ISSN: 1449-1907International Journal of Medical Sciences
Int J Med Sci 2010; 7(5):290-299. doi:10.7150/ijms.7.290 This issue Cite
Research Paper
1, 25-dihydroxyvitamin D3 decreases adriamycin-induced podocyte apoptosis and loss
1. Department of Pediatrics, Wuhan General Hospital of Guangzhou Command, Wuhan 430070, China
2. Department of Nephrology, Affiliated Children Hospital of Shanghai Jiaotong University, Shanghai, China
Abstract
Background: Selective proteinuria is frequently observed in glomerular diseases characterized by podocyte injury. Although, 1,25-dihydroxyvitamin D3 [1,25(OH)2D3] has potential therapeutic effects on chronic kidney diseases through decreasing podocyte loss, the mechanism underlying the beneficial effects of 1,25(OH)2D3 on podocytes remains still unknown. The present study tested the hypothesis that 1,25(OH)2D3 directly reduced podocyte apoptosis and loss.
Methods: Sprague-Dawley (SD) rats were randomly assigned into three groups: Adriamycin (ADR) group (n=15), ADR+1,25-(OH)2D3 group (n=16), and control group (n=16). Rats in ADR+1,25-(OH)2D3 group were treated with 1,25(OH)2D3 for 8 weeks. The number of podocytes and foot process width (FPW) were detected by transmission electron microscopy. The number of apoptotic podocytes per glomerulus and that of apoptotic nuclei and caspase-3 activity in cultured podocytes were determined by TUNEL staining. The average number of podocytes per glomerulus was quantified by immunohistochemistry. Expressions of p-Smad2/3, p-Smad1/5/8, Fas, Fas-Associated protein with Death Domain (FADD), Bax, and Bcl-2 proteins were examined by Western blot assay.
Results: Compared with control group, proteinuria, FPW, apoptotic podocytes, caspase-3 activity, the protein expressions of p-Smad2/3, Fas, FADD, and Bax were significantly increased, podocyte density, p-Smad1/5/8 and Bcl-2 expression were decreased in ADR group. 1,25(OH)2D3 significantly reduced proteinuria, FPW, caspase-3 activity, expressions of p-Smad2/3, Fas, FADD, and Bax and apoptosis of podocytes, but increased serum albumin, number of viable podocytes , p-Smad1/5/8 and Bcl-2 expression in ADR treated rats.
Conclusion: ADR-induced podocyte apoptosis was associated with the imbalance of p-Smad2/3, p-Smad1/5/8 the activity of caspase-3 and aberrant expressions of, Fas, FADD, Bax and Bcl-2. The beneficial effects of 1,25(OH)2D3 on podocytes may be attributable to inhibit podocyte apoptosis and the amelioration of podocytopenia.
Keywords: 1, 25-dihydroxyvitamin D3, podocyte, proteinuria
Introduction
Podocytes are highly specialized and terminally differentiated cells with limited mitotic capacity. Therefore, once lost, the regeneration of podocytes is limited. There is a growing body of literature showing that podocytopenia is a critical determinant in the development of glomerulosclerosis that leads to progressive renal failure. Podocytopenia is caused by detachment of podocytes from glomerular basement membrane (GBM) and/or apoptosis of podocytes (1). Caspase-3 is a newly characterized mammalian cysteine protease that promotes cell apoptosis in many different conditions. The most common cause of apoptosis in kidney diseases is mediated by Fas (2). Albumin overload also resulted in a dose-dependent up-regulation of Fas and Fas-associated protein with death domain (FADD) (3). Bcl-2 family which consists of both pro-apoptotic protein (e.g., Bax) and anti-apoptotic protein (e.g., Bcl-2) appears to play crucial roles in regulating the balance between apoptosis and survival (4).
It has been demonstrated that 1,25-(OH)2D3 can reduce proteinuria in active Thy1-nephritis rats (5), inhibit progressive glomerulosclerosis and decrease albuminuria in subtotally nephrectomized rats (6), and down-regulate the renin-angiotensin system (RAS) by inhibiting renin production (7). Recent reports also indicate that 1,25(OH)2D3 can decrease podocyte loss and inhibit podocyte hypertrophy in subtotally nephrectomized rats (8). In puromycin aminonucleoside nephropathy rats, podocyte injury is suppressed by 1,25(OH)2D3 via modulation of transforming growth factor-beta 1 (TGF-1)/bone morphogenetic protein-7 (BMP-7) signalling (9). Those findings clearly suggest podocytes serve as a potentially important target for vitamin D in the treatment of kidney diseases.
Recent studies have shown that podocytes undergo apoptosis in glomerular disease (10). Xiao et al. (11) demonstrated that 1,25(OH)2D3 prevented puromycin aminonucleoside-induced apoptosis of glomerular podocytes by activating the phosphatidylinositol 3-kinase/Akt-signaling pathway. Gassler et al. (12) revealed extensive apoptosis and markedly decreased number of nephrons in bcl-2 deficient mice. Schiffer et al. (13) demonstrated podocytes undergo apoptosis at early stages in the course of progressive glomerulosclerosis in TGF-β1 transgenic mice. Their results suggested a novel functional role for Smad7 as amplifier of TGF-β-induced apoptosis in podocytes and a new pathomechanism for podocyte depletion in progressive glomerulosclerosis.
