and duration of the inflammatory response [23].
Given the diversity of pathologies associated with silica exposure, it is unlikely that one common mechanism is responsible for all of the possible diseases. For silicaosis, it is generally accepted that the alveolar macrophage is a relevant cell type to study. Researchers studying the alveolar macrophage /silica particle interaction have several explanations on how silica is toxic to alveolar macrophage. For example, producing reactive oxygen species (ROS) and triggering cell-signaling pathways initiating cytokine release and apoptosis [24] or silica-binding receptors are involved in inflammation regulation and normal immune function [25,26]. More than one mechanism may explain the silica-induced damage to alveolar macrophages. This explained why there is a diversity of pathologies linked to silica exposure and inhalation.
In conclusion, our work demonstrated that silica induced inflammation in U937-differentiated macrophages was mediated by TLR4/MyD88/TIRAP signal pathway and the activation of NFκB p65 cascade further induced the release of IL-1β, IL-6, IL-10 and TNFα. This help to fill some missing gaps in the mechanisms of silicosis.
Acknowledgements
This study was supported by Pneumoconiosis Compensation Fund Board.
Competing Interests
The authors have declared that no competing interest exists.
References
1. Leung CC, Yu IT, Chen W. Silicosis. Lancet. 2012;379:2008-2018
2. Minnesota Department of Labor, Industry Occupational Safety, Health Division (Minnesota OSH). Health Considerations for Workplace Exposure to Silica. August. 2012
3. Fujimura N. Pathology and pathophysiology of pneumoconiosis. Curr Opin Pulm Med. 2000;6:140-144
4. Fubini B, Hubbard A. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) generation by silica in inflammation and fibrosis. Free Radic Biol Med. 2003;34:1507-1516
5. Joshi GN, Knecht DA. Silica phagocytosis causes apoptosis and necrosis by different temporal and molecular pathways in alveolar macrophages. Apoptosis. 2013;18:271-285
6. Shen HM, Zhang Z, Zhang QF. et al. Reactive oxygen species and caspase activation mediate silica-induced apoptosis in alveolar macrophages. Am J Physiol Lung Cell Mol Physiol. 2001;280:L10-17
7. Rimal B, Greenberg AK, Rom WN. Basic pathogenetic mechanisms in silicosis: current understanding. Curr Opin Pulm Med. 2005;11(2):169-73
8. Rathinam VA, Vanaja SK, Fitzgerald KA. Regulation of inflammasome signaling. Nat. Immunol. 2012;13:333-342
9. Peeters PM, Eurlings IM, Perkins TN. et al. Silica-induced NLRP3 inflammasome activation in vitro and in rat lungs. Part Fibre Toxicol. 2014;11:58
10. Rastrick J, Birrell M. The role of the inflammasome in fibrotic respiratory diseases. Minerva Med. 2014;105:9-23
11. Re SL, Giordano G, Yakoub Y. et al. Uncoupling between inflammatory and fibrotic responses to silica: evidence from MyD88 knockout mice. PLoS One. 2014;9:e99383
12. Chan JYW, Tsui JCC, Law PTW. et al. RNA-Seq revealed ATF3-regulated inflammation induced by silica. Toxicology. 2018;393:34-41
13. Chan JY, Tsui JC, Law PT. et al. Profiling of the silica-induced molecular events in lung epithelial cells using the RNA-Seq approach. J Appl Toxicol. 2017;37:1162-1173
14. Wang L, Chan JY, Zhou X. et al. A novel agent enhances the chemotherapeutic efficacy of doxorubicin in MCF-7 breast cancer cells. Front Pharmacol. 2016;7:249
15. Huang YC. The role of in vitro gene expression profiling in particulate matter health research. J Toxicol Environ Health. 2013;16:381-394
16. Sellamuthu R, Umbright C, Li S. et al. Mechanisms of crystalline silica-induced pulmonary toxicity revealed by global gene expression profiling. Inhal Toxicogical. 2011;23:927-937
17. Sellamuthu R, Umbright C, Roberts JR. et al. Transcriptomics analysis of lungs and peripheral blood of crystalline silica-exposed rats. Inhal Toxicogical. 2012;24:570-579
18. Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010;11:373-384
19. Matsunaga N, Tsuchimori N, Matsumoto T. et al. TAK-242 (resatorvid), a small-molecule inhibitor of Toll-like receptor (TLR) 4 signaling, binds selectively to TLR4 and interferes with interactions between TLR4 and its adaptor molecules. Mol Pharmacol. 2011;79:34-41
20. Chanput W, Peters V, Wichers H. (2015) THP-1 and U937 Cells. Verhoeckx K. et al (eds) The Impact of Food Bioactives on Health Springer, Cham
21. Guo S, Al-Sadi R, Said HM. et al. Lipopolysaccharide causes an increase in intestinal tight junction permeability in vitro and in vivo by inducing enterocyte membrane expression and localization of TLR-4 and CD14. Am J Pathol. 2013;182:375-387
22. Tomita M, Ohkubo R, Hayashi M. Lipopolysaccharide transport system across colonic epithelial cells in normal and infective rat. Drug Metab Pharmacokinet. 2004;19:33-40
23. Schroder MW, Heine H, Alexander C. et al. Lipopolysaccharide binding protein binds to triacylated and diacylated lipopeptides and mediates innate immune responses. J Immunol. 2004;173:2683-2691
24. Fubini B, Hubbard A. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) generation by silica in inflammation and fibrosis. Free Radic Biol Med. 2003;34:1507-1516
25. Gough PJ, Gordon S. The role of scavenger receptors in the innate immune system. Microbes Infect. 2000;2:305-311
26. Peiser L, Gordon S. The function of scavenger receptors expressed by macrophages and their role in the regulation of inflammation. Microbes Infect. 2001;3:149-159
Author contact
Corresponding authors: Carmen Chan: Address: Room 732, Esther Lee Building, The Chinese University of Hong Kong, Shatin, Hong Kong; Tel: +852 39436218; Email: whchanedu.hk and Judy Chan: Address: Room 816, Esther Lee Building, The Chinese University of Hong Kong, Shatin, Hong Kong; Tel: +852 39433515; Email: judychanywedu.hk

 Global reach, higher impact
Global reach, higher impact