In clinical practice, 1,25-(OH)2D3 is widely used in the treatment of hypocalcemia and hyperphosphatemia of patients with chronic kidney disease (CKD). However, little is known about the effects of 1,25-(OH)2D3 on podocyte apoptosis and podocytopenia. The present study aimed to investigate the effects of 1,25-(OH)2D3 on podocyte apoptosis and podocytopenia induced by adriamycin (ADR) and the potential mechanisms.
Materials and Methods
Animals and experimental design
Forty-eight male Sprague-Dawley (SD) rats were housed in a temperature-controlled room with a 12 h light/12 h dark cycle and given ad libitum access to food and water. These rats were randomly divided into three groups (n=16, per group): Adriamycin (ADR) group, ADR+1,25-(OH)2D3 group, and control group. ADR induced nephropathy was introduced by a single injection of 7.5 mg/kg ADR (0.75 mg/ml in normal saline; Sigma) via the tail vein (14). Rats in ADR+1,25-(OH)2D3 group were treated with 1,25-(OH)2D3 (3 ng·100 g body weight-1·day-1) (8) by a subcutaneous osmotic minipump for 8 weeks. One rat died in the ADR group and was excluded from the experiment. Rats in ADR group and control group were subcutaneously given normal saline of equivalent volume once daily for 8 weeks. Eight weeks later, 24-h urine samples were collected through metabolic cages followed by sacrifice of mice. The 24-h urine protein (24 h UP) was determined by colorimetric assay. Levels of serum albumin (SA), creatinine, total cholesterol (TC) and triglyeride (TG) were measured with an automatic biochemical analyzer (HITACHI 7080).
Detection of podocytes and foot process width by transmission electron microscopy
Part of renal cortex was fixed in 1.5% glutaraldehyde and 1% paraformaldehyde, dehydrated, and embedded in Spurr resin. Ultrathin sections were prepared and stained with lead citrate for transmission electron microscopy. Ten fields (three glomeruli per field) in transverse sections of each rat were randomly selected under a transmission electron microscope (×3000; Philips, Netherlands). The length of peripheral GBM was measured and the number of foot processes on GBM was counted. The mean foot process width (FPW) was calculated as follow: FPW=π/4×(∑GBM length/ ∑foot process); where ∑GBM length is the total length of GBM in one glomerulus, ∑foot process is the total number of foot process, and π/4, a correction factor, serves to correct the random orientation under which foot processes are sectioned (15).
Detection of podocyte number per glomerulus by immunohistochemistry
Renal tissues were fixed in formalin, embedded in paraffin and cut into sections followed by staining with periodic-acid schiff (PAS) reagent. The number of podocytes in 3-µm frozen sections was determined through double-staining with rabbit anti-Wilms tumor antigen-1 (WT-1) polyclonal antibody (1:50) (Santa Cruz Biotechnology, Inc., USA) and mouse anti-synaptopodin monoclonal antibody (1:50) (Biofriendship Inc., Beijing, China). Cells positive for both synaptopodin and WT-1 were counted in 30 glomeruli of transverse sections. Results were presented as average number of podocytes per glomerulus in transverse sections.
Detection of apoptotic podocytes per glomerulus by TUNEL staining
Apoptotic nuclei were detected through a transferase-mediated dUTP nick-end labeling (TUNEL) staining of kidney sections followed by counterstaining with hematoxylin and PAS, as previously described (13). TUNEL staining was performed according to the manufacturer's instructions (Biosynthesis Biotechnology Co., Ltd, Beijing). In brief, 3 µm paraffin-embedded kidney sections were deparaffinized with xylene, blocked with 3% H2O2 for 30 min, washed with PBS, permeabilized with 0.5% Triton X-100 for 10 min and then washed with PBS. Terminal deoxynucleotidyl transferase (TdT) and biotin-11-dUTP were supplemented followed by incubation for 60 min. Then, sections were washed with PBS, incubated with avidin-biotinylated horseradish peroxidase complex followed by color development with DAB. Counterstaining for nuclei, dehydration, clearing and mounting were performed. Cells with brown granules in nuclei were TUNEL positive cells and TUNEL positive podocytes on the GBM were counted in 30 glomeruli of transverse section, Results were expressed as average numbers of TUNEL-positive podocytes per glomerulus.
Detection of apoptotic podocytes nuclei and the activity of caspase-3 in cultured podocytes
Podocytes were isolated from glomeruli of SD rats and maintained in medium as previously described (16). Briefly, glomeruli were isolated by differential sieving of minced cortices, followed by digestion with collagenase and culture. Early cellular outgrowths at 5-7 days were selectively removed by a cylinder cloning technique. Cells were replated, and plates growing pure colonies were then expanded and identified. Podocytes between passages 8 and 10 were used for experiments. Terminal deoxynucleotidedyl transferase (TUNEL) assay (ApoAlert Assay Kit; BD Biosciences Clontech, San Jose, CA) was performed as previously described (17). Briefly, cells were grown on cover slips, fixed with 4% formaldehyde for 30 min at 4°C, permeabilized with 0.2% Triton X-100 for 15 min at 4°C, and incubated with a mixture of nucleotides and TdT enzyme for 60 min at 37°C in humidified air in dark. Reaction was terminated with 2X SSC, and the cover slips were mounted on glass slides. Apoptotic nuclei were detected under fluorescence microscope, and cells with characteristic morphology of apoptosis, including nuclear fragmentation, nuclear condensation, and intensely fluorescent nuclei were counted. A total of 100 cells were counted for each sample. Results were expressed as the percentage of TUNEL positive nuclei in all nuclei visualized by phase contrast microscope.
Detection of caspase-3 activity was performed with BD ApoAlert Caspase Colorimetric Assay Kit (BD Biosciences, Palo Alto, CA), according to the manufacturer's instructions. This assay uses the spectrophotometric detection of the chromophore p-nitroaniline after its cleavage by caspase-3 from the labeled caspase-specific substrates. Adherent cells together with floating cells in the medium were collected, and lysed with lysis buffer included in the kit. After the cell lysate was incubated with caspase-3 substrate DEVD-p-nitroaniline (final concentration, 50 µM) at 37°C for 1 h, the absorbance (caspase-3 activity) was measured by at 405 nm with a microplate reader (Packard Spectracount).
Western blot assay
After removal of kidney capsule, the outer cortex was minced in 1- to 2-mm fragments, passed through consecutive 80- and 120-mesh sieves, and recovered from the 200-mesh sieve. After centrifugation, supernatant was abandoned and glomeruli were obtained. Glomeruli were lysed with RIPA buffer (0.1% sodium dodecyl sulfate, 1% sodium deoxycholate, 1% Triton X-100, 150 mM NaCl, 10 mM EDTA, and 25 mM Tris·HCl, pH 7.2) and protease inhibitors (1 mM phenylmethylsulfonyl fluoride, 1 µg/ml leupeptin, and 1 µg/ml pepstatin) followed by ultrasonic homogenation on ice. Lysates were centrifuged at 12,000 g for 10 min, and supernatants containing proteins were collected. Then, 75 μg of total proteins were subjected to SDS-polyacrylamide gel electrophoresis and transferred onto nitrocellulose membranes. After being blocked in 5% low-fat milk, the membranes were incubated with anti-p-smad2/3(1:500), anti-p-Smad1/5/8(1:500), anti-Fas (1:1000), anti-FADD (1:100), anti-Bax (1:500) or anti-Bcl2 (1:500) antibodies (Santa Cruz Biotechnology, CA). Subsequently, the membranes were rinsed in Tris-buffered saline containing 0.02% Tween-20 and incubated with horseradish peroxidase conjugated to anti-rabbit or mouse IgG antibody (1:4000, Santa Cruz). After washing, the membranes were developed using an enhanced chemiluminescence reagent (Santa Cruz), and the specific protein bands were scanned and quantitated. Expression of specific protein was normalized by that of GAPDH (36 kDa) as an endogenous reference.
Statistical Analyses
Data were presented as means ± standard deviation (SD). Results were analyzed using the Kruskal-Wallis non-parametric test for multiple comparisons followed by Mann-Whitney U-test. A value of P<0.05 was considered statistically significant. Correlations were assessed by Pearson correlation analysis. Statistical analyses were performed with SPSS version 11.0.
Results
Changes in 24 h UP, SA, TC and TG
The levels of 24 h UP, TC and TG in ADR group were significantly higher than those in control group, and the SA level in ADR group was markedly lower than that in control group. As compared with ADR group, the levels of 24 UP, TC and TG were dramatically decreased and SA increased after 1,25-(OH)2D3 treatment. Results are shown in Table 1.
24 h UP, SA, TC and TG levels in different groups
| Group | n | 24 h UP (mg) | SA (g/L) | TC (mmol/L) | TG (mmol/L) |
|---|---|---|---|---|---|
| control | 16 | 1.01±0.29 | 35.10±3.33 | 0.43±0.11 | 0.89±0.21 |
| ADR | 15 | 57.05±9.28## | 18.13±3.06## | 8.06±2.00## | 8.17±1.97## |
| ADR +1,25(OH)2D3 | 16 | 37.69±5.71## △△ | 23.25±3.01##△△ | 5.09±1.42## △△ | 5.48±1.30##△△ |
Data are means ± SD. 24 h UP, 24-h urine protein; SA, serum albumin; TC, total cholesterol; TG, triglyeride.
##P < 0.01, #P < 0.01 vs. control group.
△△P<0.01 vs. ADR group.
The number of podocytes and FPW
Compared with rats in control group, the number of podocytes was decreased and FPW increased in ADR group. In addition, after 1,25-(OH)2D3 treatment, the number of podocytes and FPW in 1,25-(OH)2D3 group was higher and smaller than those in ADR group, respectively (Table 2 and Figure 1).
The number of podocytes and FPW in different groups
| Group | n | Podocytes | FPW (nm) |
|---|---|---|---|
| control | 16 | 5.46±0.51 | 271.38±52.48 |
| ADR | 15 | 3.35±0.14## | 806.13±120.19## |
| ADR +1,25(OH)2D3 | 16 | 4.44±0.23## △△ | 401.13±52.48## △△ |
Data are means ± SD. FPW, foot process width.
##P < 0.01 vs. control group.
△△P<0.01 vs. ADR group.
Effect of 1,25(OH)2D3 treatment on podocyte density
In the control group, the number of double-positive podocytes was 11.55±1.69 cells/glomerulus. In the ADR group, the number of double-positive podocytes (7.97±1.62 cells/glomerulus) was markedly lower than that in 1,25(OH)2D3 group (10.07±1.54 cells/glomerulus) (P <0.01) (Figure 2).
Podocyte apoptosis
TUNEL staining was performed to detect the number of apoptotic podocytes per glomerulus. Significant difference in the number of TUNEL-positive podocytes was found between ADR group and control group (2.50±0.77 vs. 0.19±0.40, P<0.01). After 8 weeks of treatment with 1,25(OH)2D3, the number of apoptotic podocytes was significantly higher than that in ADR group (1.19±0.37 vs. 2.50±0.77, P<0.01), indicating marked improvement in podocyte apoptosis after 1,25(OH)2D3 treatment (Figure 3).
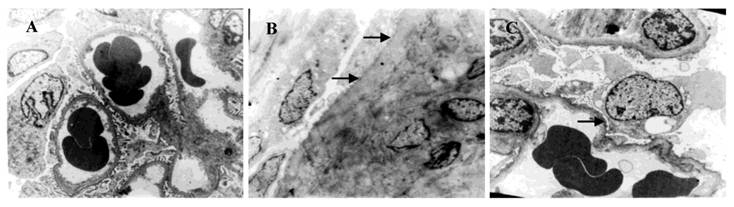
Ultrastructure of podocytes under a transmission electron microscope (×3000). A: control group; B: ADR group and C: 1,25(OH)2D3 group. The foot processes in the control group were intact. Fusion and disappearance of foot processes (arrow) were noted in the ADR group. Fusion and disappearance of foot processes were improved in 1,25(OH)2D3 group when compared with the ADR group.
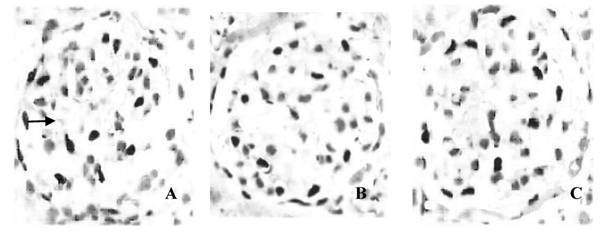
Podocytes in different groups under a light microscope (×200). Immunohistochemistry was performed to evaluate the number of podocytes. A: control group; B: ADR group and C: 1,25(OH)2D3 group. The average number of double-positive podocytes (arrow) was 11.55±1.69 cells/glomerulus in the control group, and 7.97±1.62 cells/glomerulus in the ADR group. Statistical analysis showed the average number of podocytes per glomerulus was higher in the 1,25(OH)2D3 group (10.07±1.54 cells/glomerulus) than that in the ADR group.
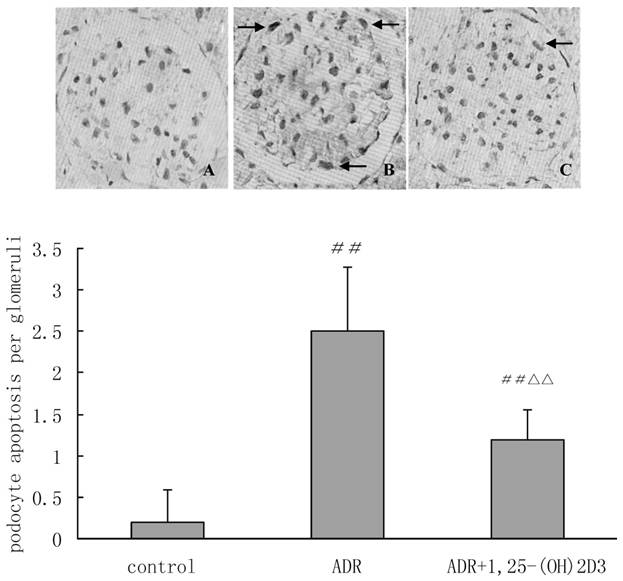
Apoptotic podocytes in different groups (×400). TUNEL staining was performed to detect the number of apoptotic podocytes per glomerulus. A: control group; B: ADR group and C: 1,25(OH)2D3 group. The average number of TUNEL-positive podocytes (arrows) was 0.19±0.40 cells/glomerulus in the control group and 2.50±0.77 cells/glomerulus in the ADR group. Statistical analysis showed the number of TUNEL-positive podocytes was lower in 1,25(OH)2D3-treated group (1.19±0.37 cells/glomerulus) than in the ADR group. ##P < 0.01 vs. control group. △△P<0.01 vs. ADR group.
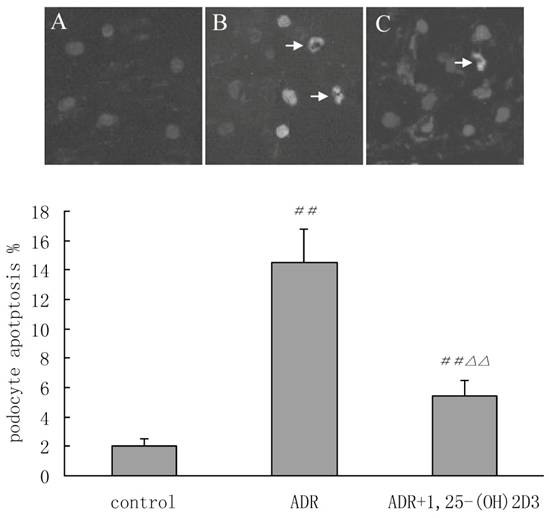
TUNEL staining was conducted in cultured podocytes treated with 1,25(OH)2D3. Representative photographs of apoptotic podocytes from fluorescence microscopy were shown in Figure 4. In the control group, TUNEL positive podocytes were almost absent, and the morphology of necrotic cells was also not observed. The arrows revealed nuclear condensation and fragmentation, characteristics of apoptosis in ADR group. Less apoptotic podocytes in 1,25(OH)2D3 group was found when compared with ADR group. These results further confirmed that 1,25(OH)2D3 could improve podocyte apoptosis.
Detection of caspase-3 activity
The caspase-3 activity was 0.13±0.05 OD405/mg protein in control group, 0.42±0.11 OD405/mg protein in ADR group and 0.33±0.09 OD405/mg protein in 1,25(OH)2D3 group. The caspase-3 activity in ADR group was 3.23 fold higher than that in control group (P < 0.01), and after 8 weeks of 1,25(OH)2D3 treatment, caspase-3 activity was down-regulated by 21.4% (P < 0.05 versus ADR group).
Western Blot
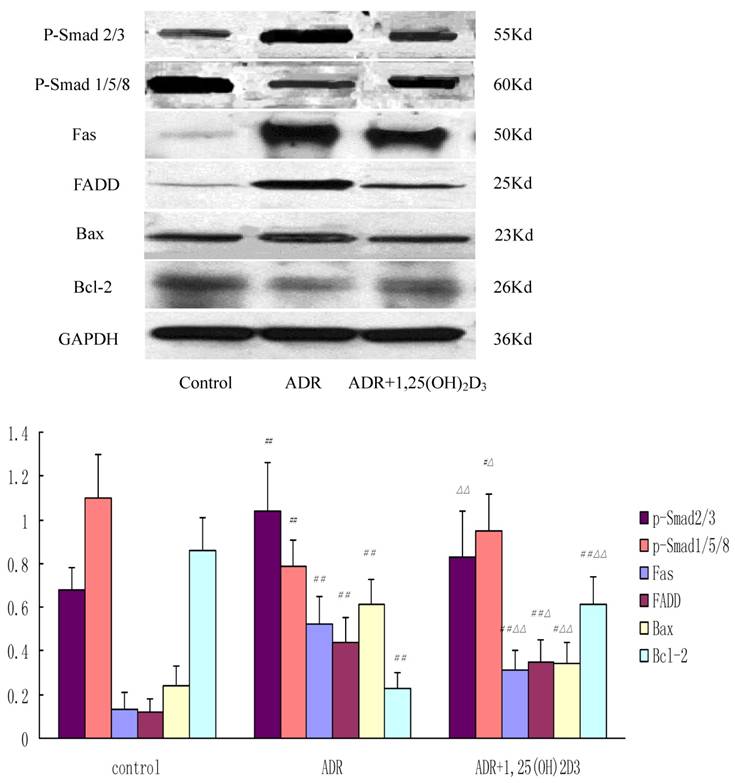
The specific protein bands of p-Smad2/3, p-Smad1/5/8, Fas, FADD, Bax, Bcl-2, and GAPDH were identified as 55 kDa, 60 kDa, 50 kDa, 25 kDa, 23 kDa, 26 kDa and 36 kDa, respectively. The expressions of p-Smad2/3, p-Smad1/5/8, Fas, FADD, Bax, and Bcl-2 were 0.68±0.10, 1.10±0.20, 0.12±0.08, 0.12±0.06, 0.24±0.09, and 0.86 ± 0.15, respectively, in the control group, 1.04±0.22, 0.79±0.12, 0.52±0.13, 0.44±0.11, 0.61±0.12, and 0.23 ± 0.07, respectively, in the ADR group, and 0.83±0.21, 0.95±0.17, 0.31±0.09, 0.35±0.10, 0.34±0.10, and 0.61±0.13, respectively, in the 1,25(OH)2D3-treated group. The expressions of p-Smad2/3, Fas, FADD, and Bax were significantly higher and p-Smad1/5/8, Bcl-2 expression lower in the ADR group than those in the control group. The decrease in the expressions of p-Smad2/3, Fas, FADD, Bax, and Bcl-2 and increase in p-Smad1/5/8, Bcl-2 expression were observed in 1,25-(OH)2D3 group when compared with the ADR group (Figure 5).
Tunel-positive nuclei in cultured podocytes (arrows) (×400). Podocytes from passages 8 to 10 were used for Tunel staining and all nuclei visualized under a phase contrast microscope. The apoptotic nuclei (arrows identify) underwent nuclear fragmentation and condensation, characteristics of apoptosis. A: control group; B: ADR group and C: 1,25(OH)2D3 group. The percentage of TUNEL-positive nuclei was 2±0.5% in the control group and 14.5±2.3% in the ADR group. The 1,25(OH)2D3 treated group had lower number (5.4±1.1%) of TUNEL-positive nuclei in cultured podocytes than ADR group did. ##P < 0.01 vs. control group; △△P < 0.01, vs. ADR group.
Western blot assay of expressions of p-Smad2/3, p-Smad1/5/8, Fas, FADD, Bax, and Bcl-2 in different groups. Compared with the control group, the ADR group had higher expressions of p-Smad2/3, Fas, FADD, and Bax and lower p-Smad1/5/8, Bcl-2 expression. In addition, compared with ADR group, the expressions of p-Smad1/5/8, Fas, FADD, Bax, and Bcl-2 were down-regulated and p-Smad2/3, Bcl-2 expression was up-regulated after 1,25(OH)2D3 treatment. Data were expressed as means ± SD. ##P < 0.01, #P < 0.05 vs. control group; △△P < 0.01, △P < 0.05 vs. ADR group.
Discussion
Podocytopenia is closely associated with the development of glomerulosclerosis in animal and human glomerular diseases. Kriz et al. (18) proposed that podocytopenia as a result of apoptosis and/or detachment of podocytes from GBM and the inability of podocytes to replicate could result in glomerulosclerosis. Podocyte apoptosis is a major factor inducing podocytopenia (19). In murine type 1 and type 2 diabetic models, podocyte apoptosis precedes podocyte depletion and urinary albumin excretion (UAE), suggesting that podocyte apoptosis/depletion represents a novel early pathomechanism leading to diabetic nephropathy (20).
1,25-(OH)2D3 can act as an immunomodulator and may be beneficial for a number of autoimmune diseases (21). The beneficial effect of active vitamin D on glomerular structures may lead to proteinuria reduction in several animal models (22, 23) and human kidney diseases (24, 25). Moreover, 1,25-(OH)2D3 can decrease podocytopenia and podocyte hypertrophy also contributing to improved albuminuria and glomerulosclerosis (8). In our study, the number of apoptotic podocytes was determined by TUNEL assay in vivo and in vitro, and consistent results were obtained that 1,25-(OH)2D3 significantly protected podocytes from ADR induced apoptosis. Of interest, 1,25-(OH)2D3 and its metabolites were previously found to induce apoptosis of embryonic renal cells (26), breast cancer cells (27, 28), ovarian cancer cells (29), and prostate cancer cells (30). In contrast, some studies also showed 1,25-(OH)2D3 could inhibit apoptosis of other cell types including human leukemic cells (31) and murine fibroblast cells (32).
To explore the potential mechanisms of ADR-induced podocyte apoptosis and the mechanisms underlying the anti-apoptotic effect of 1,25(OH)2D3, the expressions of apoptosis related proteins including Fas, FADD, Bax and Bcl-2 and caspase-3 activity were detected. Proteinuria can cause tubular cell apoptosis which is associated with activation of Fas-FADD-caspase 8 pathway (33). Bcl-2 can bind Bax to exert its effects. Therefore, over-expression of Bcl-2 can inactivate Bax suppressing apoptosis (34), Qiu et al.(35) showed that over-expression of Bcl-2 in podocytes may exert protective effects on glomerular lesions in progressive IgA nephropathy through affecting the Bax/Bcl-2 ratio and finally limiting glomerular cell apoptosis. Our study demonstrates the protective effect of 1,25-(OH)2D3 on podocyte apoptosis by down-regulating Fas, FADD and Bax and up-regulating Bcl-2, which is of great importance in preventing podocytopenia.
TGF-β1 and BMP-7 belong to transforming growth factor superfamily. In the podocytes, proteins of Smads family play an important role in the signal transduction from cell membrane to nucleus. TGF-β1 can activate smad2/3 mediating podocyte apoptosis. BMP-7 can activate PI3K/Akt /Smad l/5/8 pretecting podocytes (36). In the present study, 1,25(OH)2D3 treatment decreased the p-smad 2/3 expression and increased p-Smad l/5/8 expression in the ADR treated rats, which suggested 1,25(OH)2D3 could suppress TGF-β1 signal pathway and caspase-3 activity, inhibit expressions of apoptosis related proteins (Fas, FADD and Bax), increase anti-apoptotic protein expression and activate BMP-7 signal pathway. These processes finally alleviate podocyte apoptosis and promote podocyte survival. Our results were consistent with Xiao et al reported (9, 11) in which the protective effects of 1,25(OH)2D3 on podocytes were mediated by TGF-β1/BMP-7 and related to the activation of PI3K/Akt leading to suppression of podocyte apoptosis.
Angiotensin II, TGF-β1 (37), and reactive oxygen species (ROS) (38) have been shown to initiate podocyte apoptosis. Fornoni et al. (39) reported that hepatocyte growth factor (HGF) could protect podocytes from cyclosporine A-induced apoptosis. 1,25(OH)2D3 exerts the protective effect on kidney in multiple ways such as anti-inflammation, suppression of renin production, and reduction of fibrogenic cytokine production by regulating Smad3 and TGF-β pathways and inducing HGF mRNA expression and HGF secretion in renal interstitial fibroblasts (40). So it was possible that 1,25-(OH)2D3 could exert anti-apoptotic effect and prevent podocytopenia. Our results were partially consistent with a recent study in which dexamethasone could prevent puromycin aminonucleoside induced podocyte apoptosis (41). Podocytopenia is usually found in type 2 diabetic nephropathy and related to increased proteinuria (42).
In summary, we demonstrated that 1,25-(OH)2D3 could inhibit podocyte apoptosis through changing the imbalance of p-Smad2/3/p-Smad1/5/8, down-regulating caspase-3 activity, and expressions of Fas, FADD and Bax and up-regulating Bcl-2 expression, which maybe finally contributed to improved podocytopenia. These findings provides an experimental evidence for 1,25(OH)2D3 treatment of nephropathy characterized by podocyte injury.
Acknowledgements
We thank Dr Zhao LS (Department of Endocrinology, Wuhan General Hospital) and Qi ML (Department of Pathology, Wuhan General Hospital) for their kind help.
Conflict of Interest
The authors have declared that no conflict of interest exists.
References
1. Mundel P, Shankland SJ. Podocyte biology and response to injury. J Am Soc Nephrol. 2002;13:3005-3015
2. Ross MJ, Martinka S, D'Agati VD. et al. NF-kappaB regulates Fas-mediated apoptosis in HIV-associated nephropathy. J Am Soc Nephrol. 2005;16(8):2403-2411
3. Erkan E, De Leon M, Devarajan P. Albumin overload induces apoptosis in LLC-PK(1) cells. Am J Physiol Renal Physiol. 2001;280(6):F1107-1114
4. Tan C, Dlugosz PJ, Peng J. et al. Auto-activation of the apoptosis protein Bax increases mitochondrial membrane permeability and is inhibited by Bcl-2. J Biol Chem. 2006;281(21):14764-14775
5. Makibayashi K, Tatematsu M, Hirata M. et al. A vitamin D analog ameliorates glomerular injury on rat glomerulonephritis. Am J Pathol. 2001;158:1733-1741
6. Schwarz U, Amann K, Orth SR. et al. Effect of 1,25 (OH)2 vitamin D3 on glomerulosclerosis in subtotally nephrectomized rats. Kidney Int. 1998;53:1696-1705
7. Li YC, Kong J, Wei M. et al. 1,25-Dihydroxyvitamin D3 is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest. 2002;110:229-238
8. Kuhlmann A, Haas CS, Gross ML. et al. 1,25-Dihydroxyvitamin D3 decreases podocyte loss and podocyte hypertrophy in the subtotally nephrectomized rat. Am J Physiol Renal Physiol. 2004;286:F526-F533
9. Xiao HQ, Shi W, Liu SX. et al. Podocyte injury is suppressed by 1,25-dihydroxyvitamin D via modulation of transforming growth factor-beta 1/bone morphogenetic protein-7 signalling in puromycin aminonucleoside nephropathy rats. Clin Exp Pharmacol Physiol. 2009;36(7):682-689
10. Kim Y-H, Goyal M, Kurnit D. et al. Podocyte depletion and glomerulosclerosis have a direct relationship in the PAN-treated rat. Kidney Int. 2001;60:957-968
11. Xiao H, Shi W, Liu S. et al. 1,25-Dihydroxyvitamin D(3) prevents puromycin aminonucleoside-induced apoptosis of glomerular podocytes by activating the phosphatidylinositol 3-kinase/Akt-signaling pathway. Am J Nephrol. 2009;30(1):34-43
12. Gassler N, Elger M, Inoue D. et al. Oligonephronia, not exuberant apoptosis, accounts for the development of glomerulosclerosis in the bcl-2 knockout mouse. Nephrol. Dial. Transplant. 1998;13:2509- 2518
13. Schiffer M, Bitzer m, Roberts IS. et al. Apoptosis in podocytes induced by TGF-beta and Smad7. J Clin Invest. 2001;108:807-816
14. Deschênes G, Doucet A. Collecting duct (Na+/K+)-ATPase activity is correlated with urinary sodium excretion in rat nephrotic syndromes. J Am Soc Nephrol. 2000;11(4):604-615
15. Koop K, Eikmans M, Baelde HJ. et al. Expression of podocyte-associated molecules in acquired human kidney diseases. J Am Soc Nephrol. 2003;14(8):2063-2071
16. Sanwal V, Pandya M Bhaskaran M. et al. Puromycin aminonucleoside induces glomerular epithelial cell apoptosis. Exp Mol Pathol. 2001;70:54-64
17. Park MS, De Leon M, Devarajan P. Cisplatin induces apoptosis in LLC-PK1 cells via activation of mitochondrial pathways. J Am Soc Nephrol. 2002;13(4):858-865
18. Kriz W, Gretz N, Lemley KV. Progression of glomerular diseases: Is the podocyte the culprit?. Kidney Int. 1998;54:687-697
19. Meyer TW, Bennett PH, Nelson RG. Podocyte number predicts long-term urinary albumin excretion in Pima Indians with type II diabetes and microalbuminuria. Diabetologia. 1999;42:1341-1344
20. Susztak K, Raff AC, Schiffer M. et al. Glucose-induced reactive oxygen species cause apoptosis of podocytes and podocyte depletion at the onset of diabetic nephropathy. Diabetes. 2006;55(1):225-233
21. Boonstra A, Barrat FJ, Crain C. et al. 1alpha,25-Dihydroxyvitamin d3 has a direct effect on naive CD4(+) T cells to enhance the development of Th2 cells. J Immunol. 2001;167(9):4974-4980
22. Hirata M, Makibayashi K, Katsumata K. et al. 22-Oxacalcitriol prevents progressive glomerulosclerosis without adversely affecting calcium and phosphorus metabolism in subtotally nephrectomized rats. Nephrol Dial Transplant. 2002;17(12):2132-2137
23. Panichi V, Migliori M, Taccola D. et al. Effects of 1,25(OH)2D3 in experimental mesangial proliferative nephritis in rats. Kidney Int. 2001;60:87-95
24. Agarwal R, Acharya M, Tian J. et al. Antiproteinuric effect of oral paricalcitol in chronic kidney disease. Kidney Int. 2005;68(6):2823-2828
25. Alborzi P, Patel NA, Peterson C. et al. Paricalcitol reduces albuminuria and inflammation in chronic kidney disease: a randomized double-blind pilot trial. Hypertension. 2008;52(2):249-255
26. Wagner KD, Wagner N, Sukhatme VP. et al. Activation of vitamin D receptor by the Wilms' tumor gene product mediates apoptosis of renal cells. J Am Soc Nephrol. 2001;12(6):1188-1196
27. Welsh JE. Induction of apoptosis in breast cancer cells in response to vitamin D and anti-estrogens. Biochem Cell Biol. 1994;72:537-545
28. Simboli-Campbell M, Narvaez CJ, VanWeelden K. et al. Comparative effects of 1,25-(OH)2D3 and EB1089 on cell cycle kinetics and apoptosis in MCF-7 cells. Breast Cancer Res Treat. 1997;42:31-41
29. Jiang F, Bao J, Li P. et al. Induction of ovarian cancer cell apoptosis by 1,25-dihydroxyvitamin D3 through the down-regulation of telomerase. J Biol Chem. 2004;279(51):53213-5321
30. Meral G, Shinichi K, John C. Apoptosis induction by 1alpha,25-dihydroxyvitamin D3 in prostate cancer. Mol Cancer Ther. 2002;1(9):667-77
31. Wu YL, Jiang XR, Lillington DM. et al. 1,25-Dihydroxyvitamin D3 protects human leukemic cells from tumor necrosis factor-induced apoptosis via inactivation of cytosolic phospholipase A2. Cancer Res. 1998;58(4):633-640
32. Adams LS, Teegarden D. 1,25-dihydroxycholecalciferol inhibits apoptosis in C3H10T1/2 murine fibroblast cells through activation of nuclear factor κB. J Nutr. 2004;134(11):2948-2952
33. Abbate M, Zoja C, Remuzzi G. How does proteinuria cause progressive renal damage?. J Am Soc Nephrol. 2006;17(11):2974-2984
34. Liu JJ, Huang RW, Lin DJ. et al. Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-γ ligands induces apoptosis on human myeloid leukemia cells in vitro. Ann Oncol. 2005;16(3):455-459
35. Qiu LQ, Sinniah R, I-Hong Hsu S. Downregulation of Bcl-2 by podocytes is associated with progressive glomerular injury and clinical indices of poor renal prognosis in human IgA nephropathy. J Am Soc Nephrol. 2004;15(1):79-90
36. Boor P, Sebeková K, Ostendorf T. et al. Treatment targets in renal fibrosis. Nephrol Dial Transplant. 2007;22(12):3391-3407
37. Ding G, Reddy K, Kapasi AA. et al. Angiotensin II induces apoptosis in rat glomerular epithelial cells. Am J Physiol Renal Physiol. 2002;283(1):F173-F180
38. Wu DT, Bitzer M, Ju W. et al. TGF-beta concentration specifies differential signaling profiles of growth arrest/differentiation and apoptosis in podocytes. J Am Soc Nephrol. 2005;16(11):3211-21
39. Fornoni A, Li H, Foschi A. et al. Hepatocyte growth factor, but not insulin-like growth factor I, protects podocytes against cyclosporin A-induced apoptosis. Am J Pathol. 2001;158:275-280
40. Li Y, Spataro BC, Yang J. et al. 1,25-Dihydroxyvitamin D3 inhibits renal interstitial myofibroblast activation by inducing hepatocyte growth factor expression. Kidney Int. 2005;68:1500-1510
41. Wada T, Pippin JW, Marshall CB. et al. Dexamethasone prevents podocyte apoptosis induced by puromycin aminonucleoside: role of p53 and Bcl-2-related family proteins. J Am Soc Nephrol. 2005;16(9):2615-25
42. White KE, Bilous RW. Diabiopsies Study Group. Structural alterations to the podocyte are related to proteinuria in type 2 diabetic patients. Nephrol Dial Transplant. 2004;19(6):1437-1440
Author contact
![]() Corresponding author: Jian Yu, Department of Pediatrics, Wuhan General Hospital of Guangzhou Command, Wuhan 430070, China. Email: docjack99com
Corresponding author: Jian Yu, Department of Pediatrics, Wuhan General Hospital of Guangzhou Command, Wuhan 430070, China. Email: docjack99com
Received 2010-5-26
Accepted 2010-8-17
Published 2010-8-24
